Effect of oxygen content on optical, structural, and dielectric properties of Al$_x$Ta$_y$O$_z$$ thin films
This study reports on the optical, structural, and dielectric properties of aluminum tantalum oxide (Al$_x$Ta$_y$O$_z$) thin films deposited at low temperature on silicon and steel substrates by pulsed direct current reactive magnetron sputtering of a target containing 80 at.% aluminum and 20 at.% tantalum in Ar/O$_2$ atmosphere. Oxygen flow rates ranging from 5.0 to 20 sccm corresponded to O content changes from 57.7 to 69.6 at.% and resulted in large differences in dielectric behavior, from films with no measurable dielectric strength to a dielectric strength of 231 V$μ$m$^{-1}$, respectively. Ab initio calculations were employed to explain the large property changes, and we show that a decrease in the dielectric strength can be linked to the formation of metal-metal bonds in the material, when the O content is less than what would correspond to a stoichiometric Ta$_2$O$_5$ and Al$_2$O$_3$ mixture. The electronic states corresponding to the metal–metal bonds are located in the band gap close to the top of the valence band, leading to an effective band gap reduction, which is directly supported by X-ray photoelectron spectroscopy valence band measurements and by a broad optical absorption in the visible region.
💡 Research Summary
This paper investigates how varying the oxygen content during low‑temperature pulsed‑DC reactive magnetron sputtering influences the optical, structural, and dielectric properties of ternary aluminum‑tantalum oxide (AlₓTaᵧO_z) thin films deposited on silicon and steel substrates. A target composed of 80 at.% Al and 20 at.% Ta was sputtered in an Ar/O₂ atmosphere while the oxygen flow was adjusted from 5 to 20 sccm, producing films with oxygen atomic fractions ranging from 57.7 % to 69.6 %. Energy‑dispersive X‑ray spectroscopy confirmed that the Al/Ta ratio in the films is systematically lower than in the target, a consequence of the lighter Al atoms undergoing more scattering than the heavier Ta atoms.
X‑ray diffraction patterns displayed only broad features, indicating that all films are essentially amorphous. At the lowest oxygen flow (5 sccm) a weak diffraction contribution near 35–40° matches tantalum metal peaks, suggesting the presence of metallic Ta clusters or metal‑metal bonding motifs. With increasing oxygen flow the broad maximum shifts toward 25–30°, corresponding to Ta₂O₅ reflections, implying a transition toward a more fully oxidized network.
