Spatio-temporal air flow properties in a 3D personalised model of the human lung
We propose a multi-scale lung model to investigate spatio-temporal distributions of ventilation variables. Lung envelope and large airway geometries are derived from CT scans; smaller airways are gene
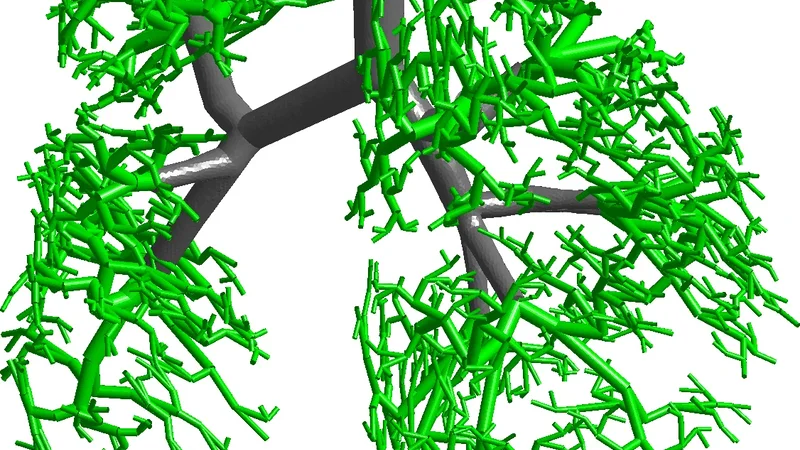
We propose a multi-scale lung model to investigate spatio-temporal distributions of ventilation variables. Lung envelope and large airway geometries are derived from CT scans; smaller airways are generated using a physiologically consistent algorithm. Tissue mechanics is modeled using nonlinear elasticity under small deformations, coupled with local air pressure from fluid dynamics within the bronchial tree. Airflow accounts for inertia and static airway compliance. Simulations employ finite elements. Using this model, we explore spatio-temporal airflows and shear stresses distributions.
💡 Research Summary
The authors present a comprehensive multi‑scale computational model of the human lung that integrates patient‑specific anatomy with physiologically realistic airway generation and tissue mechanics. Large‑scale geometry (lung envelope and the first two generations of the bronchial tree) is extracted from high‑resolution CT scans, ensuring that the model reflects the individual’s anatomical variability. To complete the airway network down to the 15th generation, a deterministic algorithm based on known branching ratios, diameter‑length relationships, and symmetry rules is employed, producing a seamless conduit from the trachea to the terminal bronchioles.
Tissue mechanics are described by a nonlinear elastic constitutive law, but the deformation is assumed to be small enough to linearize the strain‑displacement relationship. This elastic response is coupled to the local air pressure obtained from the fluid dynamics solution, establishing a two‑way feedback loop: airway pressure deforms the surrounding parenchyma, and the resulting tissue strain modifies airway cross‑sectional area and thus flow resistance.
Airflow is modeled using the incompressible Navier‑Stokes equations with the full inertia term retained, distinguishing the approach from many lung models that rely on quasi‑steady or purely resistive formulations. In addition, a static airway compliance term is introduced to capture the elastic expansion of airway walls under pressure. This combination allows the simulation to reproduce both inertial effects (important during rapid acceleration of air) and the gradual widening of airways as they experience higher transmural pressures.
The computational framework employs finite‑element (FE) discretization. High‑resolution meshes are generated for the large airways and lung parenchyma, while a parametric meshing strategy is used for the numerous small airways to keep the total number of degrees of freedom tractable. Time‑dependent simulations are performed over a full breathing cycle (typically 1 s of inhalation followed by 2 s of exhalation) with a time step of 0.01 s. At each step, the solver provides three‑dimensional fields of velocity, pressure, wall shear stress, and tissue strain, enabling a detailed spatio‑temporal mapping of ventilation variables.
Key findings include: (1) a pronounced non‑linear amplification of pressure drops from the central bronchi to the distal airways, indicating that small changes in upstream resistance can lead to large variations in terminal airway pressure; (2) localized peaks of wall shear stress during the early inhalation phase, which may be relevant to mechanotransduction pathways implicated in airway remodeling; and (3) a feedback mechanism whereby parenchymal expansion modestly enlarges airway diameters, thereby reducing flow resistance and smoothing the pressure gradient across the lung. These phenomena collectively generate heterogeneous ventilation patterns even under normal, steady breathing conditions.
The authors acknowledge several limitations. The assumption of small deformations precludes accurate representation of large tidal volumes or pathological over‑inflation. Only static compliance is considered, neglecting viscoelastic damping and dynamic airway wall behavior that become important during rapid breathing or airway obstruction. Moreover, the model does not yet incorporate patient‑specific breathing patterns, muscle forces, or the influence of gravity on regional ventilation.
Future work is outlined to address these gaps: incorporating fully nonlinear tissue mechanics, adding viscoelastic and dynamic compliance models for airway walls, and coupling the lung model with a chest wall and diaphragm mechanics module. Validation against in‑vivo measurements such as hyperpolarized gas MRI or electrical impedance tomography is also planned, which would strengthen the model’s translational potential.
In summary, this study delivers a robust, anatomically personalized lung model that simultaneously resolves airflow dynamics and tissue mechanics across multiple scales. By revealing how pressure, shear stress, and tissue deformation interact over time and space, the work provides a valuable platform for investigating the mechanistic basis of heterogeneous ventilation in health and disease, and it holds promise for informing patient‑specific diagnostic and therapeutic strategies in conditions such as asthma, chronic obstructive pulmonary disease, and pulmonary fibrosis.
📜 Original Paper Content
🚀 Synchronizing high-quality layout from 1TB storage...