Beyond Murray's Law: Non-Universal Branching Exponents from Vessel-Wall Metabolic Costs
Murray's cubic branching law ($α=3$) predicts a universal diameter scaling exponent for all hierarchical transport networks, yet arterial trees yield $α\sim 2.7-2.9$. We show that this discrepancy has a structural origin: Murray's universality is an …
Authors: Riccardo Marchesi
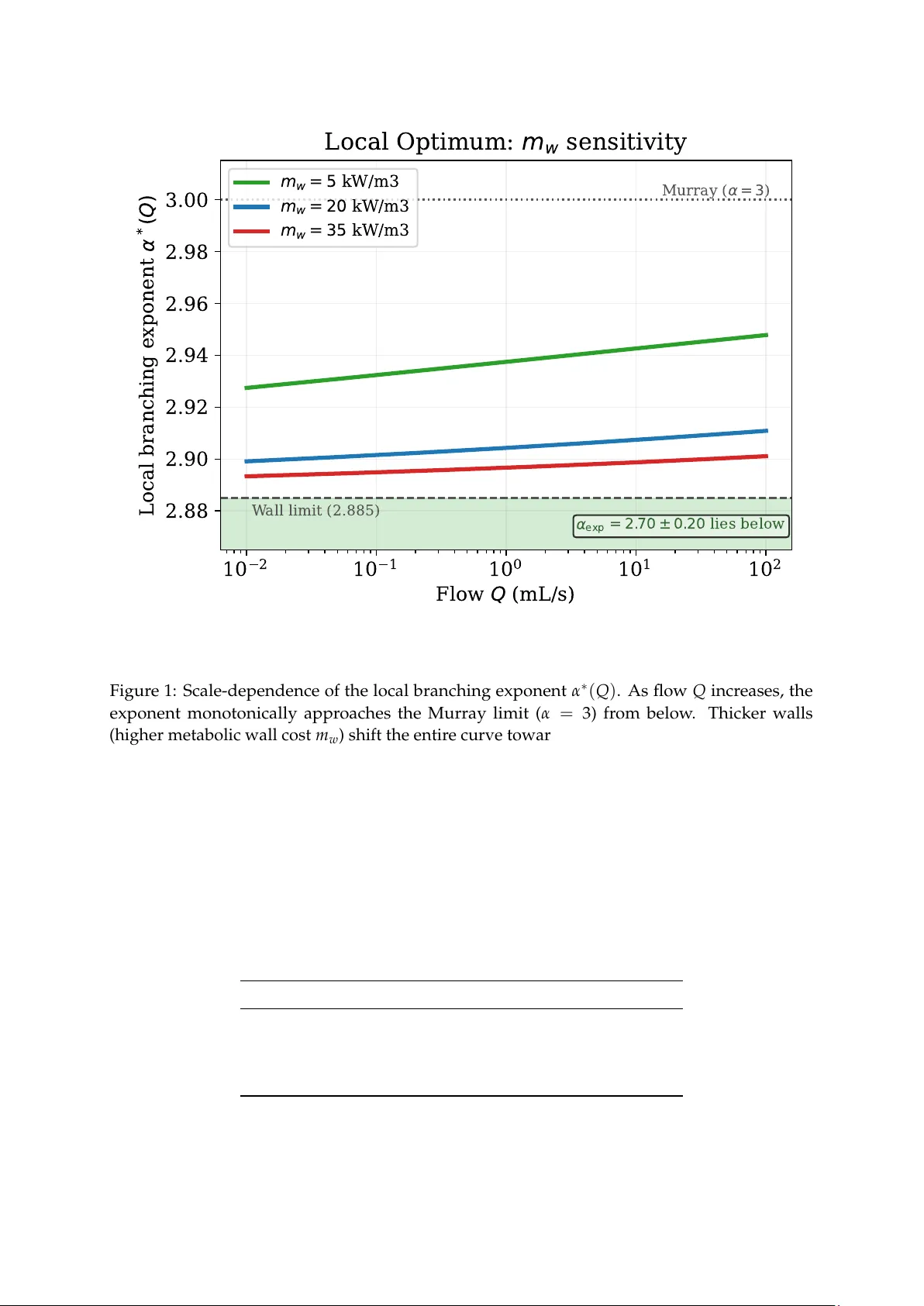
Beyond Murray’ s Law: Non-Universal Branching Exponents from V essel-W all Metabolic Costs Riccardo Mar chesi University of Pavia March 31, 2026 Abstract Murray’s cubic branching law ( α = 3) predicts a universal diameter scaling exponent for all hierarchical transport networks, yet arterial trees consistently yield α ∼ 2.7–2.9. W e show that this discrepancy has a structural origin: Murray’s universality is an artifact of his cost function’s homogeneity , not a property of biological networks. Incorporating the empirical vessel-wall thickness law h ( r ) = c 0 r p ( p ≈ 0.77 acr oss mammalian species) introduces a thir d metabolic cost term ∝ r 1 + p that renders the cost function inhomogeneous with incommensurate scaling exponents. By Cauchy’s functional equation, homogeneity is both necessary and sufficient for a universal branching exponent to exist; its absence rigorously implies non-universality , and Murray’s cubic law is thereby identified as a singular degeneracy of the cost-function family rather than a general biological principle. W e prove that the resulting scale-dependent exponent satisfies the strict bounds ( 5 + p ) / 2 < α ∗ ( Q ) < 3 independently of flow asymmetry (Theorem 4 , Corollary 5 ), and that Murray’s law is the unique member of this cost-function family admitting a universal exponent (Corollary 6 ). The static wall-tissue mechanism rigor ously bounds the symmetric bifur cation exponent to α t ∈ [ 2.90, 2.94 ] from independently measured parameters, representing a first-order symmetry breaking from Murray’s law that narr ows the empirical gap by one-thir d. The remaining discrepancy with the cardiovascular mean ( α exp ≈ 2.70) is not a model failure but a mathematical necessity that signals the independent contribution of pulsatile wave dynamics, necessitating a unified variational treatment. Additionally , the wall cost strictly breaks Murray’s topological degeneracy , bounding the optimal branching number to small finite integers and excluding star-like topologies; binary bifurcation emer ges as the physiologically selected minimum under steric constraints (Theorem 12 ). 1 1 Introduction The branching geometry of hierarchical transport networks—vascular tr ees, plant xylem, river drainage basins, engineered pipe manifolds—is commonly characterized by the exponent α in the diameter scaling law d α 0 = ∑ i d α i , (1) where d 0 is the parent diameter and d i are the daughter diameters (since d = 2 r , the exponent is the same in the radius convention used below). Murray [ 1 ] derived α = 3 by minimizing the sum of viscous dissipation and blood-volume metabolic cost over a single vessel. This r esult is elegant and well-supported for pure-flow networks (pulmonary airways, river networks, microfluidic channels), but empirical measurements of arterial trees consistently give α ≈ 2.7–2.9 [ 2 , 3 ], a systematic gap that has resisted a parameter -free explanation. The departure of empirical exponents fr om Murray’s ideal α = 3 is traditionally attributed to pulsatile wave dynamics. In the cardiovascular system, optimal wave pr opagation requir es impedance matching at bifurcations [ 4 ], which favors α = 2 (for acoustic-like waves) or α = 5 / 2 (for purely elastic walls). While unifying wave reflection costs and viscous dissipation into a single network-level Lagrangian provides a compelling physical pictur e for intermediate expo- nents, it typically r equires complex assumptions about the operational duty cycle of pulsatile versus steady flow . In this Article, we demonstrate that one does not need to invoke pulsatile wave dynamics to explain α < 3. Even in the purely static regime, simply completing Murray’s original metabolic cost function to include the structural tissue of the vessel wall is sufficient to rigorously br eak the universality of the cubic law . Recent work by Bennett [ 5 ] develops a variational framework for branching networks based on two-term cost functions: starting from a scale-free homogeneity condition on a Q 2 r − p + b r m ledger , Bennett derives generalized Murray scaling α = ( m + p ) / 2, a Y oung–Herring-type junction angle balance, concave Gilbert-type flux costs, and a single-index (rigidity index χ ) unification showing that all thr ee are contr olled by one dimensionless ratio [ 5 ]. For Poiseuille flow with volume-priced maintenance ( m = 2, p = 4), this recovers α = 3; for surface-priced maintenance ( m = 1), α = 5 / 2. The two-term limit of our Theorem 3 ( α = ( 4 + γ ) / 2 with the maintenance exponent γ physically grounded) coincides with the generalized Murray scaling independently established by Bennett thr ough abstract scale-free homogeneity , pr oviding convergent validation from two distinct theoretical starting points. However , the structural exponent m in Bennett’s framework is a phenomenological parameter whose value is not predicted fr om independently measurable tissue quantities. W e keep Murray’s energy-minimization objective and ask what happens when a third biological term is added, structurally breaking the Euler homogeneity (inhomogeneity with incommensurate scaling exponents) that underlies Bennett’s two-term class. The key observation is that vessel walls—smooth muscle cells, extracellular matrix, active tension—have a non- negligible metabolic cost that scales differently fr om blood volume. Histological measurements across species establish h ( r ) = c 0 r p with p ≈ 0.77 [ 6 , 4 , 7 ], introducing a third cost term ∝ r 1 + p with exponent strictly between 1 and 2. W e emphasize that this static mechanism r epresents a first-or der symmetry breaking fr om Murray’s law . While it narrows the gap to empirical data by one-thir d, the remaining discrepancy with the car diovascular mean ( α ≈ 2.70) is not a failure of the model but a mathematical necessity: the static wall-tissue mechanism and pulsatile wave dynamics ar e complementary , non-overlapping contributions to the full architectural optimum. The resulting thr ee-term cost function leads to several rigorous r esults, the most important of which are: Murray’s law is the unique cost function of this family that produces a universal 2 branching exponent (Corollary 6 ), the three-term case predicts a scale-dependent exponent α ∗ ( Q ) bounded strictly below 3 (Theorem 4 ), and the wall cost rigorously breaks Murray’s structural degeneracy to uniquely select N = 2 (bifurcation) as the optimal branching topology (Theorem 12 ). 