Competing Risk Analysis in Cardiovascular Outcome Trials: A Simulation Comparison of Cox and Fine-Gray Models
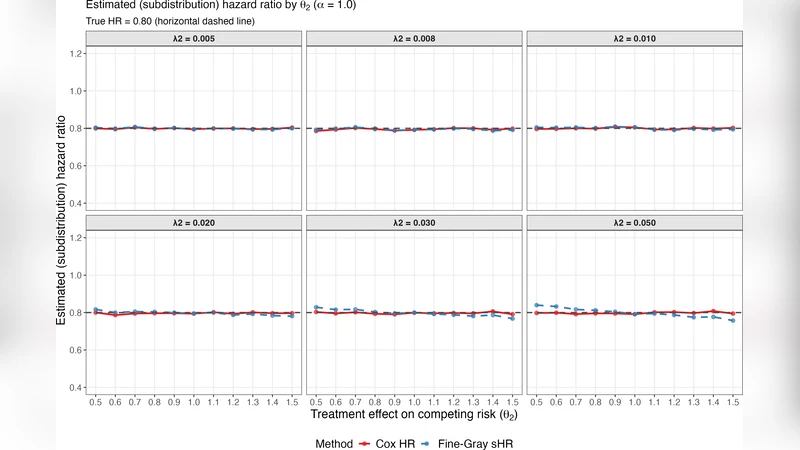
Cardiovascular outcome trials commonly face competing risks when non-CV death prevents observation of major adverse cardiovascular events (MACE). While Cox proportional hazards models treat competing events as independent censoring, Fine-Gray subdistribution hazard models explicitly handle competing risks, targeting different estimands. This simulation study using bivariate copula models systematically varies competing event rates (0.5%-5% annually), treatment effects on competing events (50% reduction to 50% increase), and correlation structures to compare these approaches. At competing event rates typical of CV outcome trials (~1% annually), Cox and Fine-Gray produce nearly identical hazard ratio estimates regardless of correlation strength or treatment effect direction. Substantial divergence occurs only with high competing rates and directionally discordant treatment effects, though neither estimator provides unbiased estimates of true marginal hazard ratios under these conditions. In typical CV trial settings with low competing event rates, Cox models remain appropriate for primary analysis due to superior interpretability. Pre-specified Cox models should not be abandoned for competing risk methods. Importantly, Fine-Gray models do not constitute proper sensitivity analyses to Cox models per ICH E9(R1), as they target different estimands rather than testing assumptions. As supplementary analysis, cumulative incidence using Aalen-Johansen estimator can provide transparency about competing risk impact. Under high competing-risk scenarios, alternative approaches such as inverse probability of censoring weighting, multiple imputation, or inclusion of all-cause mortality in primary endpoints warrant consideration.
💡 Research Summary
This paper investigates how competing risks—specifically non‑cardiovascular death that precludes observation of major adverse cardiovascular events (MACE)—affect the analysis of cardiovascular outcome trials (CVOTs). Using a bivariate copula framework, the authors generate synthetic trial data in which they systematically vary the annual competing‑risk rate (0.5 % to 5 %), the treatment effect on the competing event (from a 50 % reduction to a 50 % increase), and the correlation between the two event times. For each scenario, 10,000 virtual patients are simulated and the process is repeated 1,000 times to obtain stable performance metrics. Both the conventional Cox proportional‑hazards model, which treats competing events as independent censoring, and the Fine‑Gray subdistribution‑hazard model, which directly models the cumulative incidence of MACE in the presence of competing risks, are fitted to the data.
The key findings are threefold. First, when the competing‑risk rate is low—approximately 1 % per year, a level typical of most modern CVOTs—Cox and Fine‑Gray produce virtually identical hazard‑ratio estimates, regardless of the strength of the correlation or the direction of the treatment effect on the competing event. This demonstrates that, in routine trial settings, the simpler Cox model does not suffer from appreciable bias due to competing risks. Second, substantial divergence between the two estimators emerges only when the competing‑risk rate is high (≈5 % per year) and the treatment effect on the competing event is directionally opposite to its effect on MACE (e.g., a drug that reduces MACE but increases non‑CV death). In these extreme scenarios the Cox model tends to underestimate the true marginal effect, while the Fine‑Gray model overestimates the subdistribution effect. Third, even under these extreme conditions neither model recovers the true marginal (population‑average) hazard ratio without bias, reflecting a fundamental identifiability limitation: the marginal hazard ratio cannot be uniquely estimated from data that are subject to informative censoring by competing events.
The authors also discuss regulatory implications. According to ICH E9(R1), the Fine‑Gray model targets a different estimand than the Cox model and therefore cannot be used as a “sensitivity analysis” for the Cox model; it is not a test of the Cox model’s assumptions but an analysis of a distinct causal question. Instead, they recommend presenting cumulative‑incidence functions estimated by the Aalen‑Johansen method as a transparent descriptive supplement that quantifies the absolute impact of competing risks. For settings where competing risks are non‑negligible, they suggest alternative strategies such as inverse‑probability‑of‑censoring weighting, multiple imputation for the censored outcomes, or redefining the primary endpoint to include all‑cause mortality.
In summary, the simulation study provides strong empirical support for retaining pre‑specified Cox proportional‑hazards models as the primary analysis in typical CVOTs, where competing‑risk rates are low and the Cox estimand remains interpretable. Fine‑Gray models are valuable for exploratory or confirmatory analyses when the scientific question explicitly concerns the subdistribution hazard, but they should not replace Cox models nor be presented as a robustness check. The paper thus offers practical guidance for trial statisticians on model selection, interpretation of estimands, and appropriate supplementary analyses in the presence of competing risks.
Comments & Academic Discussion
Loading comments...
Leave a Comment