Physicochemical-Neural Fusion for Semi-Closed-Circuit Respiratory Autonomy in Extreme Environments
This paper introduces Galactic Bioware's Life Support System, a semi-closed-circuit breathing apparatus designed for integration into a positive-pressure firefighting suit and governed by an AI control system. The breathing loop incorporates a soda l…
Authors: Phillip Kingston, Nicholas Johnston
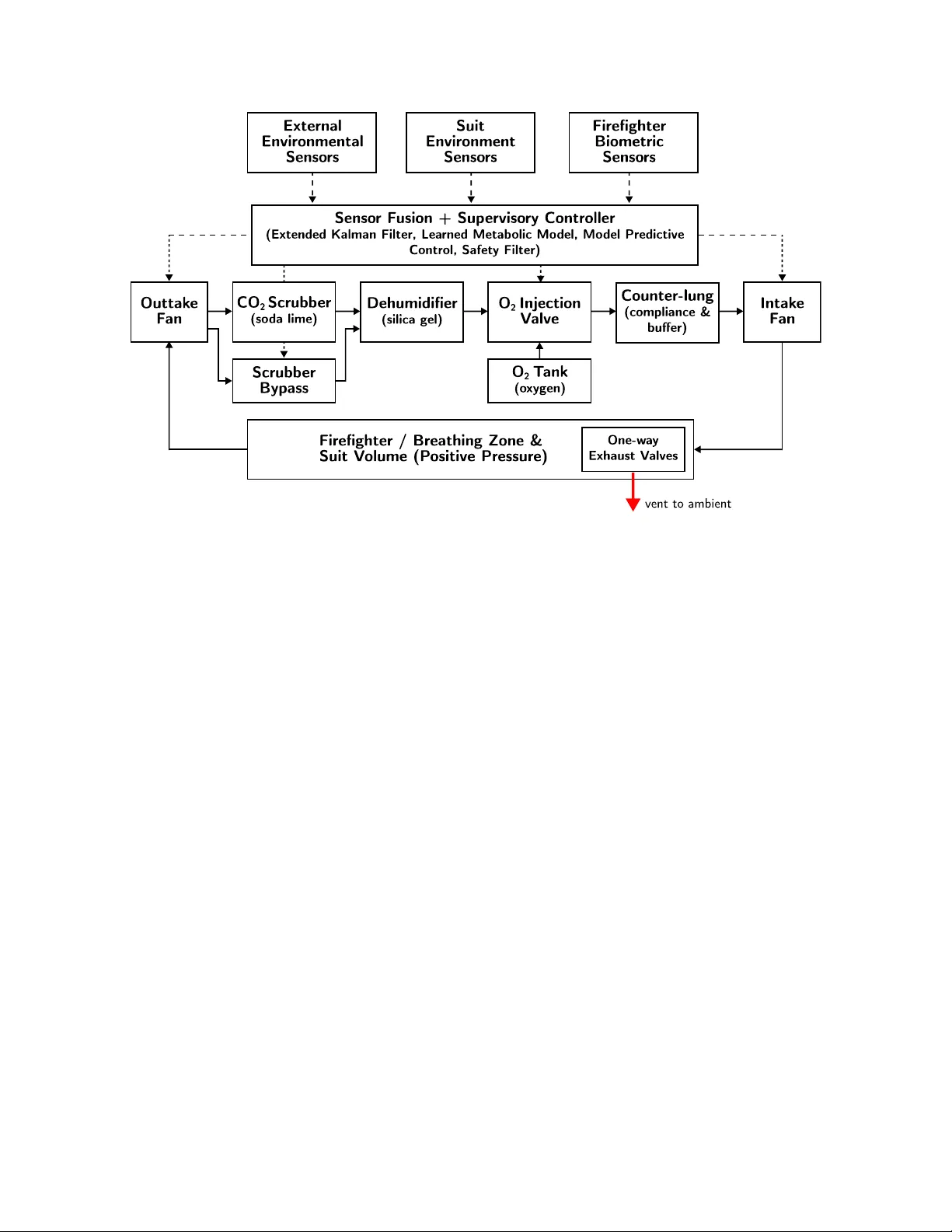
Ph ysico c hemical-Neural F usion for Se mi-Closed -Circuit Respiratory Autonom y in Extreme En vironmen ts Phillip Kingston ∗ Member of T ec hnical Staff Galactic Biow are Nic holas Johnston Member of T ec hnical Staff Galactic Biow are 1 Marc h 2026 CC BY-NC-SA 4.0 This w ork i s licensed under a Creative Commons A ttribution- Noncommercial-ShareAlike 4.0 International License (CC BY -NC-SA 4.0) Abstract This pa pe r int r o duces Galactic Bi owar e’s Life Supp ort System , a semi-close d- circuit brea th- ing apparatus des ig ned for integration into a p ositive-pressure firefighting suit and gov erned by an AI control system. The br e a thing lo o p inco r p orates a so da lime CO 2 scrubb er, a silica gel dehum idifier , and pure O 2 replenishment wit h finite consumables. One-wa y exhaust v alv es maintain p ositive pressure while c r eating a se mi- closed system in which out ward v ent ing grad- ually depletes the g as inv entory . Pa rt I develops the physico chemical foundations fr o m first principles, including sta te-consistent thermo chemistry , stoichiometric capacity limits, ads orp- tion iso therms, and oxygen-management constr aints a rising fr o m b oth fir e sa fet y and toxicit y . P art I I in tro duces an AI co nt r ol architecture that fuses three sensor tiers—ex ternal environ- men tal s ensing, internal suit atmospher e sens ing (with tr iple-redundant O 2 cells and median voting), and firefighter biometrics. The co ntroller co mbin es receding-ho rizon mo del-predictive control (MPC) with a learned metab olic mo del a nd a reinforcement learning (RL) p olicy adv is or, with all candidate actuator commands passing through a final cont r o l-barr ie r -function safety fil- ter before reaching the hardware. This architecture is in tended to optimize p erfor ma nce under unknown mission duration and exertion profiles. In th is pap er: 1. W e introduce a rigor ous first-principles physicochemical mo del of the semi-clos ed breathing lo op, including state-co nsistent thermo chemistry of so da lime scrubbing, GAB-isother m hu midity ma nagement, and the oxygen-enrichmen t dynamic driv en by exha ust-v alve ven t comp ensation with pure O 2 , sub ject to fire- safety constraints. 2. W e in tro duce an 18-state, 3-control nonlinear state- space form ulation using only sensors viable in structura l firefig ht ing , with tr iple-redundant O 2 sensing and median v oting. 3. W e in tro duce an MPC framew or k with a dynamic resour ce scarcity multiplier, an RL po licy advisor for warm-starting , and a final con trol-bar rier-function sa fety filter th r ough which all actuator commands m ust pass, demons tr ating 18–3 4 % endur ance improv ement in s imulation ov er PID baselines while maintaining tighter ph ys io logical a nd fire-sa fet y margins. ∗ Corresponding author: phillip.kingston@galacticbio ware.co m 1 Con ten ts I Chemical and Ph ysical F oundations 5 1 In tro duction 5 2 System Ov erview 5 2.1 Comp onents and La y out . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5 2.2 Airflo w P ath wa y . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6 2.2.1 Gas-T reatmen t Stage Orderin g . . . . . . . . . . . . . . . . . . . . . . . . . . 7 2.2.2 Gas Pa thw a y . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8 2.2.3 Moisture Budget Analysis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8 3 Carb on Dio xide Managemen t 9 3.1 Ph ysiological Con text . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9 3.2 P erformance Degradat ion fr om Hyp ercapnia . . . . . . . . . . . . . . . . . . . . . . . 9 3.3 Chronic Health Effects . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10 3.4 CO 2 Scrubb ing with So da Lime . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10 3.4.1 Sorb ent Choice and Safety . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10 3.4.2 Primary Reaction and Thermo chemistry . . . . . . . . . . . . . . . . . . . . . 11 3.4.3 Reaction Mec hanism and Kinetics . . . . . . . . . . . . . . . . . . . . . . . . 12 3.4.4 Stoic hiometric Capacit y . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13 3.4.5 Thermal Managemen t . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13 4 Humidit y Management 14 4.1 Source of Moi stu r e and In teraction w ith Scrubb er P lacement . . . . . . . . . . . . . 14 4.2 Silica Gel Adsorption: Ph ysical Ch emistr y . . . . . . . . . . . . . . . . . . . . . . . . 14 4.2.1 Adsorption Isotherm . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14 4.2.2 Adsorption Dynamics and Linear Driving F o r ce Mo del . . . . . . . . . . . . . 15 4.2.3 Mass Balance f or the Pac k ed Bed . . . . . . . . . . . . . . . . . . . . . . . . . 15 4.2.4 Heat of Adsorption and the T otal Thermal Budget . . . . . . . . . . . . . . . 16 5 Oxygen Replenishmen t 16 5.1 Metab olic Basis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16 2 5.2 Oxygen C on s umption Rates . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16 5.3 Oxygen T ank Endurance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17 5.4 Oxygen T oxici ty C onsiderations . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17 5.5 Oxygen En ric hment as a Fire Hazard . . . . . . . . . . . . . . . . . . . . . . . . . . . 17 6 Airflo w Ph ysics and Circulation 18 6.1 P ositiv e Pressur e Main tenance and Gas Inv en tory Dynamics . . . . . . . . . . . . . . 18 6.1.1 Correct Gas-Phase Molar Bo okk eeping . . . . . . . . . . . . . . . . . . . . . . 18 6.1.2 Exhaust V alv e V en ting: Th e Real Driv er of Gas I n ven tory Change . . . . . . 18 6.1.3 Oxygen En ric hment from V en t Comp ensation . . . . . . . . . . . . . . . . . . 19 6.1.4 Count er-Lu n g Dynamics and Suit Pressure . . . . . . . . . . . . . . . . . . . 20 6.2 In ternal Circulation: F a n Dynamics . . . . . . . . . . . . . . . . . . . . . . . . . . . 21 6.2.1 Time-V arying V oid F ractio n from Solid and Liquid V ol u m e Expans ion . . . . 21 6.3 Closed-Lo op Pr essure and V olume Dynamics . . . . . . . . . . . . . . . . . . . . . . 22 6.3.1 V en tilation Sanit y C hec k: Lo op Flu s h Rate vs. C O 2 Pro duction . . . . . . . 22 7 In tegrated Mass Balance Summary 24 I I AI-Based Control System 26 8 Motiv at ion: Wh y AI Control? 26 9 Sensor Suite 26 9.1 External Environmen tal Sen sors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 27 9.2 Suit Environmen t S ensors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 27 9.3 Firefigh ter Biometric Sensors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 27 10 St ate-Space F orm ulation 29 10.1 S tate V ector . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 29 10.2 C on trol Input V ector . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 30 10.2.1 Solenoid V alv e Stiction and Lo w-Flo w Nonlinearit y . . . . . . . . . . . . . . . 30 10.3 Distur bance V ector . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31 10.4 Nonlinear State Dynamics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31 11 C on trol Ob j e ctive: Constrained Optimization Under Uncertain ty 33 3 11.1 C ost F unction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 33 11.2 Hard Constraint s . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 34 12 AI Arc hitecture 35 12.1 S ensor F usion and State Estimation . . . . . . . . . . . . . . . . . . . . . . . . . . . 36 12.2 L earn ed Metab olic Mod el . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 36 12.3 Mo d el-Predictiv e Con troller (MPC) . . . . . . . . . . . . . . . . . . . . . . . . . . . 37 12.4 Reinf orcemen t Learning P olicy Advisor . . . . . . . . . . . . . . . . . . . . . . . . . 37 13 Sit uational Aw arene ss Integration 38 13.1 T hermal Thr eat Assessmen t . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 39 13.2 Activit y Classifi cation from Dual IMU . . . . . . . . . . . . . . . . . . . . . . . . . . 39 13.3 I n -Suit At mosph ere Monitoring and Anomaly Detection . . . . . . . . . . . . . . . . 39 13.4 Hazard Pro ximit y Estimation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 40 14 E mergency Proto cols and Graceful Degradation 40 15 Simulation Results 40 15.1 S cenario Descriptions . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 40 15.2 Performance Metrics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 41 16 Discussion 41 16.1 L on g-Duration and Multi-Sortie Op erational Gaps . . . . . . . . . . . . . . . . . . . 43 17 C onclusion 43 4 P art I Chemical and Ph ysical F oundations 1 In tro du c tion Firefigh ters op erate in en vironmen ts filled w ith sm ok e, to xic gases, and extreme temp er atur es, often exceeding 500 ◦ C in s tr uctural fires. Con v enti onal op en-circuit self-cont ained breathing appa- ratus (S CBA) e xh au s t eac h breath to the e nvironmen t, w asting roughly t wo-thirds of the d eliv ered o xygen and limiting op erating time to app ro ximately 30 minutes und er hea vy exe rtion [1 –3]. A closed-circuit br eathing apparatus ( CC BA) recycles exhale d gas, scrub s CO 2 , remov es excess mois- ture, and replenishes consumed O 2 from a finite s upply . Although this approac h is w ell established in military diving and spacecraft life supp ort [4, 5], fatal C O 2 reten tion in ciden ts in rebreather diving demons trate that it is not without risk and dep ends critically on reliable s cr u bbing [6]. If implemen ted safely , it can p oten tially triple effectiv e op erating time wh ile reducing the firefighter’s o v erall carried we ight. This pap er makes t wo con tributions. Part I dev elops th e c hemical and physic al foundations of the Galactic Bio w are Life Supp ort System from fir st principles, including complete th ermo c hemical analyses, adsorption theory , and airflo w dy n amics. P art II introdu ces an AI-based con trol sys tem that uses sensor fusion and online optimizatio n to manage finite con- sumables aga inst unkn o wn m ission du ration and dynamically evo lving fir eground conditions. The system is semi- close d : the p ositiv e-pressure suit incorp orates one-wa y exhaust v alv es (consisten t with NFP A 1991 Lev el A en capsulating suit p ractice) that v ent gas outw ard when in ternal p ressure exceeds a crac king p ressure, prev enting to xic infiltration while allo wing con trolled p ressure relief. This in termittent v en ting create s a slo w net loss of gas fr om the suit, whic h is comp ensated b y O 2 injection—the primary driv er of o xygen enric hment a nd the cen tral con trol c hallenge addressed b y the AI sy s tem. The system emphasizes th ree design constraints: (i) Semi-closed p ositive pressure: The suit v en ts outw ard through exhaust v alv es bu t never admits external air. V en ting depletes the gas inv en tory , requiring make -up O 2 injection that driv es gradual o xygen enric hment. (ii) V ariable metabolic demand: Oxygen consump tion is a n onlinear f u nction of firefighter exertion, ther m al stress, and psyc hological state. (iii) Finite cons umables: System endur ance is b ounded by limited so d a lime (1 kg), silica gel (1 kg), and o xygen supply (3 kg). 2 System Ov erview 2.1 Compo nents and La y out The Galactic Bio ware Life Supp ort Sy s tem comprises the follo wing sub systems: (i) P ositiv e-Pressure Suit with E xha ust V alv es: The s uit maintai n s an internal gauge pressure ∆ P suit = P s − P a > 0 (typica lly 2 mbar to 5 m bar) relativ e to the am bient p ressure P a . One-wa y exh au s t v alv es, consistent with NFP A 1991 Lev el A en capsulating suit design, 5 op en at a crac king pr essure P crac k ≈ P a + 5 m bar to ven t gas ou tw ard, preven ting ov erpressure while ensurin g that an y leak pathw a y resu lts in out w ard gas flo w. This mak es the su it semi- close d : gas is nev er admitted from th e en vironmen t, but is inte rm itten tly v ente d out wa rd . (ii) Air Circulat ion System: Tw o v ariable-speed bru shless DC fans—an outtake fan drawing exhaled a ir fr om the su it interio r and an inta ke f an returning trea ted air—drive a con tin uous flo w through the treatmen t train. (iii) CO 2 Scrubb er: A pac k ed-b ed ca nister of gran u lar so da lime (Ca( OH) 2 /NaOH formulat ion, mean granule diameter 2 mm to 5 mm , with pH-ind icating dye ) remov es exhaled C O 2 through irrev ersible acid–base neutralizatio n. So da lime is the standard sorb en t in closed-circuit breathing s y s tems (rebreathers, anesthesia circuits) b ecause i ts calc ium hydro xide matrix binds the caustic alk ali , prev enting th e formati on of free Na OH solution that could cause airw a y bu rns. This stage is p ositioned first in th e treatmen t train to r eceiv e moist exhaled gas directly , main taining the aqueous su rface film required for efficient scrubb ing (see section 2.2). (iv) Deh umidification Unit: A pac ked-b ed ca n ister of indicating silica g el (Typ e A, mean b ead diameter 2 mm to 5 mm) remo ves wate r v ap or from the circulating gas stream via physica l adsorption. P ositioned downstr e am of the s crubb er, it captures b oth exhaled moisture and reaction-ge n erated moisture in a single pass. (v) Oxygen Replenishmen t System: A prop ortional solenoid v alv e meters gaseous O 2 from a high-pressu re comp osite tank con taining 3 . 0 kg O 2 ( ≈ 93 . 75 mol, ≈ 2100 L at S TP), stored at 200 bar in a ∼ 11 . 7 L cylind er. The tank mass accoun ts for non-ideal gas b eha vior at high pressure. Using the r eal-ga s equation: n = P V Z R T (1) with compressibilit y factor Z ≈ 0 . 95 for oxyg en at 200 bar and 300 K (p er NIST therm o- physic al prop erties d ata for O 2 ), th e total gas mass at fill is appro ximately 3 . 16 kg. Because Z < 1 at this pressure, the cylinder holds mor e gas than an ideal- gas estimate w ould predict. Ho w eve r, the regulat or requir es a minim um inlet pressure of 10 bar to 25 bar to main tain stable d eliv ery , b elo w whic h th e residu al gas (0 . 15 kg to 0 . 38 kg) is un r eco v erable. The design therefore adopts a nominal usable ca pacit y of 3 . 0 kg , corresp ond in g to a minim u m deliv ery pressure of appro ximately 11 b ar. (vi) Coun ter-Lung (Breathing Bag): A flexible b ello ws or collapsible bag connected to the breathing lo op that accommod ates tidal br eathing oscillations and transient mismatc hes b e- t we en O 2 injection an d m etab olic consum p tion at near-constan t pressur e. (vii) Sensor Suit e and AI Con troller: T hree categories of sens ors—external en vironmental, in ternal suit en vironm ent (including trip le-redundant O 2 cells), and firefighter biometric— feed the AI con trol system describ ed in d etail in Part I I. 2.2 Airflo w Pa thw a y The ordering of treat ment stages in the c losed lo op is dictated b y a critical ph ysical-c hemistry constrain t: the so da li me scrubbing reaction r e quir es moisture to p ro ceed, while sim u ltaneously pr o ducing moisture as a bypro duct. This coupling d etermines the optimum top ology . 6 Figure 1: Cont rol-orient ed arc hitecture of the semi-closed p ositiv e-pressure b r eathing lo op. Solid lines illustrate gas fl ow and dash ed lines sensor inpu ts / cont rol links. Moist exhaled gas is drawn through the s o da-lime scrubb er firs t, then the d o wnstream silica-gel deh umidifier, b efore O 2 re- plenishment and return to the b reathing zone. Outw ard v enting through one-w a y exh aust v alv es mak es the suit semi-closed; the con troller uses external, in-suit, and biometric sensing to regulate O 2 injection, f an sp eed, and scrub b er bypass. 