2 Setup The metabolic cost per unit length of a vessel of radius r carrying volumetric flow Q has three physically distinct components. The wall tissue term is derived by integrating the volumetric metabolic rate m w over the wall cross-section: for a cylindrical vessel with thickness h ( r ) = c 0 r p , the thin-wall appr oximation ( h ≪ r , satisfied for h / r ≈ 0.18 at r = 1.5 mm ) gives a wall cr oss- sectional area 2 π r h ( r ) , yielding a cost per unit length Φ wall = 2 π m w c 0 r 1 + p . The full cost is therefor e: Φ ( r , Q ) = 8 µ Q 2 π r 4 | { z } viscous + b π r 2 | { z } blood volume + 2 π m w c 0 r 1 + p | { z } wall tissue , (2) where µ is dynamic viscosity , b is the metabolic cost per unit blood volume, m w is the metabolic rate of wall tissue, and h ( r ) = c 0 r p is wall thickness. This derivation assumes active smooth- muscle metabolism dominates over passive structural cost, consistent with m w ∈ [ 5, 35 ] kW m − 3 [ 8 ]. In compact form, Φ ( r , Q ) = A ( Q ) r − 4 + B r 2 + C r 1 + p , (3) with A ( Q ) = 8 µ Q 2 / π ∝ Q 2 , B = b π , and C = 2 π m w c 0 , all positive. Empirical inputs (stated, not derived). The viscous dissipation term assumes fully developed Poiseuille (laminar) flow; for cor onary arteries, Re ∼ 200–500, well within the laminar r egime. The wall-thickness law h ( r ) = c 0 r p is taken from published histological measurements: c 0 = 0.041, p = 0.77 [ 6 , 4 , 7 ]. Here c 0 carries units m 1 − p so that h ( r ) is expr essed in metres when r is in metres; the numerical value c 0 = 0.041 corresponds to h ≈ 274 µ m at r = 1.5 mm , consistent with histological data. All other parameters are drawn from independent sources: µ = 3.5 mPa s [ 9 ], b = 1500 W/m 3 [ 1 , 10 ], m w ∈ [ 5, 35 ] kW/m 3 [ 8 ]. No parameter is fitted to morphometric data. Crucially , as demonstrated via sensitivity analysis in the unified framework, the topological optimum α ∗ is structurally insensitive ( | S c 0 | < 0.01) to the exact absolute value of the histological pre-factor c 0 . The exponent prediction depends critically only on the scaling exponent p , ensuring the r esult is not an artifact of numerical fitting. Womersley pulsatility . Proximal pulsatile flow at W o > 1 modifies the viscous dissipation term. Since W o = r p ω ρ / µ depends explicitly on r , the corrected dissipation reads F ( W o ( r )) · A ( Q ) · r − 4 , and the optimality condition ∂ Φ / ∂ r = 0 acquires an additional term via the product rule: ∂ Φ ∂ r = − A ( Q ) ˜ F ( W o ( r )) r − 5 + 2 B r + ( 1 + p ) Cr p = 0, where ˜ F ( W o ) ≡ 4 F ( W o ) − W o F ′ ( W o ) > 0 for all physiological W o . Mathematically , this positiv- ity condition equates to the logarithmic derivative requir ement d ( ln F ) / d ( ln W o ) < 4. Because the W omersley multiplier asymptotically approaches F ∝ W o for purely inertia-dominated high-frequency flows [ 4 ], its logarithmic derivative is strictly bounded by 1 ≪ 4 across all physi- ological r egimes. The W omersley correction therefor e modifies the coefficient of the dominant r − 5 term but leaves its algebraic power unchanged. Since the proof of Theorem 4 r elies exclusively on the sign structur e and the relative powers { − 5, 1, p } of the three terms in ∂ Φ / ∂ r —not on the precise magnitude of the dissipation coefficient—the strict bounds ( 5 + p ) / 2 < α ∗ ( Q ) < 3 3 are pr eserved under arbitrary smooth F ( W o ( r )) . This is structurally identical to the ar gument for Perturbation 3 (Fahræus–Lindqvist): in both cases, an r -dependent modification of the dissipation coef ficient leaves the maintenance-term incommensurability—the true source of non- universality—intact. While the logarithmic derivative analytically guarantees the preservation of these bounds, numerical evaluation across the physiological W omersley range ( W o ∈ [ 1, 10 ] ) confirms that the actual dynamic shift of the lower bound is strictly of or der O ( 10 − 2 ) . Therefor e, the static prediction α ∗ ≈ 2.90 remains quantitatively robust even in the proximal pulsatile regime. Crucially , this perturbation strictly captur es only the altered velocity profile of the viscous drag (the active dissipated power). It explicitly does not account for the r eactive power—fluid inertia and vessel-wall compliance—associated with pulsatile pressur e-wave pr opagation. These reactive components r epresent a physically distinct, non-dissipative penalty that is invisible to a purely static metabolic ledger: they enter as dimensionless wave-r eflection losses at bifurcations, a network-level observable incommensurate with the extensive costs optimised here. Their incorporation therefor e requir es a unified network-level Lagrangian in which dissipative and non-dissipative penalties are tr eated on equal footing—a structural extension beyond the scope of the present static framework. 3 Mathematical Results Theorem 1 (Existence and uniqueness of the optimal radius) . For every Q > 0 and every A , B , C , p > 0 , the function r 7 → Φ ( r , Q ) has a unique global minimum r ∗ ( Q ) on ( 0, + ∞ ) . Proof. The derivative f ( r ) = ∂ Φ / ∂ r = − 4 Ar − 5 + 2 Br + ( 1 + p ) C r p satisfies f ( r ) → − ∞ as r → 0 + and f ( r ) → + ∞ as r → + ∞ , so by the Intermediate V alue Theorem at least one zero exists. The second derivative (acting as the 1D scalar Hessian determinant) ∂ 2 Φ ∂ r 2 = 20 A r − 6 + 2 B + p ( 1 + p ) Cr p − 1 > 0 for all r > 0 , so Φ is strictly convex, giving a unique global minimum. Moreover , r ∗ ( Q ) is smooth and strictly increasing: by the implicit function theorem applied to ∂ Φ / ∂ r = 0, dr ∗ d Q = − ∂ 2 Φ / ∂ r ∂ Q ∂ 2 Φ / ∂ r 2 > 0 , since ∂ 2 Φ / ∂ r ∂ Q = − 64 µ Q / ( π r 5 ) < 0 and ∂ 2 Φ / ∂ r 2 > 0. Lemma 2 (Power law ⇔ Murray’s law on all tr ees) . Let r ∗ ( Q ) be the optimal radius fr om Theor em 1 . The branching law r ∗ ( Q 0 ) α = r ∗ ( Q 1 ) α + r ∗ ( Q 2 ) α holds for all flow-conserving bifurcations Q 0 = Q 1 + Q 2 if and only if r ∗ ( Q ) = k Q 1/ α for some constants k , α > 0 . Proof. ( ⇐ ) If r ∗ ( Q ) = k Q 1/ α , then r ∗ ( Q 0 ) α = k α Q 0 = k α ( Q 1 + Q 2 ) = r ∗ ( Q 1 ) α + r ∗ ( Q 2 ) α . ( ⇒ ) Define g ( Q ) = r ∗ ( Q ) α . The branching condition becomes g ( Q 1 + Q 2 ) = g ( Q 1 ) + g ( Q 2 ) for all Q 1 , Q 2 > 0. This is Cauchy’s functional equation on ( 0, + ∞ ) . Since r ∗ is continuous and strictly incr easing in Q (as the minimizer of a family of strictly convex functions varying continuously in Q ), g is continuous. The unique continuous solution is g ( Q ) = c Q , giving r ∗ ( Q ) = c 1/ α Q 1/ α . Remark 1 . Lemma 2 connects local single-bifurcation optimization to the global tree law ( 1 ) : a universal branching exponent exists for a cost function if and only if its optimal radius is a power law in flow . 4 Theorem 3 (Single-term classification) . For the two-term cost Φ γ ( r , Q ) = A ( Q ) r − 4 + Br γ with A ∝ Q 2 and B , γ > 0 , the optimal radius is r ∗ ( Q ) = K γ Q 2/ ( 4 + γ ) and Murray’ s branching law holds with α = 4 + γ 2 . (4) Proof. Setting ∂ Φ γ / ∂ r = 0: 4 A ( Q ) r − 5 = B γ r γ − 1 , so r 4 + γ = [ 4 / ( B γ ) ] · A ( Q ) ∝ Q 2 , giving r ∗ ( Q ) ∝ Q 2/ ( 4 + γ ) . This is a power law , so by Lemma 2 Murray’s law holds with α = 1 / ( 2 / ( 4 + γ )) = ( 4 + γ ) /2. T able 1 lists the principal special cases. Theorem 3 identifies the Murray–Bennett family as pr ecisely the homogeneous subclass of the br oader cost family ( 3 ) . Homogeneity—the maintenance term Br γ scaling as a single power of r —is both necessary and sufficient for a universal branching exponent, as established by Lemma 2 . The Murray law ( γ = 2), the Da V inci surface law ( γ = 1), and Bennett’s EPIC result [ 5 ] are therefore not independent discoveries but distinct instances of a single degeneracy class: homogeneous cost functions admitting scale-invariant branching. The three-term cost ( 3 ) with B , C > 0 necessarily exits this class because r 2 and r 1 + p carry incommensurate scaling exponents (2 = 1 + p for p = 1), making the composite cost inho- mogeneous. Non-universality of α is therefor e a direct structural consequence of biological completeness—the inclusion of both volumetric and wall-tissue maintenance—not a numerical accident. The formula ( 4 ) coincides with Bennett’s α = ( m + 4 ) / 2 under γ ≡ m ; the distinction is that γ = 1 + p is independently measurable fr om histology , yielding a parameter -free pr ediction of m for any network with a wall-like maintenance cost, without morphometric fitting. T able 1: Single-term classification via Theorem 3 . Cost ∼ r γ α = ( 4 + γ ) /2 name γ = 0 2.000 impedance matching γ = 1 2.500 Da V inci / surface γ = 1 + p = 1.77 2.885 wall cost (limit) γ = 2 3.000 Murray (volume) Theorem 4 (Strict bounds for the thr ee-term case) . For the cost function ( 3 ) with B , C > 0 and p ∈ ( 0, 1 ) , define for each Q > 0 the local branching exponent α ∗ ( Q ) = ln 2 ln ( r ∗ ( Q ) / r ∗ ( Q /2 ) ) , the exponent at a symmetric bifurcation with inlet flow Q . The symmetric split f = 1 / 2 is chosen as the canonical definition; Corollary 5 establishes that the bounds ar e independent of f . Then 5 + p 2 < α ∗ ( Q ) < 3 for all Q > 0 . (5) Proof. Fix Q > 0. Let r 0 = r ∗ ( Q ) , r 1 = r ∗ ( Q / 2 ) , and ρ = r 1 / r 0 ∈ ( 0, 1 ) . Dividing the optimality conditions for r 0 and r 1 and writing r 1 = ρ r 0 : h ( ρ ) ≡ 4 ρ 6 + 4 λρ 5 + p − 1 − λ = 0 , (6) where λ = ( 1 + p ) C r p − 1 0 / ( 2 B ) > 0. Monotonicity . h ′ ( ρ ) = 24 ρ 5 + 4 λ ( 5 + p ) ρ 4 + p > 0 for ρ > 0, and h ( 0 ) < 0 < h ( 1 ) , so ( 6 ) has a unique root ρ ∗ ∈ ( 0, 1 ) . 