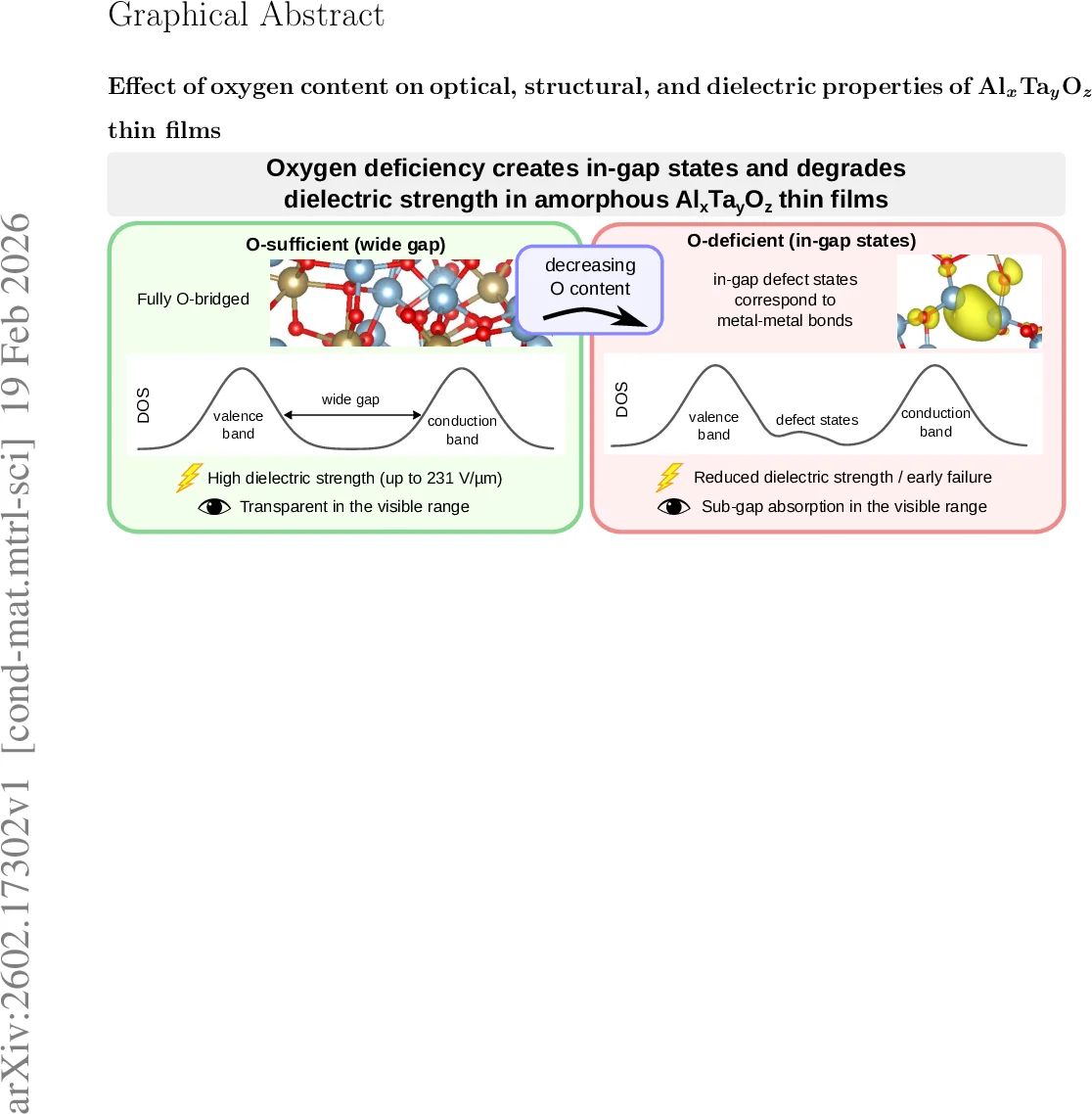
Dielectric breakdown measurements using a pin‑plate configuration revealed a dramatic dependence on oxygen content. The film deposited at 5 sccm broke down immediately upon voltage application, precluding a strength value. Films grown at 6 sccm and above exhibited measurable breakdown fields that increased monotonically from 96 V µm⁻¹ (6 sccm) to 231 V µm⁻¹ (20 sccm). The authors attribute the low breakdown strength at low oxygen to conductive pathways formed by metal‑metal clusters, whereas higher oxygen yields a more insulating, defect‑sparse matrix.
Optical constants were extracted over a spectral range of 0.003–10.8 eV by simultaneously fitting ellipsometric and spectrophotometric data with the Universal Dispersion Model. Oxygen‑deficient films showed a broad absorption tail extending into the visible region (≈2–3 eV), consistent with sub‑bandgap states. In contrast, oxygen‑rich films displayed a well‑defined optical band gap near 4.4 eV and high transparency in the visible.
To rationalize these observations, the authors performed density‑functional theory (DFT) simulations of amorphous Al‑Ta‑O networks containing ~150 atoms. Using a melt‑quench protocol followed by geometry optimization, models with 58–63 % oxygen were generated. Hybrid PBE0 calculations revealed that when the oxygen fraction falls below ~70 %, Al–Al, Ta–Ta, and Al–Ta metal‑metal bonds emerge, introducing electronic states within the band gap close to the valence‑band maximum. These in‑gap states reduce the effective band gap, explain the visible‑range absorption, and provide pathways for early dielectric breakdown. X‑ray photoelectron spectroscopy (XPS) valence‑band measurements corroborated the presence of such states in the most oxygen‑deficient films, while oxygen‑rich films showed a clean valence‑band edge.
Overall, the study demonstrates a clear structure‑property relationship: oxygen deficiency promotes metal‑metal bonding, creates in‑gap defect states, narrows the optical band gap, and severely degrades dielectric strength. Sufficient oxygen supply suppresses these motifs, leading to a fully oxidized amorphous network with a wide band gap, high dielectric breakdown strength, and excellent optical transparency. The findings provide a practical guideline for tailoring ternary Al‑Ta‑O thin films for applications ranging from high‑voltage insulation to transparent optoelectronic components.
Comments & Academic Discussion
Loading comments...
Leave a Comment