2.2.1 Gas-T reatment Stage Ordering One p ossible configuration places the dehumidifier upstream of the scrubb er; ho wev er, th is top olog y is incompatible with the underlyin g physic o chemical requirements for t w o reasons: (i) Scrubb er moisture requiremen t: As detailed in section 3, the CO 2 absorption mec h an ism pro ceeds thr ough an aqueous surface film on the sod a lime gran ules (eq. (10)–eq. (13)). If the inlet gas is aggressiv ely dried, this film desiccates, causing a hard crust of calcium carb onate to form on the gran ule surface. The crust blo c ks gas–liquid con tact, and the effectiv eness factor η ( t ) drops precipitously—p oten tially renderin g the scrubb er non-functional while significan t h ydr o xide remains unreacted in the gran ule interior. (ii) Reaction-generated mois ture : Eac h mole of CO 2 scrubb ed pro d uces one mole of H 2 O (eq. (8)). At hea vy exertion ( ˙ n CO 2 ≈ 0 . 068 mol min − 1 ), this corr esp onds to a w ater pro duction rate of: ˙ m H 2 O , rxn = ˙ n CO 2 × M H 2 O = 0 . 068 × 18 . 015 ≈ 1 . 22 g min − 1 (2) If the desiccant is upstr eam, this reaction-generate d moisture passes u nr emove d into t h e breathing gas deliv ered to the fi refight er. Combined with the moisture that the upstream desiccan t failed to in tercept (b ecause it w as already saturated or b ecause the sc r u bb er added 7 new mo istur e downstream), the suit h umidit y constrain t ( RH ≤ 60%) would b e violated within minutes und er hea vy exertion. 2.2.2 Gas P athw a y The ideal closed-lo op gas pathw a y therefore pro ceeds as f ollo ws: 1. Outtake fan: Exh aled gas (enr ic hed in CO 2 and H 2 O, depleted in O 2 ) is drawn from the suit h elmet and torso plenum. 2. So da lime scrubb er: The moist exhaled gas e nters th e scrubb er directly . The high h u- midit y of exh aled air (90 % to 100 % RH at 34 ◦ C) main tains the aqueous surf ace film on the so da lime gran ules, e n s uring efficien t gas–liquid con tact for CO 2 absorption. The s crubb er sim ultaneously remo v es CO 2 and generates additional H 2 O and heat. 3. Silica gel dehumidifier: The co oled gas, n ow carryin g b oth th e firefigh ter’s exhaled moisture and the scru bb er’s reaction-generated moisture, passes through th e silica gel canister. B ecause this stage sees the total system moisture lo ad, the desiccan t is utilized effic iently—it captures all sources of w ater in a single pass rather than missing the largest con tribu tor. How ev er, w ater adsorption o nto silica gel is itself exothermic: th e e nthalp y of adsorption comprises the laten t heat of condensation ( ∼ 2440 kJ kg − 1 ) plus the exc ess surface energy of sorp tion ( ∼ 100 kJ kg − 1 to 200 kJ kg − 1 ), tota ling approximat ely 2550 kJ kg − 1 of wate r adsorb ed . At the p eak moisture load of 4 . 2 g min − 1 (section 2.2.3), this pro du ces: ˙ Q ads ( t ) ≈ 2550 × 4 . 2 × 10 − 3 60 ≈ 179 W (3) This heat is released p ost-scrubb er directly into the gas stream appr oac hing the fi r efigh ter’s breathing zone. Without a secondary co oling stage, inspired gas temp erature could exceed 45 ◦ C—creating a “hot hair d ry er” effect that accele r ates core temp erature r ise an d thermal injury to th e upp er airw a y . 4. Oxygen injection: F resh O 2 is m etered into the co oled, deh umidified gas stream via the prop ortional v alve . 5. In take fan: The refr eshed gas (low CO 2 , cont rolled humidit y , replenished O 2 , co oled b elo w 35 ◦ C) is circulated bac k i nto t h e suit th rough distribution c hann els in the helmet, torso, and lim bs. The G alactic Bio wa re L ife Supp ort Sys tem is semi-close d : it follo ws the ideal closed-loop gas path wa y , mo dified only b y one-w a y exhaus t v alve s that v ent out w ard in termitten tly when in ternal pressure excee d s the crac king pressure, w hile prev en ting ambien t ga s ingress. The net effe ct is a slo w loss of gas-phase moles from the suit, comp ensated by O 2 injection. This v enting is the primary mec hanism driving o xygen enric h men t in the breathing lo op (see section 6.1.3). 2.2.3 Moisture Budget Analysis T o v erify th at the top ology satisfies the humidit y constraint, we examine the total moisture load on the d o wnstream d esiccan t. The t wo sources of w ater entering the silica gel canister are: 8 (i) Exhaled moisture: A firefighter und er hea vy exertion exhales appr o ximately 1 . 5 g min − 1 to 3 . 0 g min − 1 of w ater v ap or (dep endin g on minute v en tilation and b o d y temp erature). (ii) Scrubb e r reaction pro duct: F r om eq. (2), approxi mately 1 . 2 g min − 1 at hea vy exertion. (iii) T otal moisture load: 2 . 7 g m in − 1 to 4 . 2 g min − 1 , or 160 g h − 1 to 250 g h − 1 . With 1 kg of silica gel at a capacit y of ∼ 350 g of wate r, the desiccan t p r o vides 1 . 4 h to 2 . 2 h of h u- midit y con trol at hea vy exertion—w ell-matc hed to the scrubb er and o xygen supply end u rance. In the incorrect (upstream) top ology , the d esiccan t w ould capture o nly Exhale d moistur e w hile Scrub- b er r e action pr o duct passes directly to the firefigh ter, making the h umidity constr aint unac hiev a b le regardless of d esiccan t capacit y . 3 Carb on Dio xide Managemen t 3.1 Ph ysiological Context Normal atmospheric CO 2 concen tration is approximate ly 0 . 0 4 % by v olume (4 00 ppm). The human b o dy conti nuously pro duces C O 2 as the terminal pro duct of aerobic cellular respiration, transp orted via the blo o d to the lun gs and exhaled at concen trations of 3 . 5 % to 5 . 5 %. In a closed breathing lo op, CO 2 accum ulates unless activ ely remo v ed. T he system m ust main tain inspired C O 2 b elo w 0.5% (5000 ppm). This threshold derives from OSHA’s p ermissible exp osure limit (PEL-TW A, 29 CFR 19 10.1000, T able Z-1) [ 7], whic h is an 8-hour time -weig hted a ve rage for o ccupational am bien t air. A closed-loop breathing app aratus is not an o ccup ational w orksp ace in the regulatory sens e, and no d edicated standard e xists for inspir ed CO 2 in self-con tained life su pp ort systems for firefighting. W e adopt the OSHA PEL as a c onservative op er ational c eiling for three reasons: (i) firefight er deplo yments on a single air fill are su bstan tially shorter than 8 hours , so the TW A a ve raging p erio d is nev er appr oac h ed ; (ii) the PEL aligns with the NIOS H recommended exp osur e limit (REL) of 5000 ppm T W A and with sub marine atmosphere qu ality stand ards (NA VSEA SS 521-AK-HB K-010), pro vidin g cross-d omain consistency; and (iii) the MPC treats this as a soft ceiling with quadratic p enalt y onset w ell b elo w 5000 ppm (nomin al target ∼ 2000 ppm), so the controlle r activ ely minimises inspired CO 2 rather than d w elling at the limit. The hard emergency flo or in casca d e-failure mo de (section 14) is set at 3%, consisten t with s h ort-duration acute exp osure guidance. Remark 1. 1 mmHg is the pr essur e exerte d by a 1 m m c olumn of mer cury at 0 ◦ C u nder standar d gr avity, e qual to appr oxima tely 133 . 322 Pa . 3.2 P erformance Degradation from Hyp erc apnia Excessiv e CO 2 exp osure causes hyp er c apnia —an elev atio n of P a CO 2 ab o ve 45 mmHg . The dissolved CO 2 reacts with w ater in th e blo o d to form carb onic acid: CO 2 (aq) + H 2 O(l) ⇋ H 2 CO 3 (aq) ⇋ H + (aq) + HCO − 3 (aq) (4) This equilibr ium is catalyzed b y the enzyme carbonic anh ydrase (with a tur n o v er n umber of ∼ 10 6 s − 1 ) in red bloo d cells, making the resp onse nearly instan taneous. The r esu lting increa se 9 in [H + ] lo we rs blo o d pH, a condition termed r espir atory acidosis . T he Henderson–Hasselbalc h equation quantifies this relationship: pH = p K a + log 10 [HCO − 3 ] [H 2 CO 3 ] = 6 . 1 + log 10 [HCO − 3 ] 0 . 03 × P a CO 2 (5) where P a CO 2 is measured in mm Hg and 0 . 03 is the solubilit y co efficient of CO 2 in plasma (mmol L − 1 p er mmHg) at 37 ◦ C. Normal arterial pH i s 7 . 35 to 7 . 45; respiratory aci d osis dr iv es pH b elo w 7.35. Symptoms progress w ith severit y: at inspired CO 2 of 2 % to 3 % , headac he and impaired ju dgmen t; at 5 % to 7 %, confu s ion, tac h ycardia, and dysp nea; ab ov e 10 %, loss of consciousness and death within minutes [8]. 3.3 Chronic Health E ffects Rep eated subacute exp osure pro d uces chronic h yp ercapnia with renal comp ensation (elev ate d serum HCO − 3 ), placing sustained strain on the cardio v ascular system and increasing hyp ertension risk. Neur ologica l effects of c hronic hyp ercapnia include imp aired memory , concen tration deficits, and accelerat ed cognitiv e decl ine. In closed-circuit breathing en viron m en ts sp ecifically , rep eated CO 2 exp osure alte rs respiratory driv e and reduces C O 2 sensitivit y [9]. Rep eated o ccupational exp osure to elev at ed CO 2 in closed-circuit breathing environmen ts has also b een asso ciated with blunte d v entil atory c hemosensitivit y [10], whic h ma y mask early warning signs of scrubb er failure in exp erienced users. This observ atio n strengthens the requirement for direct in strumente d mon- itoring of scrub b er p erformance, b ecause physiolog ical p erception of risin g inspired CO 2 ma y b e atten uated in exp erienced user s . 3.4 CO 2 Scrubbing wit h So da Lime 3.4.1 Sorb en t C hoice and Safety The G alactic Bio wa re L ife Supp ort Sys tem uses so da lime —a gran ular form ulation of ca lcium h ydr o xide (Ca(OH) 2 , ∼ 75–80% by mass) with a small fraction of so d ium or p otassium h yd r o xide ( ∼ 3–5%) as an activ ator, plus wa ter ( ∼ 15–20%) and a pH-indicating dye. So da lime is the standard CO 2 absorb ent in closed-circuit breathing systems (military and recreational r ebreathers, anesthesia circuits, sub marine atmosphere managemen t) b ecause: 1. The calcium hydro xide matrix physica lly binds the alk a li hydro xide, prev enting formatio n of free caustic solution that could m igrate to the breathing zone and cause c hemical bu rns to the airwa y . 2. The granular f orm m aintains stru ctural in tegrit y throughout the reactio n , with less tendency to form fi ne particulate th an pur e NaOH p ellets. 3. The built-in m oisture conten t mainta ins th e aqueous su rface fi lm requir ed for efficient gas– liquid con tact without relying on external humidit y alone. Remark 2 (Wh y not pure NaOH?) . Pur e so dium hydr ox ide p e l lets ar e highly hygr osc opic and c or- r osive (pH 13–14 in solution). In a br e athing lo op , th e c ombination of high humidity, me chanic al vibr ation, and exo thermic r e action c an pr o duc e a mo bile c austic liquid—the “c austic c o cktail” do cu- mente d in r ebr e ather diving incident r ep orts. This r epr esents an unac c ept able airway inju ry ha zar d for fir efighting. So da lime mitigates this risk thr ough the c alcium hydr oxide matrix and c ontr ol le d formulation. 10 3.4.2 Primary Reaction and Thermo c hemistry The C O 2 absorption in sod a lime pro ceeds through a tw o-stage mec hanism. Th e NaOH activ at or reacts first (faster kinetics) and is regenerated b y the bulk C a(OH) 2 : Stage 1: NaOH-catalyzed absorption: CO 2 (g) + 2 NaOH(aq) − → Na 2 CO 3 (aq) + H 2 O(l) (6) Stage 2: Regeneration of NaOH by Ca(OH) 2 : Na 2 CO 3 (aq) + Ca(OH) 2 (s) − → CaCO 3 (s) + 2 NaOH(aq) (7) The NaOH is recycled; how ev er, b ecause the regeneration step (eq. (7)) is slow er than the in itial absorption (eq. (6 )), the NaOH can b ecome tran s ien tly dep leted at high CO 2 loading rates, tem- p orarily reducing the e ffectiv e scrubbin g rate unt il the Ca(OH) 2 regeneration catc hes up. The net reaction is: CO 2 (g) + Ca(OH) 2 (s) − → CaCO 3 (s) + H 2 O(l) (8) State-consisten t enthalp y via formation en thalpies: Using sta nd ard e nthalpies of f orm ation at 298 . 15 K: Sp ecies ∆ H ◦ f ( kJ mol − 1 ) CO 2 (g) − 393 . 5 Ca(OH) 2 (s) − 986 . 1 CaCO 3 (s) (calcite) − 1206 . 9 H 2 O(l) − 285 . 8 Applying Hess’s la w with well-defined initial and final states: ∆ H ◦ rxn = ∆ H ◦ f (CaCO 3 (s)) + ∆ H ◦ f (H 2 O(l)) − ∆ H ◦ f (CO 2 (g)) + ∆ H ◦ f (Ca(OH) 2 (s)) = [( − 1206 . 9) + ( − 285 . 8)] − [( − 393 . 5) + ( − 986 . 1)] = − 1492 . 7 − ( − 1379 . 6) = − 113 . 1 kJ / molCO 2 (9) This v alue is uniquely determine d by the standard-state endp oints of the net reaction and is path- indep end en t (Hess’s la w). No v alue range is needed for the standard enthalp y itself—uncertaint y in the op erating h eat release arises from non-standard conditions (elev ate d temp erature, v arying h ydr ation state of the prod uct) and is add ressed through engineering s afet y factors in th e ther- mal managemen t design rather than by adjusting the thermo dyn amic v alue. F or comparison, the analogous rea ction with pure NaOH yields ∆ H ◦ rxn ≈ − 171 kJ mol − 1 if w ater co n d enses to liquid, or − 127 kJ mol − 1 if w ater remains v ap or. The so da lime reaction is less exot her m ic p er m ole of CO 2 , whic h is a thermal man agement adv an tage. 11 3.4.3 Reaction Mec hanism and Kinet ics The gas–solid react ion pro ceeds through a m ulti-step mec hanism: Step 1: Dissolution of CO 2 in to the surface water film: So d a lime gran ules main tain a thin aqueous film d ue to their built-in moisture cont ent ( ∼ 15–20% w ater). In pure w ater, CO 2 dissolv es according to Henry’s la w: [CO 2 (aq)] = K H · p CO 2 (10) where K H = 3 . 4 × 10 − 2 mol L − 1 atm − 1 at 298 K for pure wat er (solubilit y conv en tion; [ CO 2 ] = K H · p ). Ho w ever, the so da lime surface film is a concen trated alk aline solution (pH 12–14 ), in whic h dissolv ed CO 2 is rapidly consumed by react ion with OH − (Steps 2–3). The result is r e action-enhanc e d abso rption : the effectiv e gas-phase driving force is m uc h larger than the bare Henry’s l aw equilibriu m would pr edict, b ecause the liquid-side CO 2 concen tration is h eld n ear zero b y fast c hemical consumption. This r egime is c h aracterized by a Hatta num b er Ha ≫ 1, and th e absorption rate is go ve r n ed by the pro duct K H p k OH [OH − ] D CO 2 rather than by K H alone, where k OH is the second-order rat e constan t for the CO 2 +OH − reaction and D CO 2 is the liquid-ph ase diffusivit y . Th e o v erall v olumetric scru bbing rate (e q. ( 15)) abs orb s this enhancemen t in to the lump ed co efficient k o v . Step 2: F ormation of carb onic acid and disso ciation: CO 2 (aq) + H 2 O(l) ⇋ H 2 CO 3 (aq) ⇋ H + (aq) + HCO − 3 (aq) (11) with K a 1 = 4 . 3 × 10 − 7 at 298 K. Step 3: Neutralization by h ydro xide ions: H 2 CO 3 (aq) + OH − (aq) − → HCO − 3 (aq) + H 2 O(l) (12) HCO − 3 (aq) + OH − (aq) − → CO 2 − 3 (aq) + H 2 O(l) (13 ) Step 4: Precipitation of calcium carb onate: Ca 2+ (aq) + CO 2 − 3 (aq) − → CaCO 3 (s) (14) The rate-limiting step und er typica l op erating conditions is the gas-phase mass transfer of CO 2 to the gran u le su r face (Step 1), which d ep ends on th e gas-phase velocit y , gran ule s urface area, and the dr iving force p CO 2 , bulk − p CO 2 , surface . W e m o del the ov erall vo lum etric r ate of CO 2 remo v al as: r scrub = k o v a s V bed p CO 2 − p ∗ CO 2 η ( t ) (15) where k o v is the o veral l mass transfer co efficien t (mol s − 1 P a − 1 m − 2 ), a s is the sp ecific surface area of the pac ke d b ed (m 2 m − 3 ), V bed is the b ed v olume, p ∗ CO 2 is the equilibrium partial p ressure ov er the pro d uct la yer (effectiv ely zero for fresh so da lime), and η ( t ) ∈ [0 , 1] is an effectiv eness factor that decreases as the Ca(OH) 2 is consum ed and the pro duct la ye r of CaCO 3 builds u p on granule surfaces, creating a diffusion barr ier. 12 3.4.4 Stoic hiometric C apacit y F rom the net reactio n (eq. (8)), 1 mole of Ca(OH) 2 consumes 1 mole of CO 2 . With the molecular masses M Ca(OH) 2 = 74 . 