5 Upper bound. Let ρ M = 2 − 1/ 3 (the Murray value). Evaluating: h ( ρ M ) = λ h 4 · 2 − ( 5 + p ) / 3 − 1 i > 0 , since ( 5 + p ) / 3 < 2 for p < 1, so 4 · 2 − ( 5 + p ) / 3 > 1. Thus ρ ∗ < ρ M , giving r 0 / r 1 > 2 1/3 and α ∗ ( Q ) = ln 2/ ln ( 1/ ρ ∗ ) < 3. Lower bound. Let ρ W = 2 − 2/ ( 5 + p ) (the wall-only value). Evaluating: h ( ρ W ) = 4 · 2 − 12/ ( 5 + p ) − 1 < 0 , since 12/ ( 5 + p ) > 2 for p < 1. Thus ρ ∗ > ρ W and α ∗ ( Q ) > ( 5 + p ) /2. Both bounds are strict because B , C > 0 exclude the degenerate limits. Corollary 5 (Independence fr om flow asymmetry) . For an asymmetric bifurcation where a parent vessel of flow Q splits into two daughters carrying fractions f Q and ( 1 − f ) Q with f ∈ ( 0, 1 ) , the local branching exponent α ∗ ( Q , f ) defined by r ∗ ( Q ) α = r ∗ ( f Q ) α + r ∗ ( ( 1 − f ) Q ) α obeys the same strict boundaries: ( 5 + p ) /2 < α ∗ ( Q , f ) < 3 . Proof. Since r ∗ is strictly increasing in Q (Theorem 1 ), define x = r ∗ ( f Q ) r ∗ ( Q ) ∈ ( 0, 1 ) , y = r ∗ ( ( 1 − f ) Q ) r ∗ ( Q ) ∈ ( 0, 1 ) . The branching equation r ∗ ( Q ) α = r ∗ ( f Q ) α + r ∗ ( ( 1 − f ) Q ) α is equivalent to G ( α ) ≡ x α + y α = 1. Since x , y ∈ ( 0, 1 ) , we have G ′ ( α ) = x α ln x + y α ln y < 0 (both logarithms are strictly negative), so G is strictly decreasing . Moreover G ( 0 ) = 2 > 1 and G ( α ) → 0 < 1 as α → + ∞ . By the Intermediate V alue Theor em, G ( α ∗ ) = 1 has a unique solution α ∗ ( Q , f ) > 0. In the Murray limit ( C → 0), r ∗ ∝ Q 1/3 , so x = f 1/3 , y = ( 1 − f ) 1/3 , and G ( 3 ) = f + ( 1 − f ) = 1; hence α ∗ = 3. In the wall-dominated limit ( B → 0), r ∗ ∝ Q 2/ ( 5 + p ) , so x = f 2/ ( 5 + p ) , y = ( 1 − f ) 2/ ( 5 + p ) , and G ( ( 5 + p ) /2 ) = 1; hence α ∗ = ( 5 + p ) /2. For the intermediate r egime B , C > 0, r ∗ ( Q ) is not a power law (Cor ollary 6 ); we evaluate the bounds explicitly . Upper bound G ( 3 ) < 1: Evaluate 4 A = 2 B r 6 + ( 1 + p ) Cr 5 + p at the test radius f 1/3 r ∗ ( Q ) : 2 B f 2 r ∗ ( Q ) 6 + ( 1 + p ) C f ( 5 + p ) /3 r ∗ ( Q ) 5 + p . Since p < 1, ( 5 + p ) / 3 < 2, so f ( 5 + p ) /3 > f 2 for f ∈ ( 0, 1 ) . The expression strictly exceeds f 2 [ 2 Br ∗ ( Q ) 6 + ( 1 + p ) C r ∗ ( Q ) 5 + p ] = 4 A ( f Q ) . Because the right-hand side of the optimality condition is strictly increasing in r , this forces r ∗ ( f Q ) < f 1/3 r ∗ ( Q ) , i.e. x < f 1/3 , and identically y < ( 1 − f ) 1/3 , giving G ( 3 ) = x 3 + y 3 < f + ( 1 − f ) = 1. Lower bound G 5 + p 2 > 1: Evaluate at the test radius f 2/ ( 5 + p ) r ∗ ( Q ) : 2 B f 12/ ( 5 + p ) r ∗ ( Q ) 6 + ( 1 + p ) C f 2 r ∗ ( Q ) 5 + p . Since p < 1, 12 / ( 5 + p ) > 2, so f 12/ ( 5 + p ) < f 2 . The expression is strictly less than 4 A ( f Q ) , forcing r ∗ ( f Q ) > f 2/ ( 5 + p ) r ∗ ( Q ) . Setting β = ( 5 + p ) / 2, this gives x β > f and y β > 1 − f , so G 5 + p 2 = x β + y β > 1. 6 Since G is strictly decr easing, the two bounds G ( 3 ) < 1 and G 5 + p 2 > 1 rigorously imply ( 5 + p ) / 2 < α ∗ ( Q , f ) < 3 for all f ∈ ( 0, 1 ) . Non-universality is ther efore an inher ent property of the composite cost function, not a geometric artifact of the symmetric split assumption. Corollary 6 (Uniqueness of Murray scaling) . Among all cost functions of the form ( 3 ) with B , C ≥ 0 (not both zero), Murray’ s cubic branching law ( α = 3 ) is the unique member admitting a universal, scale-independent exponent. Any deviation intr oduced by the biological wall cost ( p < 1 ) structurally breaks the Euler homogeneity of the cost function (rendering it inhomogeneous with incommensurate scaling), rendering the branching exponent dependent on the absolute flow scale Q . This repr esents a structural impossibility r esult within the additive cost-function class ( 3 ) : a universal exponent ca nnot exist in a biologically complete transport network whose wall cost scales sub-linearly ( p < 1 ). Proof. By Euler ’s homogeneous function theorem, a universal exponent α exists if and only if the maintenance cost Φ m ai nt ( r ) = B r 2 + Cr 1 + p is a homogeneous function of r . This requires r 2 and r 1 + p to satisfy the same scaling, which holds only if p = 1. For any biological network with sub- linear wall scaling ( p < 1), the maintenance term is quasi-homogeneous but not homogeneous. Consequently , the optimality condition ( 32 µ / π ) Q 2 = 2 Br 6 + ( 1 + p ) C r 5 + p cannot be solved by a single power-law r ∗ ( Q ) ∝ Q 1/ α . By Lemma 2 , no universal α exists for C > 0 and p < 1. Corollary 7 (General three-term incommensurability) . Let Φ ( r , Q ) = A ( Q ) r − n + B r m + C r k be any cost function where A ( Q ) is strictly positive and strictly increasing in Q , B , C > 0 , and the maintenance exponents are distinct : m = k . Then no universal branching exponent α exists for B , C > 0 . Proof. By Lemma 2 , a universal α exists if and only if r ∗ ( Q ) ∝ Q 1/ α , which r equires the maintenance cost Φ maint ( r ) = Br m + C r k to be homogeneous in r . Homogeneity requir es m = k . For m = k and B , C > 0 , the optimality condition n A ( Q ) r − ( n + 1 ) = m B r m − 1 + k C r k − 1 cannot be solved by any power law r ∗ ( Q ) ∝ Q 1/ α , because for m = k the right-hand side is a sum of strictly linearly independent power laws in r , preventing the factorization of a single scaling variable Q . By Lemma 2 , no universal α exists. □ Remark 2 . Corollary 6 is a special case of Corollary 7 with n = 4, m = 2, k = 1 + p , and A ( Q ) ∝ Q 2 (Poiseuille). The general result establishes that non-universality is the generic be- haviour of any additive cost function with two maintenance terms at distinct scaling exponents, independently of the transport physics encoded in A ( Q ) . The two-term homogeneous family (Theorem 3 , Bennett’s framework) is the unique class admitting a universal branching exponent. Proposition 8 (Physical determination of Bennett’s parameter) . The structural pricing parameter m in Bennett’ s EPIC framework [ 5 ] is determined analytically by the histological wall-thickness law: m = 1 + p . For por cine coronary arteries ( p = 0.77 ), this predicts m ≈ 1.77 , rigorously explaining why empirical physiological data fall between the ideal limits of m = 1 (surface-priced) and m = 2 (volume-priced) explored in the original Bennett formulation. Corollary 9 (Asymptotic behaviour) . Under the same hypotheses, α ∗ ( Q ) → 3 as Q → + ∞ and α ∗ ( Q ) → ( 5 + p ) /2 as Q → 0 + . Proof. As Q → + ∞ , r 0 → + ∞ and λ ∝ r p − 1 0 → 0 (since p < 1). Equation ( 6 ) reduces to 4 ρ 6 ≈ 1, giving ρ ∗ → 2 − 1/ 3 and α ∗ → 3. As Q → 0 + , r 0 → 0 and λ → + ∞ . Dividing ( 6 ) by λ gives 4 ρ 5 + p ≈ 1, so ρ ∗ → 2 − 2/ ( 5 + p ) and α ∗ → ( 5 + p ) /2. 