Foundation Models for Medical Imaging: Status, Challenges, and Directions
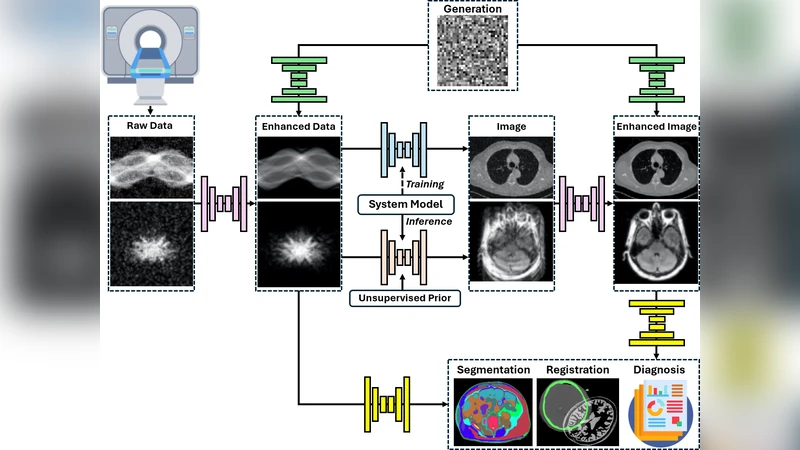
Foundation models (FMs) are rapidly reshaping medical imaging, shifting the field from narrowly trained, task-specific networks toward large, general-purpose models that can be adapted across modalities, anatomies, and clinical tasks. In this review, we synthesize the emerging landscape of medical imaging FMs along three major axes: principles of FM design, applications of FMs, and forward-looking challenges and opportunities. Taken together, this review provides a technically grounded, clinically aware, and future-facing roadmap for developing FMs that are not only powerful and versatile but also trustworthy and ready for responsible translation into clinical practice.
💡 Research Summary
This review provides a comprehensive synthesis of the emerging landscape of foundation models (FMs) in medical imaging, organizing the discussion around three major axes: design principles, application domains, and forward‑looking challenges and opportunities. The authors begin by defining an FM as a large‑scale, pre‑trained neural network that can be adapted to a wide variety of imaging modalities (CT, MRI, ultrasound, X‑ray, etc.), anatomical regions, and clinical tasks with minimal task‑specific data. They argue that the shift from narrowly trained, task‑specific networks to these general‑purpose models is driven by three core design tenets.
First, data scaling and self‑supervision are essential because high‑resolution 3D volumes are expensive to label. The review highlights multi‑task pre‑training strategies that combine image reconstruction, mask prediction, and sequence modeling to learn robust, modality‑agnostic representations. Second, multimodal integration is achieved through tokenization and cross‑attention mechanisms that fuse imaging data with electronic health records, pathology slides, and genomic information, enabling a unified patient‑level embedding space. This is particularly valuable for rare disease discovery and for institutions that possess heterogeneous data sources. Third, efficient fine‑tuning is emphasized; techniques such as Low‑Rank Adaptation (LoRA), prompt tuning, and adapter modules allow clinicians to specialize a frozen FM for a specific diagnostic or therapeutic task without retraining the entire parameter set, dramatically reducing computational cost and data requirements.
The authors then map the current applications of medical imaging FMs into four categories. In diagnostic assistance, few‑shot and zero‑shot capabilities allow rapid adaptation to new pathologies or anatomical sites, delivering sensitivity and specificity comparable to expert radiologists while reducing workload. For image reconstruction and denoising, FM‑based super‑resolution and de‑noising pipelines can recover high‑quality images from low‑dose CT or accelerated MRI acquisitions, preserving fine anatomical detail better than conventional CNNs. In treatment planning, FMs support automated segmentation of radiation therapy targets and dose‑optimization by jointly processing CT and PET data, thereby streamlining workflow and improving plan consistency. Finally, clinical report automation combines vision‑language models to generate structured radiology reports in natural language, standardizing terminology and cutting report‑writing time.
Despite these promising results, the review devotes substantial space to the trustworthiness, transparency, and ethical dimensions that are critical for clinical translation. Uncertainty quantification (via Bayesian deep learning, ensembles, or temperature scaling) and explainable AI (XAI) methods are presented as mandatory safeguards against harmful misclassifications. The authors discuss data bias—stemming from demographic or institutional variations—and advocate for federated learning and differential privacy to protect patient confidentiality while still enabling large‑scale collaborative training. Regulatory considerations are also examined; the FDA, EMA, and other agencies are still formulating frameworks for AI/ML medical devices, and the black‑box nature of FMs complicates validation, necessitating standardized benchmarks and continuous post‑deployment monitoring.
The review concludes with a roadmap for responsible FM deployment in medical imaging. The proposed pipeline includes (1) building ethically governed, multimodal datasets; (2) developing self‑supervised, multi‑task pre‑training frameworks; (3) applying parameter‑efficient adaptation methods for downstream clinical tasks; (4) embedding uncertainty estimation and XAI modules; and (5) establishing transparent documentation and regulatory compliance processes. By following this iterative, interdisciplinary approach, the field can harness the transformative potential of foundation models—high performance, versatility, and scalability—while mitigating risks related to safety, bias, and resource consumption. In sum, foundation models promise to reshape medical imaging from a collection of isolated deep‑learning solutions into an integrated, trustworthy AI ecosystem ready for real‑world clinical impact.
Comments & Academic Discussion
Loading comments...
Leave a Comment