09 g mol − 1 and M CO 2 = 44 . 01 g mol − 1 , the scru bbing capacit y dep end s on the a v ailable Ca( OH) 2 in the as-pac ked canister. So da lime as pac k ed conta ins ∼ 15–20 % w ater b y mass (required for the aqueous film that enables s crubbing); the d r y mass fraction is therefore ∼ 0.80–0 .85 of th e total, and Ca(OH) 2 constitutes ∼ 75–80% of the dry mass: m CO 2 , max = f dry × f Ca(OH) 2 × m soda lime M Ca(OH) 2 × M CO 2 = 0 . 82 × 0 . 77 × 1000 g 74 . 09 g mol − 1 × 44 . 01 g mol − 1 ≈ 375 g (16) where f dry ≈ 0 . 82 is th e d ry m ass fraction (assuming 18% water, mid-range) and f Ca(OH) 2 ≈ 0 . 7 7 is the Ca(OH) 2 fraction of dry mass. The canister can absorb a maxim u m of ∼ 375 g of C O 2 . A t a mo derate metab olic CO 2 pro du ction rate of appro ximately 200 m L min − 1 (STP), corresp onding t o ˙ m CO 2 ≈ 0 . 39 g min − 1 , the scru bb er lifetime is: T scrubber ≈ 375 0 . 39 ≈ 962 min ≈ 16 h (at rest) (17) Under h ea vy exertion, ˙ V CO 2 can r ise to 2 . 0 L min − 1 to 3 . 0 L m in − 1 , reducing s crubb er life to ap- pro ximately 1 . 0 h to 1 . 6 h. 3.4.5 Thermal Managemen t F rom eq. (9), the heat generation r ate in the scrub b er is: ˙ Q scrub ( t ) = | ∆ H ◦ rxn | × ˙ n CO 2 , scrubb ed ( t ) (18) where ˙ n CO 2 , scrubb ed is the m olar scrubbing rate. A t heavy exertion ( ˙ n CO 2 ≈ 0 . 068 mol min − 1 ): ˙ Q scrub ≈ 113 . 1 × 0 . 068 60 kJ s − 1 ≈ 128 W (19) Com bined with the desiccan t heat of adsorption ( ˙ Q ads ≈ 179 W at p eak, e q. (3)), the total in ternal heat generation from the treatmen t train reac hes up to ∼ 307 W at heavy exertion. Wh y no external heat exchanger is used: In a stru ctural fi re, the external en vironment is frequent ly hotter than the breathing loop. An y attempt to reject h eat to the suit shell can rev erse sign and add h eat to the lo op wh en T ext > T lo op . F or this reason th e design does not r ely on a shell- coupled heat exc hanger for cooling; thermal safet y is instead managed b y (i) limiting in ternal heat generation via con trol (fan sp eed, scrubb er b ypass), and (ii) con trolled out w ard ven ting strategies describ ed b elo w. Supplemen tary ev ap orativ e v enting: F or extended m issions, the system is supplemen ted b y c ontr ol le d evap or ative venting : a small amount of w ater (condensed from the desiccan t or from a se p arate reserv oir) is rele ased through a one-wa y v a lve to the exterior, where it ev ap orates, carrying a w a y ∼ 2440 kJ kg − 1 . This b reaks the “fu lly closed” thermal c onstraint a t a mo dest w ater cost ( ∼ 6 g min − 1 to reject 250 W ). Design mitigations include: • Gran ular soda lime b ed with controll ed v oid fractio n ( ε ≈ 0 . 35–0 . 45) to allo w co nv ectiv e heat remo v al by the circulating gas. 13 • Thermal fuse: if the scru bb er b ed thermocouple r egisters T bed > 80 ◦ C, the con troller au- tomatica lly in creases scr u bb er bypass fraction φ b ypass and fan sp eed, trading te mp orarily elev ated CO 2 for ther m al safet y . The transient temp erature of the scrubb er b ed can b e mo deled u sing an energy balance: ( ρc p ) bed V bed dT bed dt = ˙ Q scrub ( t ) − ˙ m air c p, air ( T bed − T air , in ) − U A wa ll ( T bed − T wa ll ) (20) where ( ρc p ) bed is th e effectiv e v olumetric heat capacit y of the pac k ed b ed, U is the ov erall heat transfer co efficien t to the canister w all. 4 Humidit y Managemen t 4.1 Source of Moisture and In teract ion wit h Scrubb er Placem ent In a closed-circuit system, wat er v ap or has t wo distinct sour ces, b oth of wh ic h must b e managed b y th e desiccan t: (i) Metab olic moisture: The firefighter’s exhaled br eath an d insensible p ers p iration. A r esting adult exhales approximat ely 200 mL to 400 mL of liquid-equiv alen t w ater p er d a y via respi- ration; und er hea vy exertion and thermal stress, this ca n increase fiv e-fold or m ore, reac hing 1 . 5 g min − 1 to 3 . 0 g min − 1 . (ii) Reaction-generated mois t ure : The so da lime scrubbin g reaction (eq. (8)) pro d uces one mole of H 2 O p er mole of CO 2 absorb ed. A t hea vy exertion, th is con tribu tes an add itional ∼ 1 . 2 g min − 1 (eq. (2)), represen ting 30 % to 45 % of the tota l moisture load. As discussed in sec tion 2.2, the silica gel desicc ant is p ositioned downstr e am of the scru bb er so that it in tercepts b oth sources in a single pass. This top ology also pr eserv es the moist gas en vironm en t required for efficien t scru bb er op eration. The system target s a relativ e humidit y (RH) b elo w 60 % at the suit breathing zone, measured by the in-suit capacitiv e RH sen s or do wns tr eam of the des- iccan t stage. Excessiv e h u midit y causes visor f ogging (onset at RH > 80% on co ol visor surfaces), skin ma ceration, and reduced ev ap orativ e co oling efficie n cy—f u rther ele v ating the firefigh ter’s core temp erature in an already thermally h ostile environmen t. 4.2 Silica Gel Adsorption: Ph ysical Chemistry Silica gel (S iO 2 · nH 2 O) is an amorp hous, highly p orous form of silicon dioxi d e. Its int ern al sur - face area—t ypically 600 m 2 g − 1 to 800 m 2 g − 1 —pro vides abun dan t sites for ph ysical adsorption (ph ysisorp tion) of wate r molecules via h ydrogen b onding with surface silanol (Si-OH) groups. 4.2.1 Adsorption Isotherm The equilibrium m oisture uptak e of silic a gel as a fu nction of r elativ e h umid it y exhibits strong m ultila yer adsorption and capillary condensation b eha vior, particularly ab o ve ∼ 40% RH where 14 the h um idit y con trol constraint operates. The simp le Langm uir isotherm (monola yer, asymptotic saturation) systematic ally un derpredicts u ptak e in this regime. W e therefore use the Guggenheim– And erson–de Bo er (GAB) isotherm, a th ree-parameter extension of BET theory that is standard in adsorption engineering for wate r/silica gel systems: q e = q m C G K G a w (1 − K G a w )(1 − K G a w + C G K G a w ) (21) where q e is the equilibriu m loading (kg wat er p er kg dry silica), a w = RH / 100 is the water ac- tivit y , q m is the monola ye r capacit y , C G is the Gu ggenheim c onstant (related to the enthalp y difference betw een m on olay er and multila y er adsorption), and K G is a m u ltila y er correction factor. F or T yp e A silica gel a t 25 ◦ C, r epresen tativ e parameters are q m ≈ 0 . 10, C G ≈ 40, K G ≈ 0 . 85, yielding q e ≈ 0 . 30–0 . 3 5 at 80% RH—consisten t with pub lish ed d ata. All three parameters are temp erature-dep en den t (decreasing q e with increasing temp erature at constan t RH), which is cap- tured by Arrhenius-t yp e expressions fitted to man ufacturer d ata. 4.2.2 Adsorption Dynamics and Linear Driving F orce Mo del The rate of w ater u ptak e is go v erned b y intraparticle diffusion through the p ore netw ork. Using the linear d riving force (LDF) appro ximation: d ¯ q ( t ) dt = k LDF q e RH in ( t ) − ¯ q ( t ) (22) where ¯ q ( t ) is the a verag e loading on the gel a t time t and k LDF is the LDF ma ss trans f er co efficien t, whic h depen ds on effectiv e p ore d iffusivit y D e , b ead radius R p , and the a pp ro ximation k LDF ≈ 15 D e /R 2 p . 4.2.3 Mass Balance for t he P ac k ed Bed F or the silica gel canister, the macroscopic w ater mass balance is: dM wa ter ( t ) dt = ˙ m air Y in ( t ) − Y out ( t ) (23) where Y in ( t ) and Y out ( t ) are the h umid ity rat ios (kg wate r p er kg dry air) at the ca n ister inlet and outlet, resp ectiv ely , and ˙ m air is the dry-air mass fl o w rate. The total silica canister mass evolv es as: M silica ( t ) = M silica , dry + M wa ter ( t ) , 0 ≤ M wa ter ( t ) ≤ M wa ter , max (24) As M wa ter ( t ) → M wa ter , max ≈ 0 . 35 kg (for 1 kg of d ry silica gel), Y out → Y in and the desiccan t is effectiv ely saturated. Expressing the dr iving force in terms of partial pressu res: dM wa ter ( t ) dt = ˙ m air α p H 2 O , in ( t ) − p H 2 O , out ( t ) (25) where α is a lump ed pr op ortionalit y constan t accoun ting for the psyc h r ometric r elationship b et w een h um idit y ratio and partial pr essure at the system’s op erating temp eratur e and total p ressure. 15 4.2.4 Heat of Adsorption and the T otal Thermal Budget W ater adsorption on to sili ca gel is exothermic. Th e enthalp y o f adsorption ∆ H ads includes the laten t heat of conden sation of w ater v ap or (∆ H v ap ≈ 2440 kJ kg − 1 at 35 ◦ C) p lus the exc ess heat o f surface binding (∆ H excess ≈ 100 kJ kg − 1 to 200 kJ kg − 1 for T yp e A silica gel, dep ending on loading). The total enthalp y of adsorption is therefore: ∆ H ads ≈ 2500 kJ / kgH 2 O to 2600 kJ / kg H 2 O (26) The heat generation rate in the d esiccan t b ed is: ˙ Q ads ( t ) = ∆ H ads × dM wa ter ( t ) dt (27) A t p eak m oisture load (4 . 2 g min − 1 , see sectio n 2.2.3), this yields ˙ Q ads ≈ 179 W—a heat load comparable to the scrub b er’s output (128 W at hea vy exertion). The com bined thermal output of b oth pac k ed b eds is therefore up to ∼ 307 W (section 3.4.5). T he desiccant b ed temp erature can b e mo d eled analogously to eq. (20): ( ρc p ) silica V silica dT silica dt = ˙ Q ads ( t ) − ˙ m air c p, air ( T silica − T air , in ) − U silica A wa ll , silica ( T silica − T wa ll , silica ) (28) 5 Oxygen Replenishmen t 5.1 Metabolic Basis Aerobic metab olism is summarized by the o xidation of glucose: C 6 H 12 O 6 (aq) + 6 O 2 (g) − → 6 CO 2 (g) + 6 H 2 O(l) ∆ H ◦ = − 2803 kJ mol − 1 (29) The respiratory e xchange r atio (RER), defin ed a s R = ˙ V CO 2 / ˙ V O 2 , ranges from ∼ 0 . 7 (pu re fat o xidation) to 1 . 0 (pure carb oh yd rate o xidation) and exceeds 1.0 ab ov e the r espiratory comp ensation p oint , when excess CO 2 is pr o duced from b icarb onate bu ffering of lactic acid. At mo derate exertion, R ≈ 0 . 85. 5.2 Oxygen Consumption Rates A firefighte r ’s metab olic O 2 consumption rate, ˙ V O 2 , dep end s strongly on work rate W ( t ): ˙ V O 2 ( t ) = ˙ V O 2 , rest + γ W ( t ) + β [ W ( t )] 2 (30) where ˙ V O 2 , rest ≈ 0 . 25 L min − 1 (STP), γ and β are s ub ject-sp ecific co efficients capturing the ad- ditional o xygen cost at high exertion (the ˙ V O 2 slo w comp onent, reflecting increased reli ance on fast-t witc h motor u nits ab o v e t h e lactate threshold), and W ( t ) is the instan taneous meta b olic w ork rate (W). P eak ˙ V O 2 for an elite firefighter can r eac h 3 . 0 L min − 1 to 4 . 0 L min − 1 , corresp ondin g to a mass consu m ption rate of: ˙ m O 2 = ˙ V O 2 × M O 2 V m = ˙ V O 2 × 32 . 00 22 . 414 ≈ 1 . 43 ˙ V O 2 [g min − 1 ] (31) 16 5.3 Oxygen T ank Endurance The fin ite oxyg en sup ply of 3 . 0 kg constrains mission du ration: Z T max 0 ˙ m O 2 , inject ( t ) dt ≤ 3000 g (32) The O 2 injection rate m ust comp ensate for b oth metab olic consump tion and the gas lost throu gh ex- haust v alve v en ting. A t p eak metab olic consump tion alone (4 . 0 L m in − 1 STP), ˙ m O 2 ≈ 5 . 7 g min − 1 , giving a metab o lic- only end urance of 3000 / 5 . 7 ≈ 526 m in. Ho w eve r , the exhaust v alv e ve nting creates an additional O 2 demand: eac h ven t ev en t exhausts gas at the cu r ren t lo op comp osition, whic h m ust b e replaced with p ure O 2 . The effectiv e en durance depen d s o n the v en ting rate, whic h in turn dep ends on thermal transien ts, b o d y m ov emen t, and the pressur e c ontrol strategy . A t mo derate sus tained exertion ( ˙ V O 2 ≈ 2 . 0 L m in − 1 ) with a t ypical ven t loss of ∼ 0 . 5 L min − 1 (equiv- alen t), the com bined O 2 consumption is ˙ m O 2 ≈ 3 . 6 g min − 1 , yiel d in g an en d urance of ∼ 830 min ( ∼ 14 hours). At hea vy sustained exertion with frequent ven ting, endurance drops to ∼ 3 h to 5 h— still substanti ally exceeding t h e 30-min u te limit of o p en-circuit SCBA. This regime is precisely where in tellig ent con trol adds v alue: the MPC optimizes the tradeoff b et ween O 2 conserv atio n, v ent frequency managemen t, and physiolo gical safet y . 5.4 Oxygen T oxicit y Considerations In a pu re-O 2 replenishment system, the in s pired partial pressur e of O 2 ( P i O 2 ) m u st b e carefully con trolled. A t sea- leve l ambien t pressure ( ∼ 1 atm), breathing 100 % O 2 yields P i O 2 ≈ 1 . 0 atm. Prolonged exp osure ab o ve P i O 2 > 0 . 5 atm risks pulmonary oxygen toxicity (Lorrain Smith effect) [11], while P i O 2 > 1 . 6 atm ca n precipitate c entr al nervous system (CN S) oxygen toxicity with seizures [5, 12–14]. The con trol system must therefore main tain P i O 2 within a safe band: 0 . 19 atm ≤ P i O 2 ( t ) ≤ 0 . 50 atm (33) corresp ondin g to an inspired O 2 fraction of appro ximately 19 % to 50 % at 1 atm. 5.5 Oxygen Enrich ment as a Fire Hazard The pap er’s O 2 to xicit y constrain t ( P i O 2 ≤ 0 . 50 atm , or ∼ 50% O 2 ) is insufficien t as the sole upp er b ound on o xygen fraction. Per NFP A standards a n d oxygen-system safety engineering (NASA, ASTM), an atmosphere ab o ve ∼ 23.5% O 2 b y v olume is classified as oxygen-enriche d , with signifi- can tly incr eased fi re risk: materia ls that are self-extinguishing in air may burn vigorously , ignition energies decrease, an d flame propagation r ates increase. In a fir efigh ting su it op erating in a fir e envir onment , this risk is comp ounded. The O 2 fraction in the b reathing lo op m u st th er efore be constrained by fir e safety as w ell as to xicit y: x O 2 ( t ) ≤ x O 2 , fire = 0 . 235 (34) where x O 2 , fire = 0 . 235 is the o xygen-enric hed-atmosphere threshold us ed here for fire safet y , based on OS HA guidance [7]. This is a substan tially tighter constr aint t h an the to xicit y limit of 0.50 and fundamentall y shap es the control problem. Th e fi re-safet y limit is the binding upp er constrain t under normal op eration; the to xicit y limit serv es as a hard b ac kup for degraded-mo de op eration where mainta inin g 23.5% is no longer feasible. 17 Remark 3 (Implications f or system arc hitecture) . The fir e-safety O 2 limit str ongly motivates c on- fining the O 2 -enriche d br e athing gas to a small in ternal breathing loop (mask, hoses, c ounter-lung, tr e atment tr a in) r ath er than flo o ding the entir e suit interior vol ume. In fu tu r e design iter ations, a sep ar ate low-O 2 gas (filter e d air or N 2 -enriche d mix) c ould pr essurize the suit shel l, with th e br e athing lo op isolate d by a mask/mouthpie c e. This p ap er analyzes th e simpler sing le- atmospher e ar c hite ctur e and applies the 23.5% c onst r aint to the ful l suit volume. 6 Airflo w Ph ysics and Circulat ion 6.1 P ositive P ressure Main tenance and Gas Inv en tory Dynamics 6.1.1 Correct Gas-Phase Molar Bo okk eeping In a closed-circuit breathing system with c hemical CO 2 scrubbin g, the gas-phase molar in ve ntory ev olv es as follo ws. C on s ider one metab olic–scrubbin g cycle: 1. The firefi gh ter consumes 1 mol of O 2 from the gas phase (up tak e into b lo o d). G as m oles: − 1. 2. The firefight er exhales R mol of CO 2 (where R ≈ 0 . 85 is the respiratory exc hange ratio) into the gas phase. Gas moles: − 1 + R . 3. The scrubb er r emo v es the R m ol of CO 2 from the gas ph ase, con v erting it to solid CaCO 3 . Gas moles: − 1 + R − R = − 1. 4. The O 2 injection system add s 1 mol of O 2 . Gas moles: − 1 + 1 = 0. The net c hange in gas-phase moles p er cycle is zero —the CO 2 pro du ced by metab olism and remo v ed by scrubbing exactly cancel in the molar b o okk eeping, and the consumed O 2 is replaced b y injection. Th ere is no in heren t “molar sink” fr om the scrub bing c hemistry in a sealed sy s tem with 1:1 O 2 replacemen t. Remark 4 (Common er r or in molar-sink analyses) . A fr e quently enc ounter e d err or is to tr e at the scrubb er as r emoving CO 2 fr om the lo op ’s initial inventory r ather than fr o m the CO 2 just pr o duc e d by metab olism. This double-c ounts the CO 2 : the b o dy add s R mol to the gas phase and the scrubb er imme diat ely r emoves R mol fr om the gas phase. The net e ff e ct on gas-phase moles is zer o fr om the CO 2 p at hway. The only gas-phase deficit is the − 1 mo l O 2 c onsume d, which is exactly c omp ensate d by inje ction. 