7 Remark 3 (V alidity of the thin-wall approximation) . The thin-wall geometry h ≪ r requir es h / r = c 0 r p − 1 ≪ 1. For p = 0.77 < 1, this ratio grows as r → 0: at the proximal coronary ( r 0 = 1.5 mm) one has h / r ≈ 0.18, well within the thin-wall regime. The approximation loses accuracy as h / r increases, reaching h / r ≈ 0.42 at r ≈ 0.04 mm (40 µ m, generation g ≈ 7–8), where the tr ue annular cross-section deviates from the thin-wall estimate by appr oximately 20%. The asymptotic limit α ∗ → ( 5 + p ) / 2 (Corollary 9 ) should therefor e be interpreted with caution at arteriolar scales approaching the capillary bed. 3.1 Structural stability of the non-universality result The non-universality established in Theorem 4 and Corollary 6 r ests on the positivity of B and C and the strict inequality p = 1. W e verify that these conditions—and hence the result—are preserved under the thr ee physiologically most relevant perturbations. Perturbation 1: Generation-dependent wall scaling p ( g ) . In real arterial trees, p may ap- proach 1 in terminal arterioles (Laplace limit for thin membranes). Let p ( g ) = ¯ p + δ p ( g ) where ¯ p = 0.77 and | δ p ( g ) | ≤ 0.2. The key structural condition for Theor em 4 is ¯ p < 1 strictly , so that the wall-cost exponent 1 + ¯ p remains strictly less than 2 (the blood-volume exponent). Under the perturbation, p ( g ) < 1 is preserved for all generations provided | δ p ( g ) | < 0.23, which is satisfied within the physiological range. The contribution of terminal generations to the network-averaged exponent is suppr essed exponentially with generation number under self- similar scaling, so local violations near the capillary limit do not propagate to the macr oscopic α ∗ . The non-universality bounds therefor e remain str ucturally intact under realistic p ( g ) variation. Perturbation 2: Active smooth-muscle tone. As discussed in Remark 7 , basal vascular tone contributes a term Φ active to leading order un der the thin-wall approximation that scales as r 2 , producing a renormalization B → ˜ B = B + B active > B . Since Theorem 4 requires only ˜ B > 0 and C > 0, the bounds ( 5 + p ) / 2 < α ∗ ( Q ) < 3 are pr eserved identically . This perturbation is structurally benign: active tone shifts the position of α ∗ within the interval but cannot move it outside. This is the most robust of the thr ee stability results. Perturbation 3: Non-Newtonian viscosity in small vessels. In arterioles below r ≈ 50 µ m, the F ˚ ahræus–Lindqvist effect intr oduces a weak r -dependence in the effective viscosity , modifying the dissipation coef ficient A ( Q ) → A ( Q , r ) . The additional term ∂ A / ∂ r appearing in the opti- mality condition repr esents a higher-or der perturbation that does not alter the sign structur e underlying Theorem 4 : the dominant balance between the viscous term ( ∼ r − 5 ) and the mainte- nance terms ( ∼ r , ∼ r p ) is pr eserved, and the two maintenance terms r emain non-commensurate (exponents 1 and 1 + p remain distinct for p = 1). Non-universality therefor e persists in the arteriolar r egime, with a quantitative shift in α ∗ that is accessible to numerical evaluation but does not affect the str uctural conclusion. T ogether , these analyses confirm that the non-universality of α ( Q ) reflects an intrinsic property of the cost-function structure—specifically , the incommensurability of the maintenance- cost exponents in Φ maint ( r ) = B r 2 + C r 1 + p —rather than an artifact of idealized parameterization. These analyses address parametric robustness within the additive cost structure ( 3 ) . Robustness to alternative functional forms (e.g. multiplicative coupling or non-additive cost terms) lies beyond the present scope; however , the cor e non-universality result depends only on this incommensurability , a property preserved under any smooth perturbation that maintains distinct powers of r with positive coefficients. 8 4 Bifurcation Angles The branching angle is determined by minimizing the total network cost with respect to the junction position. Using the generalized Fermat-T orricelli principle [ 11 , 3 ], the force balance at the optimal junction is ∑ i Φ ∗ ( r i ) ˆ e i = 0, where Φ ∗ ( r i ) is the cost per unit length evaluated at the optimum radius, and ˆ e i are unit vectors pointing fr om the junction to the endpoints. Lemma 10 (Optimal local cost) . At the optimal radius r ∗ , the local cost Φ ∗ ( r ) ≡ Φ ( r ∗ , Q ) is Φ ∗ ( r ) = 3 2 Br 2 + 5 + p 4 C r 1 + p . (7) Proof. Substituting A r − 4 = 1 4 [ 2 Br 2 + ( 1 + p ) C r 1 + p ] from the optimality condition ∂ Φ / ∂ r = 0 into Φ = A r − 4 + B r 2 + Cr 1 + p yields the result. Theorem 11 (Bifurcation angles) . Given fixed branching topology ( N = 2 ), predetermined daughter radii r 1 , r 2 independently determined by Theorem 1 (and hence fixed flows Q 1 , Q 2 ), the junction position that minimizes total local cost yields optimal angles θ 1 , θ 2 given by cos θ 1 = Φ ∗ ( r 0 ) 2 + Φ ∗ ( r 1 ) 2 − Φ ∗ ( r 2 ) 2 2 Φ ∗ ( r 0 ) Φ ∗ ( r 1 ) (8) and symmetrically for θ 2 . For a symmetric bifurcation ( r 1 = r 2 , θ 1 = θ 2 ≡ θ ), the total branching angle 2 θ ∗ is bounded by: 74.9 ◦ < 2 θ ∗ ( Q ) < 80.2 ◦ ( for p = 0.77 ) . (9) Proof. Let the junction node be at x , connecting to three fixed endpoints x i ( i ∈ { 0, 1, 2 } ). The total local cost to minimise is H ( x ) = 2 ∑ i = 0 Φ ∗ ( r i ) ∥ x i − x ∥ . Setting ∇ x H = 0 and defining outward unit vectors ˆ e i = ( x i − x ) / ∥ x i − x ∥ yields the geometric force balance : Φ ∗ ( r 0 ) ˆ e 0 + Φ ∗ ( r 1 ) ˆ e 1 + Φ ∗ ( r 2 ) ˆ e 2 = 0 . The asymmetric angles follow directly fr om the law of cosines applied to this vector sum. In the symmetric case, balancing for ces along the par ent axis gives cos θ = Φ ∗ ( r 0 ) / [ 2 Φ ∗ ( r 1 ) ] . In the Murray limit ( C → 0), Φ ∗ ( r ) ∝ r 2 . Using r 1 / r 0 = 2 − 1/ 3 from T able 1 , we find cos θ M = 2 − 1/ 3 , yielding 2 θ M ≈ 74.9 ◦ . In the wall-cost limit ( B → 0), Φ ∗ ( r ) ∝ r 1 + p . Using r 1 / r 0 = 2 − 2/ ( 5 + p ) , we find cos θ W = 2 ( p − 3 ) / ( 5 + p ) . For p = 0.77, 2 θ W ≈ 80.2 ◦ . As the parameter ratio C / B varies continuously from 0 to ∞ , the angle function cos θ = Φ ∗ ( r 0 ) / [ 2 Φ ∗ ( r 1 ) ] varies continuously between these two limits. Since the two limiting angles are distinct, the intermediate value theorem guarantees 2 θ ∗ ∈ ( 74.9 ◦ , 80.2 ◦ ) for all finite positive C / B . This generalizes Zamir ’s classical results [ 11 ] to the thr ee-term cost function and provides a tighter , parameter-free bound for the opening angle than the 75 ◦ –97 ◦ range predicted phe- nomenologically by Bennett [ 5 ]. While this geometric solid is conceptualized as planar for single bifurcations, the vector equilibrium dir ectly applies to fully 3D branching structures. 9 Empirical comparison. Three-dimensional morphometric r econstructions of porcine and hu- man coronary arterial tr ees report bifurcation angles in the range 2 θ obs ≈ 70 ◦ –82 ◦ across branch- ing or ders II–VI [ 12 , 13 ], with measur ements from symmetric or near -symmetric bifurcations (daughter diameter ratio d 1 / d 2 > 0.8) concentrating between 74 ◦ and 80 ◦ . This range is fully contained within the theoretical bound 74.9 ◦ < 2 θ ∗ < 80.2 ◦ derived above. No parameters were fitted to the angle data: the bound depends only on p = 0.77, drawn from histological measurements [ 4 , 7 ] entir ely independent of the morphometric angle dataset. 5 Optimal Branching Number Biological transport networks overwhelmingly favor bifurcations ( N = 2) over higher-order multifurcations ( N > 2). Y et under Murray’s classical cost function, the total energy is completely degenerate with respect to the branching number N . The three-term cost function r esolves this geometric degeneracy . Theorem 12 (T opological bounding of branching number) . For a space-filling fractal network where vessel length scales as L ∝ r , Murray’ s classical cost function ( p = 1 ) is topologically degenerate, yielding identical total network costs for any branching number N . Introducing the empirical sub-linear wall cost ( p < 1 ) strictly breaks this degeneracy . The competition between the metabolic volume of the vessels (which decreases with N ) and the steric tissue cost of the junctions (which increases with N ) strictly forbids star-like topologies ( N → ∞ ) and guarantees the existence of a finite, small optimal branching integer N ∗ ≥ 2 . Proof. Let the network perfuse M terminal units. Under self-similar scaling r g = r 0 N − 2 g / ( 5 + p ) , the metabolic cost of the vessels across generations forms a geometric series. Summing over the network yields the total tube cost: C tu be s ( N ) = K 1 [ 1 − N − α 1 ] − 1 . The series convergence argument fundamentally holds for any finite physiological tree depth G : the truncated sum ∑ G g = 0 N − g α 1 preserves strict monotonicity in N identically to the infinite limit, since each partial sum is a strictly decreasing function of N for α 1 > 0. For a finite network depth G ∼ log N M , the truncated series yields a macr oscopic scaling for the network junction cost: C ju nc ti o ns ( N ) = K 2 N 1 − p 5 + p . where K 2 > 0. For the empirically observed sub-linear wall scaling p < 1, the exponent α 2 = 1 − p 5 + p is strictly positive, making the junction cost strictly increasing with N . The total topological cost is C to t ( N ) = C tu be s ( N ) + C ju nc ti o ns ( N ) . T reating N ≥ 2 as a continu- ous real variable, C to t ( N ) is continuous. Evaluating the asymptotic limit: lim N → + ∞ C to t ( N ) = ∞ . Because C to t ( N ) is coercive on the interval [ 2, + ∞ ) , it admits at least one global minimizer N ∗ re a l ≥ 2. Biologically , branching is restricted to integers, so the optimal physical topology N ∗ is the integer that minimizes this sequence. Furthermore, because the exponent α 2 is small ( ≈ 0.04 for p ≈ 0.77), the junction penalty grows very slowly , naturally restricting the optimum to very small integers (typically N ∈ { 2, 3, 4 } ). Finally , in the classical Murray limit ( p = 1), the exponent α 2 = 0, so that C ju nc ti o ns ( N ) becomes independent of N . Because the tube cost is also N -independent under Murray scaling, the total cost becomes completely flat with respect to the branching number , producing a topological degeneracy . Since H ( N ) is strictly decr easing (established above), the unique critical point of C tot on [ 2, + ∞ ) satisfies N ∗ real ∈ [ 2, + ∞ ) . For all integers N ≥ N ∗ real , strict unimodality 10 guarantees C tot ( N + 1 ) > C tot ( N ) , so the integer minimizer is well-defined without further approximation. Remark 4 (Necessary versus sufficient condition for N = 2) . Theorem 12 establishes that the metabolic junction penalty strictly bounds the optimal branching number to small finite integers, excluding star-like topologies ( N → ∞ ). However , the slow gr owth of the junction-cost exponent ( 1 − p ) / ( 5 + p ) ≈ 0.040 for p = 0.77 implies that the purely static cost differ ence between N = 2 and N = 3 is small: junction maintenance alone provides a necessary condition (small N ) but not a sufficient one (unique N = 2). A complementary criterion is needed. Corollary 13 (Biological Pr ediction: T olerance to Developmental Noise) . The junction-cost ex- ponent α 2 ≈ 0.04 for p = 0.77 structurally bounds the optimum to N = 2 , but implies a shallow thermodynamic energy gradient between binary branching and higher-order multifurcations. Rather than a model weakness, this shallow gradient physically explains a well-documented biological real- ity: occasional trifurcations ( N = 3 ) are not catastrophic violations of the variational principle, but quasi-degenerate morphological solutions lying only O ( α 2 ) above the global minimum. Quantita- tively , in the wall-dominated limit the incr emental cost gap is C junctions ( N + 1 ) − C junctions ( N ) = K 2 [ ( N + 1 ) α 2 − N α 2 ] = O ( α 2 · N α 2 − 1 · K 2 ) , which for p = 0.77 evaluates to ≈ 0.017 K 2 at N = 2 . This is the pr ecise sense in which trifur cations are quasi-degenerate: they lie within 1.6% of the binary minimum in the junction-dominated r egime. T rifurcations are therefor e accessible under local develop- mental noise or spatial boundary constraints. The shallow gradient is therefor e itself a strong, falsifiable prediction of the model. This quasi-degeneracy obtains in the asymptotic wall-dominated regime ( κ → ∞ ), where the junction term governs the cost function. In the physiological regime ( κ ≈ 13 , K 2 / K 1 ≈ 0.19 ), the full three-term competition reverses this conclusion: the tube-cost savings of trifurcations dominate and N = 3 is statically pr eferred (Cor ollary 15 ). The shallow-gradient interpretation ther efore applies to evolutionary perturbations around a dynamically-enforced N = 2 baseline, not to unconstrained static optimisation. W e note that translating the cost gap ∆ C ∼ O ( α 2 K 2 ) into a developmental noise tolerance requir es an implicit mapping to a stochastic or dynamical scale, which lies outside the present deterministic framework. This constitutes a testable hypothesis rather than a derived consequence. Proposition 14 (Static topological uniqueness of binary branching) . For a hierarchically branching network governed by sub-linear wall cost ( p < 1 ), binary bifurcation ( N = 2 ) is the unique optimal topology among all integers N ≥ 2 if and only if the junction-to-tube structural cost ratio K 2 / K 1 exceeds a critical analytical threshold τ ( p ) . In physiological transport networks, the macroscopic volumetric penalty of the junction tissue strongly satisfies this condition, rigor ously selecting N = 2 as the static optimum. Proof. The complete topological cost over the continuous domain N ≥ 2 is: C to t ( N ) = K 1 1 − N − α 1 − 1 + K 2 N α 2 . where α 1 = 1 + p 5 + p and α 2 = 1 − p 5 + p . T o prove that the minimum is unique, we analyze the critical points where d C to t / d N = 0. T aking the derivative and equating to zero yields: K 2 α 2 N α 2 − 1 = K 1 α 1 N − α 1 − 1 1 − N − α 1 − 2 . Rearranging all N -dependent terms to one side defines a function H ( N ) : K 2 α 2 K 1 α 1 = N − α 1 − α 2 1 − N − α 1 − 2 ≡ H ( N ) . The r oots of the derivative correspond to the intersections of H ( N ) with a constant. The monotonicity of H ( N ) is given by its derivative: H ′ ( N ) = N − α 1 − α 2 − 1 ( 1 − N − α 1 ) 3 − ( α 1 + α 2 ) + ( α 2 − α 1 ) N − α 1 . 11 For physiological sub-linear walls ( p > 0), we have α 1 > α 2 > 0. Consequently , both the sum − ( α 1 + α 2 ) and the dif ference ( α 2 − α 1 ) are strictly negative. Because the bracketed term is strictly negative and the prefactor is strictly positive for N ≥ 2, it follows that H ′ ( N ) < 0 universally . Because H ( N ) is strictly monotonically decreasing, the condition H ( N ) = K 2 α 2 K 1 α 1 can have at most one real solution. A single critical point for a coercive function ( lim N → ∞ C to t = ∞ ) analytically guarantees that C to t ( N ) is strictly unimodal. Possessing exactly one global minimum and no other local extrema, the function strictly increases for all N beyond its minimum. Thus, N = 2 is the unique integer minimizer if and only if C to t ( 2 ) < C to t ( 3 ) . Evaluating this exact inequality yields the critical threshold: K 2 K 1 > [ 1 − 2 − α 1 ] − 1 − [ 1 − 3 − α 1 ] − 1 3 α 2 − 2 α 2 ≡ τ ( p ) . The exact topological selection requires evaluating the physiological ratio K 2 / K 1 against the analytical threshold τ ( p ) . Using the empirical parameters for mammalian coronary arteries ( m w = 20 kW m − 3 , b = 1.5 kW m − 3 , c 0 = 0.041, p = 0.77, λ = 10) and the steric shape factor of a symmetric bifurcation manifold ( Ω junc ≈ 2.4), direct numerical evaluation yields: K 2 K 1 = m w Ω junc c 0 π λ b · r p − 1 0 ≈ 0.19. This result is structurally robust: even increasing Ω junc by two orders of magnitude yields K 2 / K 1 < 8, still well below τ ( 0.77 ) ≈ 103. Reaching the critical thr eshold would require Ω junc ≈ 1327, a value exceeding any physical junction geometry by thr ee orders of magnitude. The physiological ratio ther efore falls structurally short of the critical threshold, with a deficit robust to geometric uncertainty by thr ee orders of magnitude. Corollary 15 (Static insufficiency of binary selection) . Purely static viscous-metabolic optimisation is formally insufficient to select binary branching ( N = 2 ) in mammalian arterial networks. Proof. The analytical threshold for binary selection established in Pr oposition 14 is τ ( 0.77 ) ≈ 103 . Direct physiological evaluation yields K 2 / K 1 ≈ 0.19 (see proof above), which is robust to geometric uncertainty by a factor exceeding 10 2 . Since 0.19 ≪ 103 , the condition K 2 / K 1 > τ ( p ) is not satisfied. Physical consequence. Under purely steady-flow optimisation, the volumetric fluid savings of multifurcating architectures ( N ≥ 3 ) outweigh the steric tissue cost of junction manifolds in the physiological parameter space. The universal persistence of N = 2 in macroscopic biological reality therefor e constitutes a mathematical proof that binary branching cannot be explained by steady-flow metabolic optimisation alone. W e identify the missing constraint as the thermodynamic penalty of pulsatile wave-reflection (impedance mismatch) at high-degr ee nodes: this dynamic mechanism enforces N = 2 through a network-level variational principle that lies outside the purely static framework developed here, but whose physical necessity is mandated by the pr esent static insufficiency result. We emphasise that this identification is proposed as a resolution of model underdetermination rather than a deductive consequence: the static insufficiency result establishes that the model requires extension, and wave-reflection dynamics constitute the physically motivated candidate for that extension. As an order-of-magnitude estimate, the reactive power fraction in a pulsatile cor onary tr ee scales as W o − 2 ∼ 0.2 at the aortic root ( W o ≈ 2.3 ), providing a thermodynamic penalty of comparable order to the static cost gap — sufficient to break the degeneracy that the static framework cannot r esolve. Remark 5 (T opological cr ossover at the capillary limit) . The metabolic order parameter κ ≡ m w / b provides a direct physical explanation for the architectural transition from arterial trees to capillary meshes. In macroscopic vessels the active smooth-muscle layer enfor ces κ ≈ 13. 12 Although this value is insufficient to select N = 2 under purely static optimisation (Corollary 15 ), it does generate a thermodynamic preference for small branching integers via the junction penalty C junctions ∝ κ N α 2 (Theorem 12 ). At the capillary level the smooth-muscle layer vanishes entirely , reducing κ → 1. The junction penalty—already too weak to enforce N = 2 in arteries—collapses to the basal endothelial baseline, making higher-or der nodes ( N ≥ 3) fully accessible. The emergence of anastomotic capillary meshes is therefor e not an anomaly but the exact geometric expression of the system crossing the κ → 1 boundary: without the dynamic wave-r eflection penalty to enforce N = 2, and without sufficient junction cost to disfavour N ≥ 3, reticulated topologies become the natural ground state. Remark 6 (Interpretation and forwar d connection) . Proposition 14 pr ovides a structural argument for binary branching rooted in tree ar chitecture. For fixed reach ( M terminals), bifurcating tr ees are the deepest ( G maximized at N = 2), and depth amplifies the cumulative wall investment per unit metabolic cost. The effective stiffness ratio κ eff measures pr ecisely how far the three- term cost function departs fr om Murray’s homogeneous limit: a larger κ eff generates a steeper non-universality , so N = 2 is the architectur e in which wall-cost selection pressur e is strongest. This structural observation admits a rigor ous static completion in the wall-cost-dominated regime. When κ = m w / b ≫ 1, the junction cost overwhelms the tube cost and C tot ( N ) ≈ C junctions ( N ) ∝ N ( 1 − p ) / ( 5 + p ) , which is strictly incr easing in N ; in this limit, N = 2 is the unique minimizer of the static cost. For cor onary arteries, κ = m w / b ≈ 13 ≫ 1, so this limit is biologi- cally realized. T ogether , Theor em 12 (necessary condition: N < ∞ ) and Proposition 14 with the wall-dominated approximation pr ovide a qualitative structural ar gument for binary branching from wall-cost geometry alone. W e note, however , that κ ≈ 13 is pre-asymptotic with r espect to the exact threshold τ ( p ) ≈ 103: the wall-dominated limit C tot ≈ C junctions is not yet fully realised at physiological parameters. The asymptotic argument ther efore pr ovides a structural insight into the direct ionality of selection pressur e rather than a quantitative demonstration of binary selection—a role filled by Cor ollary 15 . The conclusion is thus robust not thr ough parametric precision but thr ough order -of-magnitude separation: any multiplicative perturbation of K 1 or K 2 by a factor less than 10 2 leaves the inequality K 2 / K 1 ≪ τ ( p ) intact. 5.1 Robustness to Non-Isometric Scaling Theorem 12 assumes the standard self-similar scaling L ∝ r . In real physiological networks, average arterial length-radius data are well described by a generalized power law L = k r β , where β repr esents the arboreal extension invariant. Substituting this empirical scaling into the local cost function yields T ( N ) = Φ ∗ ( r 0 ) · k r β 0 + N · Φ ∗ ( r 1 ) · kr β 1 . In the wall-dominated limit, Φ ∗ ∝ r 1 + p and r 1 = N − 2/ ( 5 + p ) r 0 . The combined local length-cost scales as r 1 + p + β , leading to a total daughter cost of: N · N − 2 5 + p r 0 1 + p + β = N 1 − 2 ( 1 + p + β ) 5 + p r 1 + p + β 0 (10) The strict prefer ence for bifurcations ( N = 2) requires this exponent to be strictly positive, meaning 1 > 2 ( 1 + p + β ) 5 + p , which simplifies to the condition β < 3 − p 2 . For the cardiovascular system wher e p ≈ 0.77, the framework strictly pr eserves N = 2 for all β < 1.115. This perfectly bounds the physiological range ( β ≈ 1.0) observed acr oss species [ 2 ], proving that binary branching is str ucturally robust against macroscopic tissue distortions. 13 6 Numerical V erification T o ensur e no circular reasoning, all parameters ar e drawn from sources independent of branching- exponent measurements. Parameters. µ = 3.5 mPa s [ 9 ]; b = 1500 W/m 3 [ 1 , 10 ]; m w ∈ { 5, 20, 35 } kW/m 3 [ 8 ]; c 0 = 0.041, p = 0.77 [ 6 , 4 , 7 ]; Q = 1.3 mL/s, r 0 = 1.5 mm [ 2 ] (both referring to the same proximal cor onary segment). Results. T able 2 shows α ∗ ( Q ) for the thr ee values of m w . The Murray limit is r ecovered exactly: α ∗ ( m w → 0 ) = 3.000000. Across the literature range m w ∈ [ 5, 35 ] kW/m 3 , the predicted exponent spans α ∗ ∈ [ 2.90, 2.94 ] , consistent with the experimental value α exp = 2.7 ± 0.2 [ 2 ] (deviations of 1.0–1.2 σ from the mean). The local exponent varies by only 0.012 across four decades of flow , confirming that α ∗ is nearly but not exactly scale-independent (Corollary 9 ). Remark 7 (Optimal passive radius and active tone) . T able 2 predicts r ∗ ∈ [ 0.94, 1.20 ] mm at Q = 1.3 mL/s, systematically below the morphometric value r 0 = 1.5 mm [ 2 ] by 0.3–0.6 mm. The pr esent model minimizes the purely passive metabolic cost and therefore predicts the optimal radius absent active tone contributions, not the in-vivo active radius under physiological perfusion pressur e. This offset is consistent with basal smooth-muscle tone: pharmacological vasodilation in porcine cor onary preparations incr eases luminal diameter by approximately 20–35% relative to the r esting state [ 10 , 2 ], a range compatible with the observed discr epancy . Active tone enters the cost function to leading order as an additional term ∝ r 2 , equivalent to a renormalization B → B + B active with B active > 0. Since Theor em 4 and Cor ollary 6 requir e only that B , C > 0, the bounds ( 5 + p ) / 2 < α ∗ ( Q ) < 3 are pr eserved exactly under any positive renormalization of B . The predicted passive radius therefor e repr esents a testable lower bound on coronary caliber under maximal pharmacological vasodilation, accessible to standar d pressur e-myograph or hyper emic flow protocols. This discrepancy is physiologically consistent with active vasomotor tone. While our framework predicts the structural (passive) optimum r ∗ ≈ 1.2 mm, in vivo coronary arteries exhibit significant basal constriction. A vasomotor contraction of approximately 20% would reconcile our theor etical optimum with the higher morphometric values observed in dilated or fixed specimens. T able 2: Predicted α ∗ for porcine cor onary arteries. No fitting; all parameters from independent literature. m w (kW/m 3 ) r ∗ (mm) α ∗ Agreement with α exp 5 1.200 2.938 within 1.2 σ 20 1.019 2.905 within 1.0 σ 35 0.937 2.897 within 1.0 σ Murray limit ( m w → 0) 3.000000 1.5 σ above mean Experimental [ 2 ] 2.7 ± 0.2 – σ computed from experimental uncertainty α exp = 2.70 ± 0.20 [ 2 ]. All thr ee-term predictions lie within 1–1.2 σ of the measured mean, r epresenting a measurable reduction r elative to the Murray limit (1.5 σ ). T o rigor ously assess parametric robustness, α ∗ was evaluated acr oss the full physiological range m w ∈ [ 5, 35 ] kW/m 3 . As shown in T able 2 , the predicted exponent varies only from 2.897 to 2.938, remaining strictly within the theor etical bounds ( 5 + p ) / 2 < α ∗ < 3 throughout. The 14 1 0 2 1 0 1 1 0 0 1 0 1 1 0 2 F l o w Q ( m L / s ) 2.88 2.90 2.92 2.94 2.96 2.98 3.00 L o c a l b r a n c h i n g e x p o n e n t * ( Q ) M u r r a y ( = 3 ) W all limit (2.885) e x p = 2 . 