Formation of Hydroxyl Anion via a 2-Particle 1-Hole Feshbach Resonance in DEA to 2-Propanol: A Joint Experimental and Theoretical Study
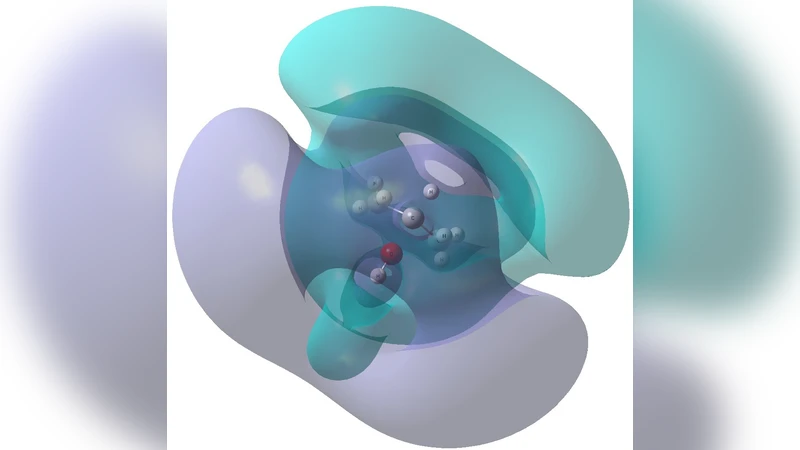
Absolute cross sections for the formation of OH- from 2-propanol (CH3CH(OH)CH3) via dissociative electron attachment (DEA) are reported in the incident electron energy range of 3.5-13 eV. Four fragment anions are observed: OH-, C2H2O-, C2H4O-, and C3H7O-. The OH- yield exhibits a pronounced resonance centered at 8.2 eV together with a broader structure extending over the 8-10 eV region. Equation-of-Motion Coupled-Cluster (electron attached) calculations with Singles and Doubles combined with a Complex Absorbing Potential (CAP/EOM-EA-CCSD) assign this feature to a two-particle-one-hole (2p-1h) core-excited Feshbach resonance. Potential energy curves along the C-OH dissociation coordinate reveal that core-excited anion states in this energy range promote efficient cleavage of the hydroxyl group. Analysis of Dyson orbitals and resonance widths demonstrates that only states with repulsive antibonding sigma(C-OH) character and sufficiently long lifetimes contribute significantly to the observed OH- production. These results provide fundamental insight into the DEA dynamics of secondary alcohols and highlight the role of multi-electron-attached resonances in site-specific bond rupture induced by low-energy electrons.
💡 Research Summary
The paper presents a combined experimental‑theoretical investigation of dissociative electron attachment (DEA) to 2‑propanol (CH₃CH(OH)CH₃) with a focus on the formation of the hydroxyl anion (OH⁻). Absolute cross sections for four fragment anions—OH⁻, C₂H₂O⁻, C₂H₄O⁻, and C₃H₇O⁻—were measured over the incident electron energy range of 3.5–13 eV using a crossed‑beam apparatus equipped with a magnetic‑field‑free electron gun and a time‑of‑flight mass spectrometer. The OH⁻ yield displays a sharp resonance centered at 8.2 eV and a broader, less structured component extending from roughly 8 to 10 eV.
To interpret these features, the authors performed Equation‑of‑Motion Coupled‑Cluster calculations for electron‑attached states (EOM‑EA‑CCSD) augmented with a Complex Absorbing Potential (CAP) to treat metastable resonances. The CAP/EOM‑EA‑CCSD results reveal a family of two‑particle‑one‑hole (2p‑1h) core‑excited Feshbach resonances in the 8–10 eV region. In a 2p‑1h configuration two electrons are promoted to virtual orbitals while a core electron is left vacant, producing a bound‑like anionic state that can decay by autodetachment or by nuclear motion.
Potential energy curves were generated by scanning the C–O bond length for the identified resonances. All 2p‑1h states possess a repulsive σ*(C‑OH) character: as the C–O distance increases, the resonance energy drops sharply, indicating that electron attachment directly weakens the C–O bond and drives its dissociation. Dyson orbital analysis shows that the excess electron density resides primarily in the σ* orbital localized on the C–O bond, confirming the antibonding nature of the resonance. The calculated resonance widths (Γ ≈ 0.1–0.3 eV) correspond to lifetimes of tens to a few hundred femtoseconds, long enough for the nuclei to move appreciably before autodetachment. Consequently, only those resonances that are both antibonding along the C–O coordinate and sufficiently long‑lived contribute significantly to the observed OH⁻ production.
The other fragment anions (C₂H₂O⁻, C₂H₄O⁻, C₃H₇O⁻) arise from higher‑energy shape resonances or from different core‑excited states that promote cleavage of C–C or C–H bonds rather than the C–O bond. The study therefore demonstrates that multi‑electron‑attached Feshbach resonances can provide a highly selective pathway for bond rupture in secondary alcohols, a mechanism that is not captured by simple one‑electron shape‑resonance models.
By integrating absolute DEA cross‑section measurements with state‑of‑the‑art CAP/EOM‑EA‑CCSD calculations, the work delivers a detailed mechanistic picture of how low‑energy electrons induce site‑specific fragmentation in 2‑propanol. The findings have broader implications for radiation chemistry (where secondary electrons are abundant), plasma processing, and the design of electron‑driven catalytic processes, highlighting the importance of multi‑electron resonances in controlling chemical reactivity at the molecular level.