6.1.2 Exhaust V alve V enting: The Real Driv er of Gas In ven tory Change In the Galactic Bio w are semi-closed su it, the exhaust valves are the pr imary mechanism th at changes the gas-phase inv en tory . When internal p ressure exceeds the c r acking pressu re P crac k , ga s v en ts out wa rd . Th e v alv e b eha v es as a compressib le orifice; in the subsonic regime (applicable for the small ∆ P in v olv ed), the mass fl o w rate follo ws: ˙ n v ent ( t ) = C d A v ¯ M q 2 ρ s max 0 , P s ( t ) − P crac k (35) where C d is the v alv e d ischarge co efficient, A v is the effectiv e v alve orifice area, ¯ M is th e mean molar mass of the lo op gas, and ρ s is the gas densit y at suit conditions. The √ ∆ P dep en dence— rather than a linear relati onsh ip—is the standard orifice flo w la w and is imp ortan t b ecause the O 2 18 enric hment analysis (section 6.1.3) is dir ectly sensitive to the ven t r ate fu nctional f orm. The v ented gas has the cu rr ent lo o p c omp osition : a mixture of N 2 , O 2 , r esidual CO 2 , and water v ap or. Th is depletes all sp ecies prop ortionally to their mol e fractions. Pressure rises (trigge rin g ve nting) occur due to: • Body mo v ement compressing th e suit volume (b endin g, crouc hing, impact) • Thermal expansion of the gas fr om external heating • O 2 injection temp orarily exceeding metab olic consu mption • Tidal exhalation p eaks 6.1.3 Oxygen Enric hmen t from V ent Comp ensation Because v en ted gas con tains the current N 2 /O 2 mixture but is replaced with pur e O 2 , eac h v en t– refill cycle in creases the O 2 mole fraction. The enr ic hment dynamic is: dx O 2 dt = (1 − x O 2 ) ˙ n O 2 , inject − ˙ n O 2 , consumed n total (36) Under the p ressure-holding approximat ion ˙ n O 2 , inject ≈ ˙ n O 2 , consumed + ˙ n v ent , the enrichmen t dy n amics reduce to: dx O 2 dt ≈ ˙ n v ent n total (1 − x O 2 ) (37) F or a reactiv e pac k ed b ed, th e appropriate sizing relation is obtained from a plu g-flo w absorb er form ulation: d ˙ n CO 2 dz = − k o v a s A c p CO 2 − p ∗ CO 2 (38) In tegrating along the b ed length yields the required num b er of transfer u nits (NTU): ln p CO 2 , in − p ∗ CO 2 p CO 2 , out − p ∗ CO 2 ! = NT U = k o v a s V bed ˙ V circ (39) The circulation rate is therefore determined b y requir ed NTU and mass-transfer kinetics rather than by simple dilution assump tions. Th e enrichmen t rate is pr op ortional to th e v ent r ate , not to the metab olic or scru bbing rate. Starting from air ( x O 2 = 0 . 21), th e O 2 fraction rises gradually as N 2 is diluted. The time to reac h the fire-safet y limit of 23.5% dep ends str ongly on the ven ting frequency , w hic h is controlle d b y b o dy m ov emen t p atterns, thermal transients, and the pressu r e con trol strateg y . F or a lo op co ntaining n total ≈ 4 mol ( ∼ 95 L to 100 L at 1 atm, 35 ◦ C) and an upp er-b ound a v erage v en t rate of ∼ 0 . 05 m ol min − 1 (equiv ale nt to ∼ 1 . 1 L min − 1 STP): dx O 2 dt x O 2 =0 . 21 ≈ 0 . 05 4 (1 − 0 . 21) ≈ 0 . 010 min − 1 (40) The fir e-safet y limit of x O 2 = 0 . 235 would b e reac hed in appro ximately 0 . 025 / 0 . 01 0 ≈ 2 . 5 min- utes at this su s tained v ent r ate. This sh ould b e int erp reted as a b ounding w orst-case: ˙ n v ent ∼ 0 . 05 mol min − 1 corresp onds to ∼ 1 . 1 L min − 1 STP-equiv alen t, wh ic h is substan tially higher than 19 t ypical leak/v en t rates rep orted for positive -pr essure suits a t 2 mbar to 5 m bar o v erpr essure. I f the effectiv e ven t rate is closer to 0 . 1 L min − 1 to 0 . 3 L min − 1 equiv alen t, the enrichmen t timescale increases pr op ortionally to ∼ 8 min to 25 min for the same loop v olume. This is an extremely tigh t timeline that renders the single-atmosphere architec tur e op erationally marginal for an ything b e- y ond v ery short deploymen ts with minimal b o dy m o v ement (and hence m in imal ven ting). This result should b e understoo d as a bounding analysis : the sin gle-atmo sp here design repre- sen ts the w orst case for O 2 enric hment because the en tire suit v olume ( ∼ 100 L) participates in the ven t–refill cycle. Typica l positiv e-pressu re suits exhibit leak/v en t rates closer to 0 . 1 L min − 1 to 0 . 3 L min − 1 at 2 mbar to 5 mbar o v erpressu re. The 0 . 05 mol min − 1 v alue therefore r ep resen ts a stress-test s cenario rather than a nominal op erating condition. A separated arc h itecture (breathing lo op confin ed to mask/coun ter-lung/treatmen t train at ∼ 10 L, with suit pressurization via an inert or filtered-air source) would reduce the enr ic hment rate by roughly an order of magnitude. The AI con troller’s ability to manage the enric hmen t–pressur e tradeoff is therefore necessary but lik ely insufficient on its o wn; the separated arc hitecture discussed in the future-wo r k sec tion is the viable path to fielded hardware. Nev ertheless, the con trol p roblem structure and the MPC formulati on remain v alid regardless of the lo op vo lum e, s o we analyze the harder single-atmosphere case: • High p ositiv e pressure margin : b etter suit in tegrit y p r otectio n but more fr equen t v ent ing, faster O 2 enric hment. • Lo w p ositiv e pressure margin : reduced ven ting and slo w er enric hment b ut in creased risk of ambien t gas infi ltration through s uit imp erf ections. The MPC optimizes this tradeoff in real time. 6.1.4 Coun ter-Lung Dynamics and Suit Pressure The coun ter-lung accommod ates transient v olume fluctuations. Rat h er than treating suit pressu r e P s as an in dep end ent dy n amic sta te—which w ould create a differenti al-algebraic in consistency with the appro ximately isobaric regi me enforced by the co mp liant coun ter-lung—w e mo del P s algebraical ly fr om the count er-lun g’s ela stic restoring force: P s ( t ) = P a ( t ) + k CL V CL ( t ) − V CL , 0 (41) where k CL is the counter-lung stiffness (Pa L − 1 ), a small v alue for a complian t b ello ws (typical ly 50 P a L − 1 to 200 Pa L − 1 ), and V CL , 0 is the n eutral volume. The exh aust v alv e op ens when P s > P crac k , i.e., w hen V CL exceeds th e threshold V CL , 0 +( P crac k − P a ) /k CL . This coup les the v ent dyn amics naturally to the count er-lun g state withou t r equiring a separate pressu re ODE. The coun ter-lung v olume ev olv es according to: dV CL dt = R g T suit P s ˙ n O 2 , inject − ˙ n O 2 , consumed − ˙ n v ent | {z } net molar chang e + V gas ( t ) T suit ( t ) dT suit dt | {z } thermal expansion + ˙ V breath ( t ) (42) The first term captur es the net molar balance: O 2 injected min u s O 2 consumed minus gas v ent ed. When the cont roller trac ks metab olic consum ption pr ecisely and v enting is minimal, this term is near zero and the counter-lung v olume remains stable. Th e thermal expansion term accoun ts for gas exp an s ion/con traction with temp erature. ˙ V breath ( t ) i s the oscillatory tidal b r eathing flow term. Thermal expansion comp ensation in the EKF: The r a w coun ter-lung p osition measurement V meas CL ( t ) conflates molar c hanges with therm al expansion. In a cata stroph ic fire scenario wh ere 20 external temp eratures escalate r apidly , the thermal expansion term can inflate the counter-lung ev en a s g as is b eing lost th rough v enti n g. The EKF i n corp orates the thermal expans ion te rm directly in its process mo d el, using the in-suit temp erature measurements ( T suit , bz and T suit , torso ) to separate thermal effects fr om molar c hanges. This cross-mod al consistency chec k—v olume sen s or v ersus temp erature sensors v ersus gas c omp osition sensors—is a k ey adv an tage of the m ulti-sensor EKF arc hitecture. 6.2 In ternal Circulation: F an Dynamics The fans do n ot exc hange mass with the environmen t; they circulate gas thr ough the tr eatmen t train. The v olumetric flo w r ate through the closed loop is: Q circ = ∆ P fan R sys (43) where ∆ P fan is the fan’s develo p ed pr essu re and R sys is the total flow resistance. The gas p asses sequen tially th r ough the so da lime s crubb er, and the silica gel canister (plus interco n n ecting tu bing and the su it in terior). F or a p ac k ed b ed of sp herical particles, the p ressure drop p er unit length is giv en by the Ergu n equ ation [15]: ∆ P L = 150 µ (1 − ε ) 2 ε 3 d 2 p v s + 1 . 75 ρ (1 − ε ) ε 3 d p v 2 s (44) where µ is gas dyn amic viscosit y , ε is b ed vo id fr action, d p is particle diameter, v s is s u p erfi cial v elo cit y , L is b ed length, and ρ is gas d en sit y . 6.2.1 Time-V arying V oid F raction from Solid and Liquid V olume Expansion The Er gun equation is con ven tionally app lied with a constan t ε , but th e so d a lime scru bb er b ed undergo es a vo lum e c hange as the r eactio n pr o ceeds. The reactan t Ca(OH) 2 has a molar vo lume of ¯ V Ca(OH) 2 ≈ 33 . 0 cm 3 mol − 1 , wh ile the p ro duct C aCO 3 (calcit e) has ¯ V CaCO 3 ≈ 36 . 9 cm 3 mol − 1 . Crucially , the r eactio n also pr o duces 1 mol of liquid H 2 O p er mol of C O 2 (eq. (8)), with molar v olume ¯ V H 2 O(l) ≈ 18 . 0 cm 3 mol − 1 . The pap er explici tly requires t h is w ater to remain a s an aqueous surface film on the granules for scrubbing to function; it therefore o ccupies void space within the b ed. Not all reaction w ater remains in the b ed: a fraction ev ap orates in to the gas s tr eam and is carried d o wnstream to the desiccan t (this is the moisture load already accoun ted for in section 2.2). W e in tro duce a wate r reten tion fr action χ w ∈ [0 , 1], where χ w = 1 means all r eactio n w ater r emains in the b ed and χ w = 0 means it all ev ap orates. The effectiv e v olume displacing voids p er mole reacted is then: ¯ V products ( χ w ) = ¯ V CaCO 3 + χ w ¯ V H 2 O(l) = 36 . 9 + 18 . 0 χ w [cm 3 mol − 1 ] (45) Let ξ ( t ) ∈ [0 , 1] b e the fractional con v ersion of Ca(OH) 2 . Th e com bined s olid-plus-liquid volume in the canister ev olv es as: V solid+liquid ( t ) = V solid , 0 1 + ξ ( t )( σ ( χ w ) − 1) (46) where the effectiv e swe lling ratio is: σ ( χ w ) = ¯ V CaCO 3 + χ w ¯ V H 2 O(l) ¯ V Ca(OH) 2 = 36 . 9 + 18 . 0 χ w 33 . 0 (47) 21 A t χ w = 0 (all w ater ev ap orated): σ = 1 . 12, reco vering the solid-only analysis. A t χ w = 0 . 5 (half retained): σ ≈ 1 . 39. A t χ w = 1 (all w ater retained): σ ≈ 1 . 66. The v oid fr action in the fixed-v olume canister then decreases: ε ( t ) = 1 − (1 − ε 0 ) 1 + ξ ( t )( σ ( χ w ) − 1) (48) where ε 0 is the in itial vo id fraction. The follo wing table summarizes the impact: χ w = 0 χ w = 0 . 5 χ w = 1 σ 1 . 12 1 . 39 1 . 66 ε ( ξ = 1) for ε 0 = 0 . 40 0 . 33 0 . 17 0 . 00 Appro x. flo w resistance increase † 2 . 2 × 25 × → ∞ † Estimated fr om the Ergun equation’s dominan t viscous term, whic h scales as (1 − ε ) 2 /ε 3 . T he ratio at ε = 0 . 33 vs. ε 0 = 0 . 40 is (0 . 67 2 / 0 . 33 3 ) / (0 . 6 0 2 / 0 . 40 3 ) = 12 . 5 / 5 . 6 ≈ 2 . 2 × ; at ε ≈ 0 . 17 (rounded fr om 0.166) the r atio is (0 . 83 2 / 0 . 17 3 ) / (0 . 6 0 2 / 0 . 40 3 ) ≈ 25 × ; using the unround ed v alue ε = 0 . 166 gives ≈ 27 × . Both are rep orted as ∼ 25 × giv en the larger u ncertain t y in χ w . The χ w = 1 case (complete b ed blo c k age) is physically unr ealistic b ecause the b ed w ould c hoke and force gas through bypass c hann els lo ng before complete conv ersion. In practice, the gas flo w through the b ed activ ely ev ap orates surface w ater, so χ w is itself a function of flow rate , temp erature, and h um idit y: at high circulation r ates and lo w inlet humidit y , χ w is dr iven to w ard zero. The Ergun equation’s ε 3 denominator amp lifi es e ven mo d est voi d -fraction reductions into large flo w resistance increases, making this a critical coupling: the state-space mo del m ust trac k ξ ( t ), and the s en sitivit y to χ w should b e included in the MPC’s un certain t y propagatio n . F or the baseline d esign analysis, w e adopt χ w ≈ 0 . 3–0 . 5 (consisten t w ith the observ ation th at the gas flo w remov es a sub s tan tial fraction of reactio n w ater), yiel d in g σ ≈ 1 . 28–1 . 3 9 and ε ( ξ =1) ≈ 0 . 17–0 . 23. Th is represents a 8–25 × increase in flo w resistance o v er the scrubb er ’s lifetime—significan tly more sev ere than the solid-only estimate and a b inding constraint on scrubb er sizing and fan p ow er. Both the so da lime and silica gel pac ke d b eds con tribute to tota l resistance. 6.3 Closed-Loop Pressure and V olume Dynamics With the coun ter-lung, the system op erates in an appr o ximately isob aric regime rather than an iso c horic one. The total loop v olume is the sum of the rigi d comp onen ts (suit shell, canisters, tubing) and th e complian t coun ter-lung: V lo op ( t ) = V rigid + V CL ( t ) (49) The c ounter-lung v olume ev olv es according to eq. (42). The fans recirculate gas int ern ally through the treatmen t train; net mass c hanges (O 2 injection, CO 2 scrubbin g t o solid, H 2 O adsorption) are accommodated by coun ter-lung disp lacemen t r ather than pressu re v ariation. Small residual pr es- sure o scillations from tidal b reathing ( ∼ ± 1 m bar) are smoothed b y the counter-lung’s compliance. 6.3.1 V en tila tion Sanit y Check: Lo op Flush Rate vs. C O 2 Pro duction A basic rebreather d esign v erification e n sures that the fan circu lation rate is sufficien t to keep inspired CO 2 b elo w the 5000 pp m threshold. A t hea vy exertion, C O 2 pro du ction r eac hes ˙ n CO 2 ≈ 22 0 . 068 mol min − 1 . The scru bb er remo ve s CO 2 at a rate prop ortional to the inlet concen tration and con tact time. F or the lo op to m aintain steady-state p CO 2 ≤ 50 00 ppm = 0 . 005 atm, the minim um circulation fl ow rate m ust satisfy: Q circ , min ≥ ˙ n CO 2 · R g T p CO 2 , max − p CO 2 , out (50) where p CO 2 , out is the scrubb er outlet concen tration (e ffectiv ely z ero for fresh so d a lime). Ev aluating in litre-atmosphere units with R = 0 . 082 06 L atm / mol / K at T = 308 K (35 ◦ C) and P = 1 atm: Q circ , min ≥ ˙ n CO 2 · R T p CO 2 , max − p CO 2 , out = 0 . 068 × 0 . 08206 × 308 0 . 005 − 0 ≈ 344 L min − 1 (51) This is a high fl o w rate bu t consistent with the ∼ 200 L min − 1 to 40 0 L m in − 1 circulation rate s us ed in military and diving rebreathers u nder hea vy w orkloads. Accordingly , the lo op flo w sensor and fan subsys tem s hould b e sp ecified to at least 400 L min − 1 full scale. Th e fan system must deliv er this flow against the total lo op pressu re drop (scrubb er + d esiccan t + tub ing), whic h is the fan sizing constraint . Mo deling ca v eat: T his estimate assu mes a p erfectly mixed con trol volume (CSTR approxima- tion) and therefore repr esents a conserv ativ e upp er b ound on required circulation. In realit y , the scrubb er is a r eactiv e pac k ed b ed w here remo v al effic iency dep ends on residence t ime and the Damk¨ ohler num b er r ather than simp le dilution. Prop erly sized beds therefore require substantia lly lo w er circulation rates than the CST R b ound suggests. 23 7 In tegrated Mass Balance Summary Collecting the preceding results, the complete state of th e c losed-lo op system is describ ed by a coupled system of ordinary differenti al equations: dm O 2 , tank dt = − ˙ m O 2 , inject ( t ) (52) dn O 2 , suit dt = ˙ n O 2 , inject ( t ) − ˙ n O 2 , consumed ( t ) − x O 2 ( t ) ˙ n v ent ( t ) (53) dn CO 2 , suit dt = ˙ n CO 2 , pro duced ( t ) − r scrub ( t ) − x CO 2 ( t ) ˙ n v ent ( t ) (54) dn H 2 O , suit dt = ˙ n H 2 O , exhaled ( t ) + ˙ n H 2 O , rxn ( t ) − ˙ n H 2 O , ads ( t ) − x H 2 O ( t ) ˙ n v ent ( t ) (55) dn N 2 , suit dt = − x N 2 ( t ) ˙ n v ent ( t ) (56) dV CL dt = R g T suit P s ˙ n O 2 , inject − ˙ n O 2 , consumed − ˙ n v ent + V gas T suit dT suit dt + ˙ V breath ( t ) (57) dM wa ter dt = ˙ m air Y in ( t ) − Y out ( t ) (58) dm Ca(OH) 2 dt = − r scrub ( t ) M Ca(OH) 2 (59) dT bed dt = 1 ( ρc p ) bed V bed ˙ Q scrub ( t ) − ˙ m air c p, air ( T bed − T air , in ) − U A wa ll ( T bed − T wa ll ) (60) d UPTD dt = 1 60 P iO 2 ( t ) − 0 . 