7 0 ± 0 . 2 0 l i e s b e l o w L o c a l O p t i m u m : m w s e n s i t i v i t y m w = 5 k W / m 3 m w = 2 0 k W / m 3 m w = 3 5 k W / m 3 Figure 1: Scale-dependence of the local branching exponent α ∗ ( Q ) . As flow Q increases, the exponent monotonically approaches the Murray limit ( α = 3) fr om below . Thicker walls (higher metabolic wall cost m w ) shift the entire curve toward the wall-dominated lower bound ( 5 + p ) / 2 ≈ 2.885. The shaded region marks the empirical morphometric range for porcine coronary arteries [ 2 ]. non-universality result is therefor e insensitive to pr ecise biochemical parameterization of wall metabolic rate. T able 3: Cross-network validation. Measured wall scaling exponent p and the associated single- term limit α = ( 5 + p ) / 2. When boundary thickness is governed exactly by vessel radius (e.g. Barlow’s stress formula for internal pr essure: h ∝ r ), p = 1 and the bounds collapse, recovering Murray’s theoretical α = 3 perfectly . Network p α Limit α exp Human pulmonary artery [ 14 ] 0.60 2.800 2.7–2.8 Porcine cor onary artery [ 2 , 6 ] 0.77 2.885 2.7 ± 0.2 Plant xylem [ 15 ] 1.00 3.000 (Murray) ∼ 3 Engineered pipes (pr essure) 1.00 3.000 (Murray) ∼ 3 The wall-thickness law also allows robust comparisons across highly disparate physical systems (T able 3 ). Human pulmonary arterial trees exhibit an independent morphometric exponent of α ≈ 2.7–2.8 [ 14 ]. Their characteristically thinner walls ( p ≈ 0.60) naturally push the 15 theoretical limit lower than the systemic cir culation, matching the dir ection of the empirical shift without requiring parameter fitting. Conversely , for networks designed statically to withstand internal pr essure (engineered pipes or certain plant xylem [ 15 ]), mechanics dictate h ∝ r 1 . Setting p = 1 forces the bounds to collapse: ( 5 + 1 ) / 2 = 3 ≤ α ∗ ≤ 3. The optimization forces Murray’s law to be recover ed exactly . 7 T estable Predictions 1. Scale gradient. Since λ ∝ r p − 1 0 is a strictly decreasing function of r 0 (and hence of Q ) for p < 1, and ρ ∗ is strictly decreasing in λ (by implicit differ entiation of ( 6 ) ), α ∗ ( Q ) = ln 2 / ln ( 1 / ρ ∗ ) is strictly incr easing in Q . Thus α ∗ ( Q ) monotonically approaches 3 from below as Q increases (Corollary 9 ). Distal bifurcations (small Q ) should have slightly smaller α than proximal bifur cations. Predicted range: ∆ α ∗ < 0.015 across four decades. 2. Wall-cost sensitivity. Thicker-walled vessels (higher h / r , i.e. larger C / B ) should have smaller α ∗ , approaching the wall-cost limit ( 5 + p ) /2. 3. Physical grounding of γ . Our Theorem 3 gives α = ( 4 + γ ) / 2 where γ = 1 + p is the wall-cost exponent. The same formula was obtained by Bennett [ 5 ] as α = ( m + 4 ) / 2 with m as a fr ee pricing parameter . Our framework pr edicts m = 1 + p for any network whose maintenance cost is dominated by a wall-like sheath. Networks with known p (from histology or materials data) can test this prediction without morphometric fitting. W e emphasize that this physical identification γ = 1 + p , and hence the Bennett formula α = ( 4 + γ ) / 2, holds rigorously onl y in the two-term limit where either B = 0 or C = 0. In the physically complete three-term case ( B , C > 0), Corollary 6 guarantees that no universal exponent exists and the Bennett single-index formula does not apply . This restriction to the two-term class is now fully generalised: Corollary 7 establishes that for any cost function Φ = A ( Q ) r − n + Br m + C r k with m = k and B , C > 0, no universal α exists regar dless of the specific transport physics encoded in A ( Q ) . 8 Relation to Prior W ork Murray [ 1 ]: two-term cost, α = 3 (exact). Our Corollary 6 shows this is the only instantiation of the cost-function family ( 3 ) with a universal exponent. Bennett [ 5 ]: develops a variational framework for the homogeneous two-term class, deriving generalized Murray scaling, Y oung–Herring junction geometry , concave (Gilbert-type) flux costs, and a single-index (rigidity index χ ) unification showing that the two-term form is the unique quadratic scale-free ledger compatible with simultaneous power-law flux–radius scaling and power-law flux-only concavity [ 5 ]. The two-term limit of our Theorem 3 ( α = ( 4 + γ ) / 2 with the exponent γ = 1 + p derived fr om histological data) coincides with the generalized Murray scaling independently established by Bennett thr ough abstract homogeneity axioms. Our Theorem 4 and Corollary 6 address the r egime that lies outside Bennett’s two-term class: incorporating the third biological term ( h ( r ) ∝ r p , sublinear conduit tissue) br eaks Euler homogeneity altogether , pr oducing a rigorously non-universal regime with scale-dependent α ∗ ( Q ) . Liu & Kassab [ 16 ] and Kim & W agenseil [ 17 ] previously incorporated metabolic wall costs into branching models, but explicitly concluded that the standard scaling relations ( α = 3) remained lar gely invariant. This discrepancy underscor es our central thesis: it is specifically the 16 sub-linear allometric scaling of the wall ( p < 1) that br eaks the cubic law . W ithout it, standar d three-term ener gy models natively recover Murray’s limits. T aylor et al. [ 18 ] report optimal Murray exponents of P = 2.15 (for volumetric flow) and P = 2.38 (for microvascular r esistance) in human epicardial coronary arteries, using a CFD model constrained by pressur e-wire measurements. W e note that these values lie below our the- oretical lower bound ( 5 + p ) / 2 ≈ 2.885. This is not a contradiction: T aylor et al. ’s exponents are hemodynamic optimality exponents—the best-fit P in the relationship Q ∝ D P that repr oduces in-vivo flow distributions in a clinical population with cor onary disease—rather than morpho- metric diameter-scaling exponents in the sense of Eq. ( 1 ) . The two quantities coincide only if the tree were exactly Murray-optimal; in diseased or developmentally constrained vascular beds the hemodynamic exponent r eflects additional physiological constraints absent from the static cost function. The morphometric exponent most dir ectly comparable to our framework is that of Kassab et al. [ 2 ], α exp = 2.7 ± 0.2, measured from perfusion-fixed casts of healthy porcine cor onary trees, which lies within our predicted range. The sub-2.885 values reported by T aylor et al. suggest that pulsatile wave costs introduce an additional ar chitectural constraint not captured by the static cost function analyzed her e. Smink et al. [ 19 ]: extend Murray’s two-term optimization to turbulent flows, non-Newtonian fluids, and rough-wall channels, deriving the exponent x in Q ∝ r x across the full Reynolds- number parameter space. Their framework covers the fluid-rheology dimension of the opti- mization problem, while ours covers the biological wall-tissue dimension. Neither framework addresses the signal-transport dimension (such as pulsatile wave reflection in arteries or electri- cal signal attenuation in neur ons), which we pr opose as the final axis requir ed for a complete universal classification. W est, Br own & Enquist [ 20 ]: minimize total network r esistance over a self-similar infinite tree, obtaining Kleiber ’s 3 / 4 law . Our optimization is at the single-vessel level; the network structur e enters only through Murray’s branching condition (Lemma 2 ). Rall [ 21 ]: the branching exponent α = 3 / 2 for electrotonic signal propagation in neurons is derived fr om impedance-matching in cable theory ( Z ∝ r − 3/ 2 ), not fr om cost minimization. Corollary 6 ther efore does not contradict Rall: the two universality results arise from physically distinct mechanisms (energy minimization vs. impedance matching) and cover non-overlapping parameter ranges. However , this mathematical structure str ongly suggests that a generalized Lagrangian unifying metabolic maintenance cost with signal attenuation impedance could natively bridge vascular and neural morphologies. 9 Conclusion W e have shown that the three-term cost function ( 2 ) leads to several rigor ous results unavailable in the existing two-term Murray framework (generalized by Bennett [ 5 ] via a single-index formalism): unique optimal radius (Theorem 1 ), equivalence of local optimization with global tree law (Lemma 2 ), exact single-term classification α = ( 4 + γ ) / 2 with physical grounding of γ (Theorem 3 ), strict non-universality of the three-term branching exponent extending to asymmetric flows (Theorem 4 , Corollary 5 ), breaking of structural degeneracy to bound the optimal topology to small branching integers, excluding star-like multifurcations (Theor em 12 ), and tight theor etical bounds on the optimal symmetric bifurcation angle, independently con- firmed by three-dimensional morphometric data (74.9 ◦ < 2 θ ∗ < 80.2 ◦ ), parameterizing the deviation from Murray’s geometry (Theorem 11 ). The central prediction α ∗ ∈ [ 2.90, 2.94 ] from independently measured parameters measurably r educes the gap between Murray’s cubic law and cardiovascular data. 17 The static attractor as a diagnostic bound. The purely static wall-tissue mechanism establishes a strict theoretical expectation of α ∗ ≈ 2.90 for porcine coronary arteries, demonstrating that ≈ 2.90—not Murray’s 3.0—is the structural baseline for purely dissipative biological networks. This reframing transforms the r esidual gap with empirical cardiovascular data ( α exp ≈ 2.5– 2.7) from a modelling discr epancy into a precise physical diagnostic. The macroscopic depression of the empirical exponent below the 2.90 static attractor serves as an explicit mathematical signature that the cardiovascular tree is subject to physical constraints invisible to a purely dissipative ledger . Furthermore, Cor ollary 15 establishes that static optimisation cannot explain why the vascu- lar tree is binary . Both failures—the exponent gap and the topological selection—signal that the final physiological architectur e must emerge fr om a variational principle that balances extensive metabolic dissipation against dimensionless wave-reflection penalties. Formulating such a unified network-level framework repr esents the necessary definitive extension of the static analysis presented her e. Data and Code A vailability . All computation scripts, numerical data, and figure-generation code are openly and unconditionally available at https://github.com/rikymarche- ctrl/ vascular- networks- theory under the CC BY 4.0 Licence. No access request is r equired. References [1] C. D. Murray . The physiological principle of minimum work. Proc. Natl. Acad. Sci. U.S.A. , 12:207–214, 1926. https://doi.org/10.1073/pnas.12.3.207 . [2] G. S. Kassab, C. A. Rider , N. J. T ang, and Y . C. Fung. Morphometry of pig coronary arterial trees. Am. J. Physiol. , 265:H350–H365, 1993. https://doi.org/10.1152/ajpheart.1993. 265.1.H350 . [3] M. Zamir and W . C. Bigelow . Cost of departure fr om optimality in arterial branching. J. Theor . Biol. , 197:517–523, 1999. https://doi.org/10.1016/S0022- 5193(84)80089- 2 . [4] W . W . Nichols, M. F . O’Rourke, and C. Vlachopoulos. McDonald’ s Blood Flow in Arteries . Hodder Arnold, London, 6th edition, 2011. https://doi.org/10.1201/b13568 . [5] J. Bennett. Murray’s law as an entropy-per -information-cost extremum; and: A single- index theory of optimal branching. arXiv:2511.04022, 2511.19915 , 2025. pr eprint, https: //doi.org/10.48550/arXiv.2511.04022 . [6] J. A. G. Rhodin. The ultrastructur e of mammalian arterioles and precapillary sphincters. J. Ultrastruct. Res. , 18:181–223, 1967. https://doi.org/10.1016/S0022- 5320(67)80239- 9 . [7] H. W olinsky and S. Glagov . A lamellar unit of aortic medial structure and function in mammals. Circ. Res. , 20:99–111, 1967. https://doi.org/10.1161/01.res.20.1.99 . [8] R. J. Paul. Chemical energetics of vascular smooth muscle. In Handbook of Physiology , volume 2, pages 201–235. American Physiological Society , 1980. https://doi.org/10. 1002/cphy.cp020209 . [9] C. G. Caro, T . J. Pedley , R. C. Schroter , and W . A. Seed. The Mechanics of the Circulation . Oxford University Pr ess, Oxford, 1978. https://www.worldcat.org/isbn/0192633236 . 18 [10] L. A. T aber . An optimization principle for vascular radius including the effects of smooth muscle tone. Biophys. J. , 74:109–114, 1998. https://doi.org/10.1016/S0006- 3495(98) 77772- 0 . [11] M. Zamir . Nonsymmetrical bifurcations in arterial branching. J. Gen. Physiol. , 72:837–845, 1978. https://doi.org/10.1085/jgp.72.6.837 . [12] M. Zamir and J. A. Medeiros. Arterial branching in man and monkey . J. Gen. Physiol. , 79(3):353–360, 1982. https://doi.org/10.1085/jgp.79.3.353 . [13] B. Kaimovitz, Y . Lanir , and G. S. Kassab. Large-scale 3-d geometric r econstruction of the porcine cor onary arterial vasculature based on detailed anatomical data. Ann. Biomed. Eng. , 33(11):1517–1535, 2005. https://doi.org/10.1007/s10439- 005- 7544- 3 . [14] W . Huang, R. T . Y en, M. McLaurine, and G. Bledsoe. Morphometry of the human pulmonary vasculature. J. Appl. Physiol. , 81:2123–2133, 1996. https://doi.org/10.1152/jappl.1996. 81.5.2123 . [15] U. G. Hacke, J. S. Sperry , W . T . Pockman, S. D. Davis, and K. A. McCulloh. T rends in wood density and structure are linked to prevention of xylem cavitation by negative pressur e. Oecologia , 126:457–461, 2001. https://doi.org/10.1007/s004420100628 . [16] Y . Liu and G. S. Kassab. V ascular metabolic dissipation in murray’s law . Am. J. Physiol. Heart Circ. Physiol. , 292(3):H1336–H1339, 2007. https://doi.org/10.1152/ajpheart.00906. 2006 . [17] J. Kim and J. E. W agenseil. Bio-chemo-mechanical models of vascular mechanics. Ann. Biomed. Eng. , 43(7):1477–1487, 2015. https://doi.org/10.1007/s10439- 014- 1201- 7 . [18] D. J. T aylor , J. Feher , I. Halliday , D. R. Hose, R. Gosling, L. Aubiniere-Robb, et al. Refining our understanding of the flow thr ough coronary artery branches; r evisiting murray’s law in human epicardial cor onary arteries. Front. Physiol. , 13:871912, 2022. https://doi.org/ 10.3389/fphys.2022.871912 . [19] J. S. Smink, R. Hagmeijer , C. H. V enner , and C. W . V isser . Optimising branched fluidic networks: a unifying approach including laminar and turbulent flows, rough walls and non-newtonian fluids. J. Fluid Mech. , 1011:A42, 2025. https://doi.org/10.1017/jfm. 2025.393 . [20] G. B. W est, J. H. Br own, and B. J. Enquist. A general model for the origin of allometric scaling laws in biology . Science , 276:122–126, 1997. https://doi.org/10.1126/science. 276.5309.122 . [21] W . Rall. Branching dendritic trees and motoneuron membrane resistivity . Exp. Neurol. , 1:491–527, 1959. https://doi.org/10.1016/0014- 4886(59)90046- 9 . 19
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment