Bistability to Quad-stability: Emergence of Hybrid Phenotypes & Enhanced Spatio-temporal Plasticity in Presence of Host-Circuit Coupling
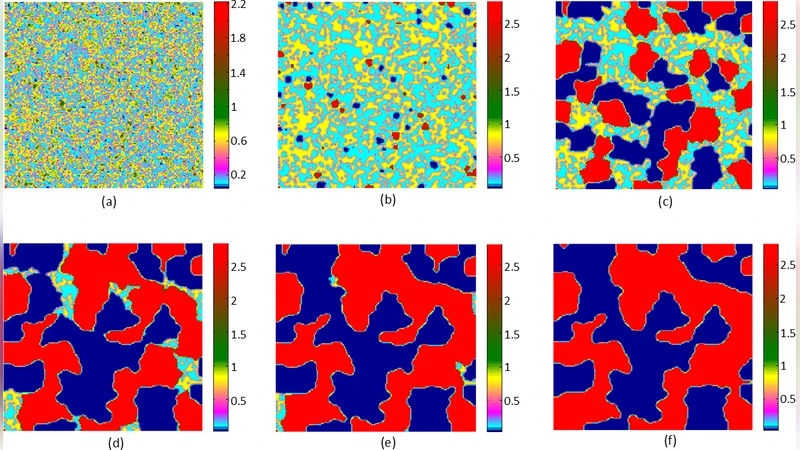
In the context of multistability driven diseases, like cancer, spatiotemporal plasticity plays a significant role to achieve a spectrum of phenotypic variations. The interplay between gene regulatory networks and environmental factors, such as resource competition and spatial diffusion, plays a crucial role in determining cellular behaviour and phenotypic heterogeneity. Though reaction diffusion frameworks have been widely applied in developmental biology, less attention has been paid to the simultaneous effects of resource competition and growth feedback on spatial organization. In this paper, we observed that a bistable genetic circuit under high resource competition due to growth feedback gives rise to multiple emergent phenotypes, as observed in cancer systems. Furthermore, we observed how spatial diffusion coupled with intrinsic nonlinearity can drive the emergence of distinct spatial dynamics over time. The observed spatiotemporal plasticity can also be driven by the comparative stability of the fixed points, diffusivity, and asymmetry of diffusion. Our findings highlight that growth-induced resource competition combined with diffusion can provide deeper insights into metastasis and cancer progression.
💡 Research Summary
The paper investigates how the interplay of growth‑induced resource competition and spatial diffusion can transform a classic bistable genetic circuit into a system exhibiting quad‑stability, thereby generating hybrid phenotypes and complex spatio‑temporal plasticity reminiscent of cancer progression. The authors begin by outlining the biological motivation: multistable gene regulatory networks are known to underlie phenotypic heterogeneity in tumors, yet most theoretical work has focused on intrinsic circuit non‑linearity or external signals, neglecting the simultaneous impact of cellular growth dynamics and diffusion of intracellular components.
To fill this gap, they construct a reaction‑diffusion model that couples three core elements: (1) a positive‑feedback toggle switch comprising two mutually inhibitory transcription factors (X and Y), which on its own yields two stable steady states; (2) a growth feedback loop in which cellular proliferation consumes a shared resource R (e.g., ATP, nutrients, ribosomes), thereby reducing the effective production rates of X and Y as the population expands; and (3) spatial diffusion of X, Y, and R with potentially distinct diffusion coefficients (D_X, D_Y, D_R), allowing for asymmetric spread of signaling molecules versus resources.
Mathematically, the system is described by a set of partial differential equations:
∂X/∂t = f_X(X,Y,R) + D_X∇²X,
∂Y/∂t = f_Y(X,Y,R) + D_Y∇²Y,
∂R/∂t = σ – δ·N·R – λ·R + D_R∇²R,
where N denotes cell number, σ is resource influx, δ captures consumption per cell, and λ represents natural decay. The production functions f_X and f_Y incorporate Hill‑type activation and repression terms, modulated by the instantaneous resource level R.
A bifurcation analysis reveals that when the product of the competition strength (γ) and the growth‑feedback coefficient (α) exceeds a critical threshold, the system acquires two additional fixed points: one stable intermediate state and one unstable saddle. Consequently, the phase space expands from two to four equilibria (two stable, two unstable), a transition the authors label “bistable → quad‑stable.” Linear stability is assessed via the Jacobian eigenvalues; the new stable equilibrium exhibits a more negative real part, indicating heightened robustness under resource‑limited conditions.
The spatial dimension is explored through Turing‑type analysis and extensive numerical simulations on one‑ and two‑dimensional lattices with Neumann boundary conditions. The authors demonstrate that diffusion asymmetry (e.g., D_X ≠ D_Y) critically shapes pattern formation. Low diffusion of the resource (small D_R) creates localized “nuclei” where one of the stable states dominates, while higher diffusion of the transcription factors allows these nuclei to interact, generating traveling fronts and oscillatory patches. When γ·α is modest, only classic bistable domains appear, alternating between X‑high and Y‑high regions. As γ·α increases, hybrid domains (partial EMT‑like states) emerge, co‑existing with the original extremes. At the highest competition levels, all four equilibria manifest as distinct spatial clusters, and the fronts between them accelerate dramatically, mimicking invasive fronts observed in metastatic tumors.
Biologically, the intermediate stable state corresponds to hybrid epithelial/mesenchymal phenotypes that have been implicated in collective migration and therapy resistance. The model predicts that aggressive tumors, which experience strong growth‑driven resource depletion, are more likely to occupy this hybrid basin, thereby explaining the prevalence of mixed phenotypes in high‑grade cancers. Moreover, the sensitivity of front speed to diffusion ratios suggests that microenvironmental heterogeneities—such as variable extracellular matrix density or differential cytokine diffusion—could bias the direction and speed of metastatic spread.
The authors discuss therapeutic implications: interventions that alleviate resource competition (e.g., metabolic inhibitors) or homogenize diffusion (e.g., matrix‑modifying agents) could collapse the quad‑stable regime back to bistability, reducing the emergence of hybrid phenotypes and slowing invasive front propagation.
Limitations are acknowledged. The model treats the resource as a single scalar, whereas real cells juggle multiple metabolites (oxygen, glucose, amino acids). Extending the framework to multi‑resource dynamics and incorporating three‑dimensional tissue architecture, vascular networks, and stochastic gene expression would enhance realism. Experimental validation is proposed via microfluidic tumor‑on‑a‑chip platforms where nutrient flow, cell density, and diffusion barriers can be precisely tuned.
In conclusion, the study provides a rigorous theoretical demonstration that growth‑induced resource competition, when coupled with spatial diffusion, can fundamentally reshape the stability landscape of a genetic circuit, giving rise to quad‑stability, hybrid cell states, and rich spatio‑temporal dynamics. These insights deepen our mechanistic understanding of cancer heterogeneity and metastasis, and open new avenues for targeting the metabolic‑diffusive axis in therapeutic strategies.
Comments & Academic Discussion
Loading comments...
Leave a Comment