50 atm 0 . 50 atm 0 . 83 , if P iO 2 ( t ) > 0 . 50 atm , 0 , otherwise, (61) where t is measured in seconds, so the factor 1 / 60 con verts the standard minute-based UPTD accum ulation la w into SI time units. The gas-phase w ater balance (eq. (55)) couples the h umid it y state to the silica gel adsorption mo del: ˙ n H 2 O , exhaled is the metabolic w ater v ap or pro duction, ˙ n H 2 O , rxn = r scrub ( t ) (1:1 stoic hiometry , eq. (8)), ˙ n H 2 O , ads is the molar adsorp tion rate in the silica gel (eq. (22)), and the last term is ven t loss prop ortional to the w ater v ap or mole fraction. T he n itrogen balance (eq. (56)) is the simplest ODE—N 2 is neither pro d uced nor consumed, only lost thr ough v ent ing—b ut it is essential : the total gas-phase m olar inv en tory n total ( t ) = n O 2 + n CO 2 + n H 2 O + n N 2 app ears in the d en ominator of ev ery mole-fraction computation, and without trac kin g n N 2 it cannot b e ev aluated. The s orb ent d epletion (eq. (59)) tracks calcium h ydro xide mass directly , where r scrub is the molar scrubb in g rate (mo l C O 2 /s) and M Ca(OH) 2 = 74 . 09 g mol − 1 ; the 1:1 stoic hiometry of the net r eaction (eq. (8)) means eac h mole of CO 2 scrubb ed consumes one mole of Ca(OH) 2 . The cumulativ e o xygen to xicit y dose UPTD (eq. (61 )) accum u lates whenev er P iO 2 exceeds the pulmonary to xicit y threshold of 0 . 50 atm, follo wing the sta n dard unit p ulmonary to xici ty dose (UPTD) p o wer-la w accum ulation mod el used in diving and h yp erbaric medicine. This enables the MPC to trade transient high-O 2 exp osure against a cumulati ve dose bud get o v er th e mission, rather 24 than enforcing only an instanta neous threshold. Sub ject to the constraints: m O 2 , tank ( t ) ≥ 0 (62) m Ca(OH) 2 ( t ) ≥ 0 (63) M wa ter ( t ) ≤ M wa ter , max (64) 0 . 19 atm ≤ P iO 2 ( t ) ≤ 0 . 50 atm (65) x O 2 ( t ) ≤ 0 . 235 (66) x CO 2 , suit ( t ) ≤ 5000 ppm (67) RH suit ( t ) ≤ 60 % (68) V CL ( t ) ≥ V CL , min (69) UPTD( t ) ≤ UPTD max (70) where inspired O 2 partial p ressure is computed on a wet-gas b asis to reflect the p h ysiologically relev an t alve olar gas equation: P iO 2 ( t ) = P s ( t ) − P H 2 O T suit , bz , RH suit × x O 2 ( t ) (71) The magnitude of this correction dep end s on conditions at the breathing zone. A t the suit breathing-zone temp erature T suit , bz ≈ 35 ◦ C, the saturation v ap or pressure is P sat H 2 O ≈ 42 mmHg ; at the cont r ol target of RH = 60%, P H 2 O ≈ 25 mmHg ≈ 0 . 033 atm , reducing P iO 2 b y ∼ 3% relativ e to a dry -gas computation. F or comparison , the classical alv eolar gas equation u ses P H 2 O = 47 mmHg (b o dy co re temp erature, 37 ◦ C, full saturation in the alv eolar space)—the relev an t correctio n wh en estimating alv eolar P A O 2 from in spired P iO 2 , but not applicable to the inspir ed-gas calculation here. A t the humidit y constrain t b ound ary (RH = 60%), the inspired-gas correctio n is mo dest; it b ecomes clinically s ignifican t if the desiccant saturates and RH appr oac hes 100%, where P H 2 O → 42 mmHg and the correction a p proac hes ∼ 5.5 %. Th e cumulativ e to xicit y dose co n strain t (e q. (70)) limits total pulmonary exp osur e via the UPTD (un it pulmonary toxic ity dose) mod el, enabling the MPC to tr ad e brief excursions ab o v e 0 . 50 atm against a mission-duration d ose bud get r ather than relying solely on the in stan taneous thresh old. V CL , min is the minimum counter-lung v olume r equired to accommodate tidal breathing oscillatio ns (typica lly 1 L to 2 L). The oxyg en mole fract ion is explic- itly constrained by the normal-mo de fi re-safet y limit x O 2 ( t ) ≤ 0 . 235 (eq. (66)), w hile the higher to xicit y ceiling of 0 . 50 atm in wet-basis in s pired p artial pressure serves as a d egraded-mo de b ac kup constrain t rat h er than the primary op erating ta rget. The dominant d riv er of O 2 enric hment is ve nt comp ensation (eq. (37)): eac h v ent– r efi ll cycle replaces mixed gas w ith pure O 2 . The MPC must join tly manage the suit pressu re (minim izing u nnecessary v ent ing) and O 2 fraction tra jectory o v er its pr ediction horizon. 25 P art I I AI-Based Con trol System 8 Motiv ation: Why A I Con trol ? A conv en tional fixed-setp oin t PID (prop ortional-int egral-deriv ativ e) con troller (as commonly used in rebreather s y s tems) computes its output from thr ee terms— a prop ortional resp onse to the cur- ren t error, an integral term that eliminates steady-state offset, and a der iv ativ e term that damps o v ersh o ot—and regulates eac h v ariable in dep end en tly: O 2 partial pressu re, C O 2 concen tration, h um idit y , and f an sp eed. While adequate for steady-state diving, this app roac h is fund amen tally limited for fi refigh ting b ecause: (a) Unkno wn mission duration: The firefigh ter do es not kno w in adv ance ho w long they will b e deplo yed. A PID con troller cannot antic ipate resource depletion and w ill m ain tain nominal setp oin ts un til a consumable is exhausted, leading to abru pt system failure. (b) Coupled dynamics: O 2 injection, CO 2 scrubbin g, and humidit y are thermo d ynamically coupled (e.g., faster scrub bing generates more heat and w ater). F u rthermore, ve nt comp en- sation w ith pure O 2 creates a direct tradeoff b et w een suit pressure maint enance and o xygen enric hment (section 6.1.3) that PID lo ops op erating indep enden tly cannot r esolv e. (c) Rapidly c hanging conditions: Fireground conditions—temp eratur e, to xic gas exp osure, structural c ollapse risk—c hange on timescales of seconds to min utes. T h e con troller m ust incorp orate situational a w areness to mo d ify its strategy . (d) Firefigh ter state v ariabilit y: Exertion, stress, and environmen tal heat load pr o duce highly nonlinear and time-v arying metab olic demand that fi x ed -gain controlle rs cannot trac k effi- cien tly . W e adopt an AI con trol arc hitecture that addresses these chall enges throu gh sensor fusion, online state estimation, and mo del-predictiv e optimizat ion. 9 Sensor Suite The Galactic Bio ware Life Su pp ort System in tegrates three categories of s en sors: external envi- ronment al s ensors that c haracterize the fireground hazard, internal suit en vironment sensors that monitor the b reathing gas and micro climate t h e fir efi gh ter actually experiences, and fi refigh ter bio - metric s ensors th at trac k physiolog ical state. A k ey design constrain t is th at all biometric s ensors m ust b e compatible with h ea vy glo ve s, a fully encapsu lating s u it, and violen t physic al activit y— ruling out clinical-grade devices su c h as ingestible core temp erature pills, fi nger/ear p ulse o ximeters, c hest imp edance b elts, and transcutaneous C O 2 monitors, none of whic h survive the mec hanical and thermal stresses of structural firefigh ting. 26 9.1 External En vironmen tal Sensors These sensors face out ward through the suit sh ell or are mounted on the exterior h elmet surface, c haracterizing the am bient hazard environmen t. Sensor Measurement Range / Res o lution Radiant heat flux sensor External thermal threa t ˙ q rad 0–200 kW m − 2 External t her mistor (shielded) Am bient temp era ture T ext − 40 to 500 ◦ C, ± 2 ◦ C T oxic ga s sensor (MOS a rray) CO, HCN, NO x (external) Multi-gas, semi-quant itative Barometric pressure senso r Am bient pr essure P a 800–1 100 hP a, ± 0 . 5 hPa 9.2 Suit En vironmen t S ensors These s ensors are p ositioned within th e closed br eathing lo op and the su it in terior, pro viding d irect measuremen t of the gas the fir efigh ter is breathing and the micro climate surroun ding th eir b od y . Because they op erate inside the sealed, p ositiv e-pressu r e en v elop e, they are shielded from external com bustion pro ducts and extreme temp eratures. Sensor Measurement Range / Res o lution NDIR CO 2 sensor In-suit x CO 2 0–10 %, ± 0 . 01 % Galv anic O 2 ( × 3, median voting) In-suit x O 2 0–100 %, ± 0 . 1 % Capacitive RH senso r In-suit re lative hum idity 0–10 0 % RH, ± 1 . 5 % In-suit thermistor (breathing zone) Breathing gas temp erature T suit , bz 10 to 60 ◦ C, ± 0 . 5 ◦ C In-suit thermistor (torso) Suit in terior temperature T suit , torso 10 to 70 ◦ C, ± 0 . 5 ◦ C Thermo couple (K-t yp e) Scrubb er bed tempera ture T b ed − 40 to 100 0 ◦ C, ± 1 ◦ C Different ia l pressur e transducer Suit gauge pres s ure ∆ P suit 0–50 mbar, ± 0 . 1 mbar Flow sensor ( ho t-wire anemometer) Lo o p circulation rate Q circ 0–400 L min − 1 , ± 2 % Counter-lung po sition sensor Counter-lung volume V CL 0–10 L, ± 0 . 05 L The in -suit CO 2 and O 2 sensors are particularly critical: th ey provide a direct, real-time measur e of the br eathing-gas comp osition the fir efigh ter is actually inh aling. The th ree galv anic O 2 cells use median vot in g with plaus ib ilit y c hec ks (rejecting r eadings th at drift more than 2 % from the median), pro vidin g f ault tolerance against the sensor drift and failure mo des th at are a dominant acciden t dr iv er in r ebreather sys tems. Th e coun ter-lung p osition sensor pr o vides information ab out the system’s net m olar balance and suit in tegrit y . The d ual-location temp erature sensors (breathing zone and torso) allo w the con troller to distinguish b et w een a rise in breathing gas temp erature (indicating scrub b er exotherm or external heat soak) and a r ise in b o d y-pro ximate temp eratur e (indicating metab olic heat accum ulation), informing different con trol r esp onses. 9.3 Firefigh ter Biometric Sensors All b iometric sensors are designed for compatibilit y with structur al firefi gh ting: ruggedized, swea t- resistan t, tole rant of high-g impacts, and requiring n o exposed skin c ontac t on fingers, ears, or m ucous mem br anes. The c hest-strap ECG and torso-mounted IMU are integ rated in to the suit’s 27 inner ga rm en t la y er. A ruggedized w rist mod u le worn und er th e suit glov e pro vides redundant heart rate measuremen t via photopleth ysmography (PPG) and additional motion sensing. Sensor Measurement Range / Res o lution Chest-strap ECG (dry electro de) Heart rate (HR) 30–24 0 bpm, ± 1 bpm Chest-strap ECG Heart rate v aria bilit y (H R V) R–R interv als, 1 ms T ors o IMU (accele r ometer + gyro) Activit y level / postur e 3-axis, 100 Hz W rist PPG sensor Redundan t HR, p erfusio n index 30–24 0 bpm, ± 3 bpm W rist accelerometer W rist motion / activity 3-axis, 5 0 Hz Remark 5 (On the absence of SpO 2 and P tc CO 2 sensing) . Clinic al pulse oximetry and tr anscu- tane o us CO 2 monitoring r e quir e stable skin c ontact, c ontr ol le d temp er atur e , and minimal mo tion artifact—c onditions funda mental ly inc omp atible with structur al fir efighting. Without dir e ct blo o d gas me asur ement, the Galactic Biowar e Lif e Supp o rt System do es not attempt to estimate arterial blo o d gas values (which ar e p o orly identifiable fr om inspir e d gas c omp osition alone due t o c on- founders including V/Q mismatch, shunt fr action, and hemo globin variability). Inste ad, the system c omp utes risk indices : • Hyp o xia risk index , d riven by wet-b asis P iO 2 (e q. (71) ), estimate d m inu te ventilation (fr om HR and activity data ), and the metab olic mo del’s O 2 c onsumpt ion estimate. • Hyp er c apnia r isk index , driven by inspir e d x CO 2 , estimate d CO 2 pr o duction r ate, and the scrubb er’s mo dele d r emoval c ap acity. These risk indic es ar e sufficient for c ontr ol de cisions (the M PC p enalizes high risk, not sp e cific blo o d gas values) and avoid claiming identifiability that the sensor suite c annot supp ort. Remark 6 (On core temp erature estimat ion) . Cor e b o dy temp er atur e T c is a critic al ph ysiolo gic al variable f or he at str ess ma nagement, but dir e ct me asur ement via ingestible telemetry pil ls is im- pr actic al for r outine fir efighting deployment (pr e- ingestion timing, sing le- use c ost, gastr ointestinal c onc erns). The Galactic Biowar e Lif e Supp ort System inste ad estimates T c using a Kalman-filter– based thermal mo del that c ombines HR, HR V, in-suit torso temp e r at ur e, activity intensity fr om IMU, and the known external thermal envir o nment. This appr o ach, validat e d in military he at str ess r ese ar ch [16], pr ovides T c estimates within ± 0 . 3 ◦ C of pil l-me asur e d val ue s under mo der ate to he avy exertion. A c cur acy may de gr ade to ± 0 . 5 ◦ C – 0 . 7 ◦ C under e xtr eme he at—pr e cisely the c on- ditions enc ounter e d in structur al fir efighting—which the EKF’s u nc ertainty quantific ation c aptur es as incr e ase d state c ovarianc e. 28 10 State-Space F orm ulatio n 10.1 State V ect or W e define the s y s tem state vec tor x ( t ) ∈ R 18 : x ( t ) = n O 2 , suit n CO 2 , suit n H 2 O , suit n N 2 , suit x O 2 V CL m O 2 , tank m Ca(OH) 2 ξ M wa ter T bed T suit , bz T suit , torso HR ˆ T c ˆ ˙ V O 2 ˆ W UPTD (72) The first thirteen sta tes are physical /c h emical plant and suit envi r onmen t states from P art I. n H 2 O , suit is the gas-phase w ater in v ento r y in the breathing lo op (mol), g ov erned b y the bala nce of exhaled moisture, reaction-generated m oisture, silica gel adsorption, and v ent losses (eq. (55)). n N 2 , suit is the n itrogen inv en tory (mol), wh ic h starts at ∼ 79% of the initial gas fi ll and is con tinuously depleted by exh aust-v alv e v enti n g bu t nev er replenished (eq. (56 )); tr acking n N 2 is essen tial b ecause the total molar inv en tory n total ( t ) = n O 2 + n CO 2 + n H 2 O + n N 2 cannot b e computed without it, and n total app ears in the denominators of all mole- fr action computations. The O 2 mole fraction is constrained by the sp ecies inv en tories: x O 2 ( t ) = n O 2 , suit ( t ) n O 2 , suit ( t ) + n CO 2 , suit ( t ) + n H 2 O , suit ( t ) + n N 2 , suit ( t ) (73) In implement ation, x O 2 ma y still b e carried in the EKF as a redun dan t sensed state, b ut it is tied to the molar inv en tories by this algebraic consistency relation (or an equiv alen t pseu do-measuremen t). V CL is the coun ter-lung vol u me (eq. (42)), directly measured b y a p osition sensor; suit pressure P s is co mp uted algebraically from V CL via th e c ounter-lung compliance relation (eq. (41)) rather than trac k ed as an in dep end ent state, a v oiding a differen tial-algebraic inconsistency . m Ca(OH) 2 is the remaining mass of calcium hydro xide (the limiting reagen t in so da lime), decremente d by the 1:1 stoic hiometry of the net scrubb ing rea ction (eq. (59)). ξ is the scrubb er con ve rs ion fraction, which determines the time-v arying v oid fraction ε ( t ) via eq. (48) and hen ce the scrub b er flo w resistance; it is not directly measured b ut inferred by the EKF f rom the cu m ulativ e scru bbing inte gral and 29 the obs er ved fan-sp eed–to–flo w-rate relationship. States T suit , bz and T suit , torso are the b reathing- zone and torso-interio r temp eratures. The last fi v e are firefi gh ter p h ysiological and safet y states: heart rate (directly measured), estimate d core temp erature, estimated o xygen consumption rate, estimated metab olic w ork r ate, and cum ulativ e o xygen to xicit y dose UPTD (eq. (61)). 10.2 Con trol Input V ector The con trol input u ( t ) ∈ R 3 : u ( t ) = ˙ m O 2 , inject ω fan φ b ypass (74) where ˙ m O 2 , inject is the oxyge n inj ection mass flo w r ate (prop ortional v alv e op ening), ω fan is the f an rotational sp eed (co ntrolling circulation rate), and φ b ypass ∈ [0 , 1] is the f raction of flo w bypassing the scr u bb er (a con trollable damp er), wh ich allo ws trading scru bbing rate against p ressure dr op and thermal load. 10.2.1 Solenoid V alve Stiction and Low-Flo w Nonlinearity The prop ortional solenoid v alv e that meters O 2 injection exhibits a nonlinear dead-zone a t lo w command signals due to stiction (static f riction) of the v alv e armature against its s eat. Belo w a threshold vol tage V break , the v alv e d o es not mo v e; once the break a w a y force is exceeded, the v alve jumps to a minim um op en p osition ˙ m min , creating a discon tin uity in th e con trol-to -flow mapping: ˙ m O 2 , actual ( V ) = ( 0 if V < V break ˙ m min + k v ( V − V break ) if V ≥ V break (75) where V is the command v oltage and k v is the v alv e gain in the linear r egime. The jump fr om 0 to ˙ m min (t ypically 5 % to 15 % of full-scale flo w) is non -d ifferen tiable, which creates t w o p roblems for the con troller: 1. The MPC’s gradien t-based sol ver (SQP) cannot compute a v alid descen t direction at the stiction b oundary , leading to oscillatio n b etw een “v alv e closed” and “v a lve at minim um op en.” 2. A t very lo w metab olic demand (rest p erio ds), the desired injection rate ma y fall b elo w ˙ m min regardless of the r esource cost f orm ulation. The controll er “h unts” for the un reac hable set- p oint , causing high-fr equency chatte r ag ainst the v a lve seat—accelerating exactly the me- c hanical fatigue that the RL cycling p enalt y wa s designed to p r ev ent . Pulse-width mo dulation (PWM) strategy for sub-minimum flo ws: The standard solutio n in pr ecision flu id control is to replace con tinuous lo w-vol tage commands with a pulse-width mo d- ulate d (PWM) signal: the v al ve is p erio dically op ened to a flo w rate ˙ m pulse > ˙ m min (safely ab o ve the stic tion b oun dary) for a fraction δ ( t ) ∈ [0 , 1] of eac h PWM p erio d T PWM , ac hieving the desired time-a v eraged flow: h ˙ m O 2 , inject i = δ ( t ) · ˙ m pulse (76) The MPC optimizes the d uty cycle δ ( t ) rather than the raw volt age when the desired flow falls b elo w ˙ m min . This k eeps the v alv e op erating in its linear regime (ab o v e V break ) during eac h p ulse, eliminating the stiction n onlinearit y from the optimiza tion landscap e. The PWM p erio d T PWM ≈ 2 s to 5 s is c hosen to b e: 30 • Long enough that eac h op en–close cycle k eeps the v alv e in steady fl o w (a vo id in g wat er-hammer transien ts), • Short enough that the p er-pu lse O 2 injection is a small fraction of the lo op in ven tory , b ounding the mole-fraction fluctuation (see b elo w), • Belo w the 2 Hz rev ersal threshold in the RL cycling p enalt y , so P WM op eration is n ot p enal- ized as “c hatter.” Bounding the PWM-induced O 2 fluctuation: Each pu lse injects a b olus of ∆ n pulse = ˙ m pulse · δ · T PWM / M O 2 moles of pure O 2 in to a lo op con taining n total ≈ 4 mol ( ∼ 100 L at 1 atm, 308 K). A t w orst case—full-scale pulse flo w ˙ m pulse = 1 g s − 1 , dut y cycle δ = 1, and T PWM = 5 s—the injected b olus is: ∆ n pulse = 1 . 0 × 5 32 . 0 ≈ 0 . 156 mol ( 77) The resulting p eak-to-trough mole-fraction excursion, assuming instantaneo u s injectio n in to a we ll- mixed vo lu m e (worst case; in practice the fan circulation pr ovides con tinuous mixing during the pulse), is: ∆ x O 2 ≈ ∆ n pulse n total + ∆ n pulse (1 − x O 2 ) = 0 . 156 4 . 156 (1 − 0 . 21) ≈ 0 . 030 = 3 . 0 % (78) This is the unmixe d b olus up p er b ound. In practice, the fan circulation rate ( ∼ 300 L min − 1 ≫ loop v olume / T PWM ) turns o v er the loop v olume m ultiple times durin g eac h 5 s pulse, distributing th e injected O 2 throughout t h e l o op as it enters. The relev an t mixing timescale is τ mix = V lo op /Q circ ≈ 100 / 3 00 ≈ 0 . 3 3 min ≈ 20 s, and the pu lse du ration is a f raction of this, so the in s tan taneous lo cal fluctuation at the breathing zone is atten uated by appro ximately T PWM · δ / (2 τ mix ). F or a t yp ical lo w-demand dut y cycle δ ≈ 0 . 3 and T PWM = 3 s: ∆ x O 2 , bz ≈ ˙ m pulse · δ · T PWM M O 2 · n total (1 − x O 2 ) = 1 . 0 × 0 . 3 × 3 32 . 0 × 4 (0 . 79) ≈ 0 . 006 = 0 . 6 % (79) The < 0 . 5 % target is th er efore achiev able for du t y cycles δ . 0 . 25 at T PWM = 3 s, whic h corresp onds to th e lo w-demand reg ime w here PWM is actually u sed (higher d emands u se con tinuous fl ow ab o v e the stiction thresh old). F or the sep arated-loop arc hitecture ( V lo op ≈ 10 L, n total ≈ 0 . 4 mol ), the fluctuation is p r op ortionally larger and T PWM m ust b e reduced accordingly . The tran s ition b et ween con tin uou s con trol (ab o ve ˙ m min ) and PWM c ontrol (b elo w ˙ m min ) is manage d b y a h ysteresis band to a vo id mo d e-switching oscillatio n at the b ound ary . 10.3 Disturbance V ect or The un controlle d disturban ces d ( t ) in clude: d ( t ) = W ( t ) T ext ( t ) ˙ q rad ( t ) c to xic ( t ) P a ( t ) T (80) represent ing metab olic work rate, external temp erature, radian t heat flux, external to xic gas con- cen trations, and am bient barometric p ressure. 10.4 Nonlinear State Dynamics The system d y n amics are: ˙ x ( t ) = f x ( t ) , u ( t ) , d ( t ) (81) 31 where f encapsulates the coupled ODEs from eqs. (52 ) to (60) plus suit en vironment dyn amics and physio logical mo dels: Suit breathing-zone temp erature dynamics: C bz dT suit , bz dt = ˙ m air c p, air ( T bed , out − T suit , bz ) − h bz A bz ( T suit , bz − T suit , torso ) (82) where C bz is the thermal capacit ance of the breathing zone gas v olume and T bed , out is the scrubb er outlet air temp erature—capturing the direct effec t of scrubb er exotherm on inspired gas temp era- ture. Suit torso-in terior temp erature dynamics: C torso dT suit , torso dt = ˙ Q met , skin ( ˆ W ) + U shell A shell ( T ext − T suit , torso ) + ˙ q rad τ shell A shell − ˙ Q con v , suit (83) where ˙ Q met , skin is metab olic heat reac hing the skin surface, U shell is the suit sh ell’s o v erall thermal transmittance, τ shell is the shell’s r ad iant trans missivit y , and ˙ Q con v , suit is con vec tive co oling f rom the circu lating gas flo w . The rise rate of T suit , torso is a k ey obs erv able for the control ler: a rapid increase indicates either high external heat soak or risin g metab olic h eat load. Heart ra t e dynamics (first-order lag mo del): τ HR d HR dt = HR ss ( ˆ W , ˆ T c , T ext ) − HR( t ) (84) Estimated core temp erature dynamics (Kalman-filter–augment ed Stolwijk mo d el [17]): C c d ˆ T c dt = ˙ Q met ( ˆ W ) − ˙ Q resp − ˙ Q skin ( ˆ T c , T suit , torso , ˙ q rad ) + K tc T meas suit , torso − h ( ˆ T c ) (85) where t h e last term is a Kalman co r r ection: h ( ˆ T c ) is the pr edicted torso-in terior temp erature giv en the est imated core te mp erature, and K tc is the Kalman ga in. This fuses the ph ysics-based thermal mo del with th e measured in-suit torso temp erature to con tin u ou s ly u p date th e core temp eratur e estimate without requiring an ingestible p ill. Metab olic estimator with thermal and hypoxic decoupling : Heart rate elev ation in a firefighte r has three distinct ph ysiological causes: (i) muscular work (t h e metabolic signal the con troller n eeds), (ii) cardio v ascular heat strain from high humidit y and temp erature (wh ic h im- pairs ev ap orativ e co oling and triggers comp ensatory tac hycardia), and (iii) hypoxic comp ensation (c hemoreceptor-driv en HR increase when P iO 2 drops b elo w ∼ 0 . 18 atm). If the metab olic esti- mator treats all HR elev a tion as muscular w ork, t wo dangerous p ositiv e feedback lo op s emerge: Humidity–scrubbing lo op: High suit humidit y → impaired swe at ev ap oration → elev at ed HR → mo del o ver-e stimates ˆ W → cont roller increases O 2 injection and fan s p eed → more CO 2 scrubb ed p er unit time → more scru bb er heat and r eaction-g enerated moisture → higher h umidit y . This lo op is particula r ly dangerous wh en h eat reduces th e silica ge l’s adsorption capacit y ( q m in eq. ( 21 ) drops appro ximately 40% b et w een 25 ◦ C and 50 ◦ C). Hyp oxia– c onservation lo op : λ ( t ) force s O 2 fraction d o wn → mild h yp o xia elev ates HR via p eriph eral c hemoreceptor activ ation [18] → mod el o v er-estimates ˆ W → con troller in creases O 2 to “supp ort exertion” → confl icts with scarcit y mul- tiplier → v alve oscillation b etw een conserv at ion and sup p ort. T o break b oth lo ops, the metab olic 32 estimator receiv es the in-suit h umid it y and O 2 fraction as explicit in puts, enabling it to learn the non-w ork components of HR: ˆ W ( t ) = g HR( t ) , HR V ( t ) , acc torso ( t ) , acc wrist ( t ) , T suit , torso ( t ) , x CO 2 , suit ( t ) , RH suit ( t ) , x O 2 ( t ) | {z } decoupling inputs (86) The net work arc hitecture decomp oses th e HR signal inte r n ally: HR( t ) = HR wo r k ( ˆ W ) | {z } musc ular + HR heat (RH , T suit , torso , ˆ T c ) | {z } heat strain + HR h yp ox ( x O 2 ) | {z } h yp oxic (87) Only HR wo r k driv es th e metabolic w ork estimate ˆ W . The heat-strai n and h yp o xic comp onen ts are estimated by the netw ork’s inte rn al represent ation and su btracted b efore th e w ork-rate outpu t la yer. During pr e-training, the netw ork is exp osed t o lab oratory proto cols that in d ep endently manipulate h um idit y (climate c hamber), O 2 fraction (altitude simulat ion), and work rate (treadmill), pr o viding the s up ervised signal to disenta ngle th ese three HR d riv ers. The MPC cost fu nction includes an additional thermal de c oupling p enalty that p rev ent s the optimizer f rom resp onding to h umidit y- induced HR ele v ation with increased scru b bing: ℓ decouple ( x , u ) = w 9 · max (0 , RH suit − R H thresh ) · ∆ ω + fan (88) where ∆ ω + fan = max(0 , ω fan ,i − ω fan ,i − 1 ) is the p ositive f an-sp eed in cremen t. Th is term p enalizes fan-sp eed incr e a ses sp ecifically when humidit y is already elev ated, breaking the p ositiv e f eedbac k lo op by preven ting the co ntrolle r fr om “c hasing” a humidit y-driv en HR signal with more sc r ubbing activit y . 11 Con trol Ob jectiv e: Constrained Optimization Under Uncer- tain t y The fun damen tal c hallenge is to maximize the fir efigh ter’s op erational effectiv eness o v er a mission of unknown duration T , su b ject to hard sa fety constraint s. W e formulate this as a r eceding-horizon optimal con trol problem. 11.1 Cost F unction A t eac h con trol step k , with sampling p eriod ∆ t , the control ler solv es: min u k : k + N J = k + N X i = k w 1 ℓ safet y ( x i ) + w 2 ℓ comfort ( x i ) + w 3 ℓ resource ( x i , u i ) + w 4 k ∆ u i k 2 + ℓ decouple ( x i , u i ) (89) where N is the pr ediction horizon, ∆ u i = u i − u i − 1 , and: Safety p enalty (barrier fun ction): ℓ safet y ( x ) = X j ∈S max 0 , x j − x max j x max j − x nom j ! 2 + m ax 0 , x min j − x j x nom j − x min j ! 2 (90) 33 enforcing soft constrain ts on P iO 2 , x O 2 , x CO 2 , RH, V CL , ˆ T c , T suit , torso , HR, and scrubb er temp erature with quadr atic p enalt y as s tates approac h limits. Comfort cost: Penaliz es deviations from ideal breathing conditions (e.g ., RH of 40 %, T suit of 28 ◦ C). Resource conserv ation: ℓ resource ( x , u ) = λ ( t ) · ˙ n v ent ( x ) (91) where ˙ n v ent ( x ) is th e e xh aust-v alv e v ent rate (eq. (35)), whic h is a function of the suit pressur e state (and hence of V CL via eq. (41)). T he p enalt y targets venting rather than injection b ecause the metab olic O 2 demand is fi xed and u na vo idable—p enalizing inj ection merely forces the optimizer to dela y it, driving the count er-lun g to V CL , min and creating pathological b an g-bang v alv e c hatter. The actual con trollable w aste is v en ted gas, whic h ca rr ies O 2 (at the curren t enric hed fr action) irrev ersibly out of the suit. By p enalizing the ven t rate, the MPC learns to maint ain su it pressur e just b elo w the crac king threshold, minimizing v en t losses while preserving t h e p ositiv e-pressure in tegrit y margin. λ ( t ) is a dyna mic r esour c e sc ar city multiplier th at in cr eases as consu mables deplete: λ ( t ) = λ 0 m O 2 , tank (0) m O 2 , tank ( t ) α (92) with α > 1 creating in creasingly aggressiv e conserv ation as th e tank empties. Th is is the k ey mec hanism by which the con troller han d les unkno wn mission duration: as resources diminish, the optimizer automatica lly shifts to a m ore conserv ativ e pressure-managemen t regime, tolerating sligh tly lo w er suit gauge pr essu re to reduce ven t fr equency . 11.2 Hard Constrain ts The optimization is sub ject to: x i +1 = A d,i x i + B d,i u i + g d,i ∀ i ∈ [ k , k + N ] (93) u min ≤ u i ≤ u max ∀ i (94) m O 2 , tank ,i ≥ 0 ∀ i (95) m Ca(OH) 2 ,i ≥ 0 ∀ i (96) V CL ,i ≥ V CL , min ∀ i (breathing margin) (97) x O 2 ,i ≤ x O 2 , max ( m i ) ∀ i (98) P iO 2 ,i ≥ 0 . 16 atm ∀ i (h ard h yp o xia limit, wet basis) (99) UPTD i ≤ UPT D max ∀ i (cum u lative to xicit y dose) (100) where the mo d e-dep endent o xygen upp er b ound is x O 2 , max ( m i ) = ( 0 . 235 , m i = n ormal mo de , 0 . 50 , m i = d egraded mo de . Under n orm al op eration, the b inding upp er o xygen co nstr ain t is x O 2 ,i ≤ 0 . 235 . In a declared degraded mo d e, the u pp er b ound ma y b e temp orarily relaxed to x O 2 ,i ≤ 0 . 50, bu t this must b e treated as an emergency fallbac k with immediate e v acuation ala rm rather than a n orm al op erating 34 regime. The enrichmen t is dr iven by exhaust-v alv e v en ting (eq. (37)): eac h v ent ev ent replaces mixed gas with pu re O 2 . The MPC must therefore plan pressure control to minimize un necessary v ent ing while mainta inin g the p ositiv e-pressure margin against to xic infiltration. Discretization of the L TV prediction mo del: The dyn amic constraint (93) is wr itten in terms of the L T V matrices ( A d,i , B d,i , g d,i ) obtained by exact zero-order-hold discretization of th e Jacobian linearizatio n at eac h predicted state: A d,i = e A i ∆ t , B d,i = R ∆ t 0 e A i τ dτ B i , and g d,i is the affine residual from the linearization p oint. In implemen tation, B d,i is ev a luated via an augmen ted matrix exp onenti al, wh ic h remains v alid ev en when A i is singular. Because the coup led system exhibits stiffness—the scrubbin g kinetics and gas-mixing time constants ( τ mix ≈ 20 s) are fast relativ e to the thermal and so rb en t-depletion dynamics ( τ ∼ 10 2 –10 3 s)—explicit forw ard Euler discretization of th e full n onlinear mo del wo uld risk n u merical instabilit y at ∆ t = 1 s. The m atrix-exp onen tial discretization is unconditionally sta b le f or an y ∆ t and preserv es the eigen v alue structure of t h e con tin uou s -time linearization, making it the app ropriate c hoice for the emb ed ded QP form ulation. The EKF’s o wn prediction step uses f ou r th-order Runge–Kutta (RK4) on t h e full nonlinear mo del f , which is not sub ject to the same compu tational constrain t as the MPC horizon rollout. 12 AI Arc hitecture Figure 2 : AI con trol arc hitecture with explicit safet y g ating. E xternal, in-suit, and b iometric sensors fee d the EKF-based sensor-fusion and state-esti mation la yer. The learned metabolic mod el pro vides physiolog ical e stimates to the MPC , while the RL p olicy advisor supplies a w arm-start p olicy h in t during n omin al op eration and a fallback candidate ac tion if the MPC fails or ti mes out. All candidate actuat or commands p ass through the co ntrol-barrier-function safet y filter b efore reac hing th e actuators and, through them, th e ph ysical life-supp ort plan t and firefighte r . The arc hitecture (fig. 2) compr ises fiv e k ey m o dules, with the safet y filter ac ting as the final sup erv isory lay er b et w een the decision logic and the actuators: 35 12.1 Sensor F usion and State Est imation An extende d Kalman filter (EKF) [1 9] estimate s the fu ll state vecto r ˆ x ( t ) from noisy , async hronous measuremen ts across all three sensor tiers. The EKF process mo del uses the nonlinear dynamics f from eq. (81), while the measuremen t mod el maps states to sensor observ ati ons with kno wn noise co v ariances. Cr itically , states th at are not directly m easur ed— ˆ T c , ˆ W , and ˆ ˙ V O 2 —are estimated as laten t v ariables, w ith their uncertain ty explicitly trac ked in the state error co v ariance ˆ P ( t ). Th e MPC uses this uncertaint y quan tification for robust constraint satisfaction: when the EKF is less certain ab out core temp erature (e.g., d uring rapid transien ts), the control ler applies wider s afet y margins. 12.2 Learned Metab olic Mo del A compact neur al net wo r k g θ , parameterized by θ , maps observ able biometric and su it-en vironment signals to metabolic wo rk rate and O 2 consumption, with explicit thermal and hyp oxic de c oup ling inputs : [ ˆ W ( t ) , ˆ ˙ V O 2 ( t )] = g θ HR , HR V , acc torso , acc wrist , T suit , torso , x CO 2 , suit , RH suit , x O 2 (101) The net w ork is pre-trained on lab oratory data collected f rom firefigh ters p erforming standardized exercises (stair clim b, hose adv ance, f orced entry , search a n d rescue) while instrumen ted with a metab olic cart, includ ing proto cols th at indep enden tly v ary h u m idit y (c limate c ham b er) and inspired O 2 fraction (altitude sim u lation) to provide sup ervised signal for the HR d ecomp osition (eq. (87)). Online adaptation w ith cata strophic-forgetting protection: Th e mo del is fi ne-tuned on- line using the EKF’s residu als as a s elf-su p ervised signal, but na ¨ ıv e online grad ient d escen t risks c atas tr ophic for ge tting : the extreme, non-stat ionary signal distr ibution on the fi r eground (rapid HR spik es, thermal transients, nov el exertion patterns) can o verwrite the baseline ph ysiologica l calibra- tions learned dur ing pr e-training. If this o ccurs, the HR d ecomp osition (eq. (87)) d egrades—the net wo rk ma y p ermanently misattribute thermal strain as m uscular work, ev en after the EK F decou- pling inpu ts atte mp t correctio n. T o pr ev ent t h is, online u p dates use Elastic Weight Consolidation (EW C). Let θ ∗ denote the pre-trained weigh ts and F the diago nal of the Fisher information ma trix computed on the pr e-training d ataset, which measures h o w sensitiv e the loss is to eac h w eigh t. The online learning ob jectiv e augments the self-sup ervised loss L EKF with a qu adratic anc h or: L total ( θ ) = L EKF ( θ ) + λ EW C 2 X i F i ( θ i − θ ∗ i ) 2 (102) where λ EW C con trols the consolidation strength. W eigh ts with h igh Fisher information ( those critical to the pre-trained HR → work mapp ing) are strongly a n chored to θ ∗ , while w eigh ts with lo w Fisher inform ation (those enco d ing con text-sp ecific adaptations like individual cardio v ascular fitness) are f ree to u p date. This p reserv es th e fund amen tal physiolog ical la ws learned in the lab while allo wing the mo del to adapt to the individual firefi ghter’s ph ysiology and to slo w sensor drift. The online lea r n ing rate is additionally clamp ed to η online ≤ 0 . 1 × η pretrain , and u p d ates are susp ended entirely when the EKF’s state un certain t y ˆ P ( t ) exceeds a thr eshold (in d icating that the residual signal is unr eliable du e to rapid transien ts). The inclusion of RH suit and x O 2 as inputs is critical: w ith ou t them, humidit y-driv en tac hycardia and h yp o xic comp ensation are misattributed 36 as muscular exertion, triggering th e p ositiv e feedb ac k lo op s describ ed in the state d ynamics section. Similarly , in-suit CO 2 concen tration serv es as a direct metab olic proxy (residu al rise ab ov e scrubb er prediction indicates increased CO 2 pro du ction), a n d the wrist acceleromet er provides indep enden t confirmation of u pp er-b o dy exe rtion that the torso IMU alone may underestimate. 12.3 Mo del-Predictiv e Con troller (MP C) A t eac h con trol in terv al ∆ t (nominally 1 s), the MPC : 1. Receiv es ˆ x k and ˆ P k from the EKF. 2. F orecasts distur b ances ˆ d k : k + N using c u r ren t sensor trends and a short-horizon extrap olati on. 3. Solv es the constrained optimiza tion (eq. (89)–eq. (99)) o v er horizon N (t ypically 15–20 steps with m o v e blo c king, i.e. , 15–20 sec ond s ahead) using a linear time-v arying (L TV) a p p ro xima- tion of the plan t d ynamics [20]. 4. P asses only th e first control action u ∗ k through the con trol-barrier-function safet y filter, app lies the resu lting safe action to the actuators, and re-solv es at the next step (receding horizon). The prediction h orizon of 15–20 seconds is short relativ e to the thermal and sorb ent- dep letion time constan ts ( τ ∼ 10 2 –10 3 s). The d ynamic scarcit y multiplier λ ( t ) in eq. (92) comp ensates for this by enco ding long-horizon resource a w areness into the in s tan taneous cost structur e, effectiv ely extending the con troller’s plannin g capabilit y we ll b ey ond its optimizat ion horizon. λ ( t ) is the critical in no v ation: as co n s umables d eplete, the c ost of ve nting increases automatica lly , causing the o p timizer to fi nd operating p oin ts that minimize u nnecessary g as loss—tigh tening the pressure margin abov e crac king pressure (reducing v ent frequency and O 2 w aste), reducing fan sp eed wh ere p ossible (sa ving O 2 b y reducing respiratory demand from in creased e ffort of br eathing against loop resistance), and increasing scrubb er b ypass to extend sorb ent life. 12.4 Reinforcemen t Learning Policy Advisor The MPC optimization is a n onlinear pr ogram that m a y con ve rge slo wly or to lo cal min ima. T o w arm-start the optimizer and p ro vide a fallbac k candidate actio n for t r u ly no v el situations, a reinforcemen t learning (RL) age nt [21] runs in parallel. The RL age nt is trained offlin e in a high- fidelit y sim ulator of the Ga lactic Bio w are Life Supp ort S ystem across thousands of randomized fire scenarios with v arying: • Mission dur ations (30 min to 4 h ours) • Exertion profiles (low, mo derate, h igh, in termittent burs t) • Am bien t temp erature tra jectories • Consumable initial state s (sim ulating partial depletion fr om prior u se) The RL p olicy π φ ( x ) provides a “p olicy hint ” that can b e u sed to wa rm -start the MPC during nominal op eration. The MPC’s nominal command is then passed through the same safety filter b efore reac hing the actuators. If the MPC op timization fails or times out (exceeding the 100 ms real-time deadline), the RL p olicy instead s upplies a fallbac k candidate ac tion, whic h is passed through that same safet y fi lter b efore app lication. Con trol barrier function safet y filter: Because th e RL p olicy is trained offline and the MPC relies on lo cal linearizations of a n on lin ear p lan t, neither cand idate command sh ould b e sent directly to the hardware without a fin al safet y c hec k. A c ont r ol b arrier function (CBF) filter [22] is therefore 37 placed a s the last la y er b efore the actuators, ensu r ing that all hard sa fety constraints are resp ected regardless of w h ether the cand idate action comes from the MPC or from the RL fallback path: u cand = u ∗ MPC if the MPC returns a v alid solution π φ ( x ) if the MPC fails or times out (103) u safe = arg min u k u − u cand k 2 s.t. ˙ h j ( x , u ) + κ j h j ( x ) ≥ 0 ∀ j ∈ C (104) where h j ( x ) are barrier f unctions for eac h hard constraint (e.g., h 1 = x O 2 , fire − x O 2 , h 2 = P iO 2 − P iO 2 , min ), κ j > 0 are class- K function co efficien ts, and C is the set of safet y constrain ts. T h e CBF fi lter is a small q u adratic pr ogram that can b e solv ed in < 1 m s , ensuring that the system alw a ys r emains within the safe in v arian t set ev en when th e RL p olicy pr o duces an aggressiv e or out-of-distribution suggestion. The conti nuous-time CBF condition ˙ h j + κ j h j ≥ 0 is applied at t h e QP’s sub-millisecond solv e cadence; for the em b edded implemen tation at ∆ t = 1 s, the equiv alen t discrete-time cond ition h j ( f ( x , u )) ≥ (1 − κ j ) h j ( x k ) is u sed to account for the sampling in terv al. This arc hitecture e n sures that the RL p olicy is neve r the d ir ect actuator authorit y: in n ominal op eration it provides only a warm-start hint to the MPC, an d in f allbac k op eration it p ro vides a substitute candidate action when the MPC fails or times out. In b oth cases, no command reac hes the actuators without first passing through the CBF safet y la y er. The safety filter therefore acts as the final inv a r iant-set guard, providing formal guaran tees that constrain t-violating actions cannot reac h the plant . The rewa r d function for RL training mirrors the MPC co st b ut m ust additionally p enalize actuato r cycling, which the MPC’s k ∆ u k 2 term handles implicitly but the RL p olicy does not inh erit: r t = − ℓ safet y ( x t ) − w 2 ℓ comfort ( x t )+ w 5 1 [still op erational at t ] − w 6 ℓ resource ( x t , u t ) − w 7 ℓ cycle ( u t , u t − 1 ) (105) where 1 [ · ] is an indicator fun ction rew ardin g surviv al time and the cycling p enalt y is: ℓ cycle ( u t , u t − 1 ) = k ∆ u t k 2 + w 8 t X k = t − K 1 sign(∆ u 1 ,k ) 6 = sign(∆ u 1 ,k − 1 ) (106) The first term p enalizes large con trol c hanges (matc hing the MPC’s smoothness cost). The second term explicitly c ounts dir e ction r eversals of the O 2 prop ortional v alv e ( u 1 = ˙ m O 2 , inject ) o v er a trailing windo w of K steps. This is critical b ecause, when the MPC times out, th e RL p olicy supplies the fallbac k candidate command present ed to the safet y filter; w ithout th is p enalt y , the p olicy can still lea rn high-frequency v alv e oscilla tions that impro v e gas concen tration trac king but cause pr emature m echanical f ailur e. Pr op ortional s olenoid v alv es are p articularly vulnerable to rapid cycling in high-vibration environmen ts: the com bination of external m ec hanical s ho c k (structural firefighti n g in vol ves impacts, falls, a n d to ol us e) and internally ind uced v alv e c hatter accele r ates seat wea r and can lead to stuck-o p en or stuck-cl osed failure mo des. The w eigh t w 8 is calibrated so th at v alv e r ev ersal r ates ab o ve 2 Hz are strongly p enalized du ring training. 13 Situational Aw areness In tegratio n A uniqu e asp ect of the Galactic Bio ware con troller is its incorp oration of external en vironmenta l in telligence in to resource managemen t d ecisions. 38 13.1 Thermal Threat Assessmen t The external radian t heat flux sensor and e xternal ther m istor, com bined with the in-suit temp era- ture sensors, f eed a thermal thr e at estimator : Θ( t ) = ˙ q rad ( t ) ˙ q rad , max + T ext ( t ) T ext , max + γ soak dT suit , torso dt dT suit , torso dt max (107) where Θ ∈ [0 , 3] is a norm alized comp osite th reat index. T he th ird term c aptur es the r ate of in -su it temp erature r ise, wh ic h d etects heat soak p enetrating the suit shell even b efore external sensors register a change (e.g., wh en the firefighter is su rround ed by h eated surfaces radiating from multiple directions). When Θ is high: • The MPC antic ipates increased metab olic demand (cardio v ascular resp onse to h eat) a nd pre- emptiv ely adjusts O 2 injection. • The fan sp eed is increased to enhance con v ectiv e co oling within the su it. • The resource scarcit y m ultiplier λ is t emp orarily reduced to prioritize su r viv al o ver c onserv a- tion. Con versely , when Θ drops (e.g., the fir efigh ter has retreated to a cooler zone), the con troller shifts aggressiv ely tow ard resource conserv ation, antici p ating that th e firefigh ter may need to re-en ter the hazard zone. 13.2 Activit y Classification from D ual I MU The torso-moun ted and wr ist-moun ted accele rometer and gyrosco p e data are join tly pro cessed b y a light w eigh t con vo lutional classifier to determine the firefighter’s curren t activit y (stationary , w alking, clim bing stairs, cra wling, forcible entry , carrying victim, hose handling). Th e d ual-IMU configuration impro v es classification accuracy: th e torso IMU captures whole-b o dy locomotion patterns, while the w rist acceleromete r disambiguat es upp er-b o dy tasks (e.g., distinguishing forcible en try f r om stair climbing, whic h h av e similar torso a cceleration profiles but v ery differen t arm kinematics). Eac h activit y class has a c haracteristic metab olic profile, enabling the metab olic mo del to p ro duce more accurate ˆ W estimates with lo we r latency than heart-rate– only estimation. 13.3 In-Suit Atmosphere Monitoring a nd A nomaly Detection The suit environmen t sensors pro vide a conti nuous, high-frequency picture o f the gas the fir efi gh ter is actually breathing. Th e cont r oller uses th is data stream for t wo p urp oses b eyond basic regulation: Scrubb er health monitoring: The con troller con tin u ously compares th e measured in-suit x CO 2 against the v alue predicted by the scrub b er kinetics mo d el (eq. (15)). A p ersistent p ositiv e residual (measured > pr edicted) indicates scrub b er degradation—either so d a lime exhaustion, c h anneling in the pack ed b ed, or pro duct-la y er build up reducing the effectiv eness factor η ( t ). Th e magnitude of this r esidual is u sed to up date η ( t ) online, impro ving the MPC’s forward predictions of remaining scrubb er life. Seal integrit y detection: A sustained dr op in ∆ P suit accompanied by unexp ected c hanges in in-su it gas comp osition (e.g., CO detection by the external to xic gas arra y coinciding with a drop in x O 2 ) triggers a suit breac h alarm. Th e con troller can distinguish a slo w leak (gradual ∆ P decline) from a catastrophic br eac h (rapid pr essu re equalizat ion) and resp ond accordingly: a slo w leak triggers an increase in O 2 injection to comp ens ate, wh ile a catastrophic br eac h triggers an immediate ev acuation alarm. 39 13.4 Hazard Pro ximity Estimation The to xic gas sensor arra y (CO, HCN, NO x ) pr o vides an indirect measure of pro ximit y to the fire seat. Increasing concen trations signal appr oac h to the fir e, triggering: • V erification of suit seal integrit y (monitoring ∆ P suit ). • Alert to the firefigh ter if the seal margin drops b elo w thr eshold. • An ticipatory O 2 b o ost (pr e-loading for exp ected exertion increase). 14 Emergency Proto cols and Graceful Degradation The AI con troller implement s a hierarc hy of op erating mo des: Normal mo de: F ull MPC optimiza- tion with c omfort and resource balancing. Conserv ation mode: Activ ated when any consumable drops b elo w 25 % remaining. The resource scarcit y m ultiplier increases sh arply , setpoints shift to w ard minim um safe v alues, and th e firefighte r r eceiv es an au d ible/haptic alert to b egin egress planning. Emergency mo de: Activ ated when any consumab le drops b elo w 10 % or a critical parameter (HR, ˆ T c , in-su it x O 2 , in-su it x CO 2 ) en ters a danger zone. The controll er o v errides all comfort ob jectiv es and op erates purely for sur viv al: minim u m O 2 flo w to maint ain P iO 2 ≥ 0 . 16 atm , minim u m fan sp eed, and con tinuous audible alarm. Cascade failure mo de: If multiple consum- ables are sim ultaneously critical, the con troller en ters a triage proto col: Priorit y: P iO 2 ≥ 0 . 16 | {z } highest > x CO 2 ≤ 3% | {z } second > RH ≤ 80% | {z } third > T bed ≤ 80 ◦ C | {z } low est (108) 15 Sim ulation Results W e presen t preliminary sim u lation resu lts comparing the AI-MPC con troller agai n st a fixed-setp oin t PID baseline across three scenarios. 15.1 Scenario Descriptions (i) Scenario A (Steady mo derate): Sustained mo derate exertion ( W = 250 W ) for an un- kno wn duration. (ii) Scenario B (Intermitten t burst): Alternating 5-min ute p erio ds of hea vy exertion ( W = 500 W) and 3-min ute rest p erio ds ( W = 80 W). (iii) Scenario C (Escalating thermal threa t): Mo derate exertion w ith am bient temp erature increasing linearly from 60 ◦ C to 300 ◦ C o ver 90 minutes. 40 15.2 P erformance Metrics Metric Scenario A Scenario B Scenario C PID: time to O 2 depletion (min) 142 98 107 MPC: time to O 2 depletion (min) 177 131 127 Impro v ement +24.6% +33.7% +18.7% PID: p eak x CO 2 (%) 0.48 0.72 0.5 5 MPC: p eak x CO 2 (%) 0.43 0.49 0.4 7 PID: p eak T c ( ◦ C) 38.4 38.9 39. 5 MPC: p eak T c ( ◦ C) 38.2 38.5 3 9.1 The MPC controlle r extends op erating time by 18–34% across all scenarios, w ith the largest im- pro vemen t in in termitten t-burst conditions wher e the dynamic r esour ce allo cation pro vides th e greatest adv an tage o v er fi xed-setp oin t con trol. Critically , the MPC main tains tigh ter physio logical safet y margins (lo w er p eak CO 2 and core temp erature) ev en while extending en d urance. 0 20 40 60 80 100 120 140 160 180 0 1 2 3 Time (min) O 2 remaining (kg) PID (Scenario A) MPC (Scenario A) Emergency thr eshold (10%) Figure 3: Oxygen tank depletion curv es for Scenario A. The MPC controll er’s dynamic conserv ation extends endu r ance b y m o dulating O 2 deliv ery rate as the tank depletes. 16 Discussion The Galactic Bio w are Life Sup p ort System represen ts a con vergence of closed-circuit life sup p ort engineering with mod er n AI con trol theory . S ev eral asp ects merit discussion: (i) Robustness to model error: The com bination of MPC (whic h r elies on a physics-based mo del) w ith RL (wh ic h is mo d el-free at d eplo yment) pro vides complemen tary robustn ess: the MPC h andles nominal op er ating conditions w ith optimalit y guaran tees, wh ile the RL p olicy pro vides a fallbac k cand idate action for scenarios outside the m o del’s v alidit y env elop e. A final con trol-barrier-function safet y filter sits b et w een the decision logic and th e actuators, so neither the n ominal MPC command nor th e RL fallbac k can r eac h the plan t without constrain t enforcemen t. 41 (ii) Computational feasibility: The full 1 8-state nonlinear mo d el is used by the EKF for state estimation but is too large for direct nonlinear MPC on a micro con troller within the 100 ms deadline. Instead, the MPC uses a line ar time-varying (L TV) app r oximation up dated at eac h con trol step: th e nonlinear d ynamics are linearized around the current E KF estimate ˆ x k , pro ducing time-v arying system matrices ( A k , B k ) that capture the lo cal p lan t b eha vior. Com bined with move blo cking (grouping con trol in puts ov er 3–5 step bloc ks to reduce the decision v ariable coun t) and a sh ortened effectiv e horizon of N = 15– 20 steps, th e resulting con v ex QP can b e solv ed on an emb ed ded ARM Cortex-A class SoC within ∼ 50 ms us in g a co de-generated in terior-p oint solve r. The RL w arm-start furth er reduces iteration coun t. The RL p olicy inference is a single forward pass through a small n eural net w ork ( ∼ 5000 parameters), r equiring < 1 m s. (iii) Sensor realism: A delib erate design c h oice of the Galactic Bio ware Life Sup p ort System is to rely exclusive ly on sensors that are op erationally viable in stru ctural firefigh ting. Clinical - grade biometric devices (ingestible temp erature p ills, finger p ulse o ximeters, transcutaneous CO 2 monitors) are replaced b y indirect estimatio n from rugged, we arable sensors fused with physic s-b ased mo d els. The in-suit en vironment sen s ors—measuring the ga s the firefi ghter actually breathes—serv e a du al role: direct feedbac k for life sup p ort regulation and indirect metab olic p ro xy (rising in-suit x CO 2 as a signal of increased exertion). This approac h trades some measur emen t fidelit y for op erational reliabilit y , a tradeoff that the EKF’s uncertain ty quan tification mak es explicit and manageable. (iv) Unkno wn duration handling: The dyn amic scarcit y multiplier λ ( t ) provides an elegan t, principled mec hanism for managing unkno wn mission duration. Unlik e a fixed timer, the system conti nuously re-optimizes its strategy based on remaining resources and curren t con- sumption, pro d ucing a n atural “slo w do wn” as su p plies diminish —analogous to ho w a distance runn er p aces without kno wing the exact finish line. (v) Ethical considerations: The AI con troller mak es decisions that d irectly affect fir efigh ter safet y . F ail-safe defaults (rev ert to conserv ativ e fi xed-setp oin t con trol if the AI mo d u le fails) and transparent logging of a ll co ntroller decisions for p ost-inciden t review are essentia l design requirement s. (vi) Cyb ersecurity in con tested en vironmen ts: AI-con trolled li fe sup p ort also introdu ces a cyb ersecurit y d imension, particularly in wartime or other con tested settings where adv er- saries ma y seek to degrade emergency resp on s e capabilit y . Pote ntial attac k su rfaces include sp o ofed telemetry if any wireless suit-to-command lin ks are exp osed, gradual p oisoning of the learned metabolic mod el through compromised biometric inputs du ring online adaptat ion, and sup ply-c hain compr omise of embed d ed firmw are or RL policy w eigh ts. In op erational terms, suc h in terference could force rev ersion to conserv ativ e fallbac k con trol or, in the w orst case, bias the system to w ard unsafe gas-managemen t decisions that acce lerate oxyge n de- pletion, su ppress scrubb er bypass, or degrade thermal protection. A fieldable design should therefore trea t cyb ers ecurit y as a safet y requiremen t rather than an auxiliary IT concern: in ternal comm unications should b e authen ticated and encrypted, external wireless in terfaces should b e disabled during deplo ymen t unless strictly necessary , fi rm ware a nd mo del artifacts should b e protected by hardwa re-ro oted secure b o ot and signature verificati on, EKF/state- tra jectory anomaly detection sh ou ld trigger a hardened fallbac k mo de based only on trusted physic al p lan t sens ors , and the RL p olicy should b e stress-tested offline against ad versarial observ ation and reward-manipulation scenarios. 42 16.1 Long-Duration and Multi-Sortie Op erational Gap s The p resen t analysis assum es a single con tinuous dep loymen t b eginnin g with a fresh consumable load. Extending the concept to long-duration or multi-sortie op eration in tro d uces additional en- gineering constrain ts that are not y et captured in the current state-space mo del. First, soda lime is effe ctive ly a s in gle-use sorb en t in this application, so the scrubb er canister m ust b e r ep laced b et ween deplo ymen ts. A field-serviceable cartridge with k ey ed a lignment, p ositiv e locking, and con trolled gask et compression wo uld lik ely b e required to enable rapid rep lacemen t with glo ve d hands and lo w visibility w h ile a voi d ing seal failure, b ed settling, or c hanneling. Second, the d esic- can t stage raises a separate lifetime question. Although silica gel is not consumed stoic hiometrically , its adsorption p erform ance under rep eated thermal cycling and exp osu r e to scrubb er-adjacen t tem- p eratures approac hing the 80 ◦ C th er m al-fuse threshold r emains u n quant ified for the pr esen t design. Capacit y retenti on o ve r realistic multi -sortie duty cycles must therefore b e measured b efore a de- fensible maintenance in terv al can b e sp ecified. Th ird, the cu rrent analysis treats electrical p ow er dra w—f an motors, prop ortional v alv e, sensor suite, and emb edded controll er—as unconstrained. A fi eldable system will requ ir e a battery sized for miss ion d uration plus reserv e, with the r esult- ing mass directly comp eting against oxyge n , sorb ent, and w ater budgets; the b attery architect ur e m ust also address el ev ated in-suit temp eratures through c hemistry selectio n , thermal isolation, and fault-con tainmen t design. Finally , the separated breathing-lo op architec tur e motiv ated b y the o xygen-enric hment analysis in section 6.1.3 r educes one dominant constraint but in tro d uces new ones: the mask-to-coun ter-lung circuit must m ain tain lo w d ead sp ace, lo w insp iratory r esistance, and a robu st seal relativ e to the h igher-pressure suit sh ell atmosphere, while the suit p ressurization stream ma y add another managed resource if su pplied fr om b ottle d inert gas. These iss u es define the critical path from the analytical framew ork presen ted here to a fieldable p rotot yp e. 17 Conclusion W e ha v e p resen ted the Galactic Biowa r e Life Sup p ort System : a semi-closed-circuit li fe supp ort apparatus for firefighting, go v erned by an AI-based control system th at fus es environmen tal and biometric s en sor data to optimize resource managemen t u nder uncertaint y . The k ey con tributions are: (i) A rigorous chemical and thermo d ynamic analysis of the so da lime C O 2 scrubb er (includin g state-co n s isten t formation-en thalp y c alculation, reactio n mec hanism, kinetic s, and capacit y limits), silica gel humidit y managemen t (a d sorption isotherms, LDF dynamics, h eat of ad- sorption), o xygen su pply c hain with correct endurance arithmetic, and th e o xygen-enric hm ent dynamic driven b y exhaust-v alve v ent comp ensation with pure O 2 . (ii) A semi-closed su it arc hitecture with one-w a y exhaust v alv es (consistent with NFP A 19 91 practice) and explicit treatmen t of o xygen e n r ic hment as b oth a fire-safet y constraint (23.5%) and a to xicit y constrain t (50%). (iii) A state-space form ulation of the life supp ort system as a constrained nonlinear dynamical system with 18 states (including coun ter-lung v olume, O 2 mole fractio n , scru b b er c onv er- sion fraction, cumula tive O 2 to xicit y dose, suit en vironment temp er atur es, and estimated core temper atur e), 3 con trols, and 5 disturbances—using only sensors viable in stru ctural firefighti n g, with trip le-redundant O 2 sensing and m edian v oting. 43 (iv) An MPC fr amew ork with a d ynamic r esource scarcit y m ultiplier that automatically adapts op erating strategy to unknown mission du r ation, augmen ted by an RL p olicy advisor and a final cont rol-barr ier-fu nction safet y filter thr ough whic h all candidate actuator commands pass b efore reac hing the hardw are, thereby formally enf orcing constrain t satisfactio n. (v) In tegration of e xternal situational a w areness (thermal threat, to xic gas pro ximit y , activit y classification) in to the resour ce managemen t lo op, enabling anticipato ry rather than pu r ely reactiv e con trol. Sim ulation results d emonstrate an 18–34% imp r o v ement in op erating end urance compared to fixed- setp oin t PI D con trol, while maintaining tigh ter physiolo gical and fire-safet y margins. F utu r e w ork will focus on empirical v alidation of the separated breathing-lo op arc hitecture, human-in-the-loop testing in con trolled fir e en vironments, supp ort for long-duration and m ulti-sortie op erations, and extension to m ulti-firefi gh ter co ordination where suit-to-suit comm u n ication enables collaborative resource p lann ing. 44 References [1] Ho op er, A. J., Cra wford , J. O ., and Thomas, D. An ev aluation of physio logical demands an d comfort b et w een the use of conv en tional and light w eigh t self-con tained breathing app aratus. Applie d Er go nomics , 32(4):3 99–406, 2001. [2] V ere ˇ so v´ a, T., S vetl ´ ık, J., and Kalu ˇ zn ´ ık, D. V erification of tactical and tec hn ical data of the breathing apparatus. P r o c e e dings of CBU in Natur al Scienc es and ICT , 2:100 –104, 2021. [3] Lo ve, R. G., John s tone, J. B. G., C r a wford, J., T esh, K. M., Gra veli n g, R. A., Ritc hie, P . J., and W etherill, G. Z . Study of the physiol ogical effects of wea rin g b reathing apparatus. T ec hnical Rep ort TM/94/05, In stitute of Occupational Medicine, Edinburgh, 1994 . [4] W o o d, W. B. NASA Firefighters Breathing Sys tem Program Rep ort. NAS A T ec hnical Note TN D-8497, 1977. [5] Butler, F. K., Jr., White, E., and Twa, M. Hyp ero xic m yo p ia in a closed-circuit mixed-gas scuba diver. Underse a & H yp erb aric Me dicine , 26(1):41–45 , 1999. [6] Mitc hell, S. J ., Cron j´ e, F. J., Mein tjes, W. A. J., and Britz, H. C. F atal respiratory failure during a “tec hnical” rebreather div e at extreme pressu r e. Aviation, Sp ac e, and Envir onmental Me dicine , 78(2):8 1–86, 2007. [7] Occupational Safet y and He alth Admin istration (OSHA). P ermissible ex- p osure limits—annotated tables (29 CFR 1 910.1000, T able Z-1); P ermit- required confined sp aces (29 CFR 1910 .146). U.S . Department of Lab or, 2024. https:// www.osha. gov/laws- regs/regulations/standardnumber/1910 [8] Lam b ertsen, C. J. Carb on dio xide tolerance and to xicit y . I n E nvir onmenta l Biome dic al Str ess Data Center, Institute for E nv i r onm ental Me dicine R ep ort No. 71-2, Un iv ersit y of Pennsylv a- nia, Philadelph ia, 1971 . [9] Morrison, J. B., Florio, J. T., and Butt, W. S . Effects of CO 2 insensitivit y and r esp iratory pattern on respiration in div ers. Underse a Biome dic al R ese ar ch , 8(4):20 9–217, 1981. [10] Earing, C. M. N. , McKeon, D. J., and Kubis, H.-P . Div ers revisited: The v en tilatory resp onse to carb on dio xide in exp erienced scuba divers. R espir atory Me dicine , 108(5):7 58–765, 2014. [11] Clark, J . M. and Lam b ertsen, C. J . Pu lmonary o xygen t oxic ity: A review. Pharmac olo gic al R eviews , 23(2):3 7–133, 1971. [12] Wingelaar, T. T ., v an Ooij, P . A. M., and v an Hulst, R. A. Oxygen to xicit y and Sp ecial Op erations F orces diving: Hidden and dangerous. F r ontiers in Psycholo gy , 8:1263, 20 17. [13] den Ouden, T. H. B., Wingelaar, T. T., Endert, E. L., and v an Ooij, P .-J. A. M. Lung diffusing capacit y in Dutc h Sp ecial Op erations F orces d iv ers exp osed to oxyg en rebreathers o v er 18 ye ars. Oxygen , 2(2):40– 47, 2022. [14] Mo on, R. E., C herry , A. D., Stolp, B. W., and Camp oresi, E. M. Pu lmonary gas exc hange in diving. Journal of Applie d Physiolo gy , 106(2):6 68–677, 2009. [15] Ergun, S. Fluid fl o w through pac k ed columns. Chemic al Engine ering Pr o gr ess , 48 (2):89–94 , 1952. 45 [16] Buller, M. J., Tharion, W. J., Cheuvront, S. N., Mon tain, S. J., Kenefick, R. W., C astellani, J., Latzk a, W. A., Roberts, W. S ., Ric h ter, M., Jenkin s, O. C., and Hoyt, R. W. E stimation of h um an core temper atur e from sequen tial heart rate observ at ions. Physiolo gic al M e asur ement , 34(7): 781–798, 2013. [17] Stolwijk, J. A. J. A mathematical mo del of physiolo gical temp erature regulation in man. NASA Con tractor Rep ort CR-1855, 1971. [18] Marshall, J. M. Pe r ipheral c hemoreceptors and cardio v a scular regulation. Physiolo gic al R e- views , 74(3):543 –594, 1994. [19] Simon, D. Optimal State Estimation: Kalman, H- Infinity, and Nonline ar Appr o aches . Wiley , 2006. [20] Ra wlings, J. B., Ma yne, D. Q., a nd D iehl, M. M o del Pr e dictive Contr ol: The ory, Computation, and Design . Nob Hill Publishing, 2nd edition, 2017 . [21] Sutton, R. S . and Ba r to, A. G. R einfor c ement L e arning: An Intr o duction . MIT Press, 2nd edition, 2018. [22] Ames, A. D., Xu, X., Grizz le, J. W., and T abuada, P . Control barrier function based qu adratic programs for safet y critical s y s tems. IEE E T r ansactions on Automatic Contr ol , 62(8):386 1– 3876, 2017. 46
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment