Mathematical modeling of 1,2-propanediol utilization bacterial microcompartments in vivo activity
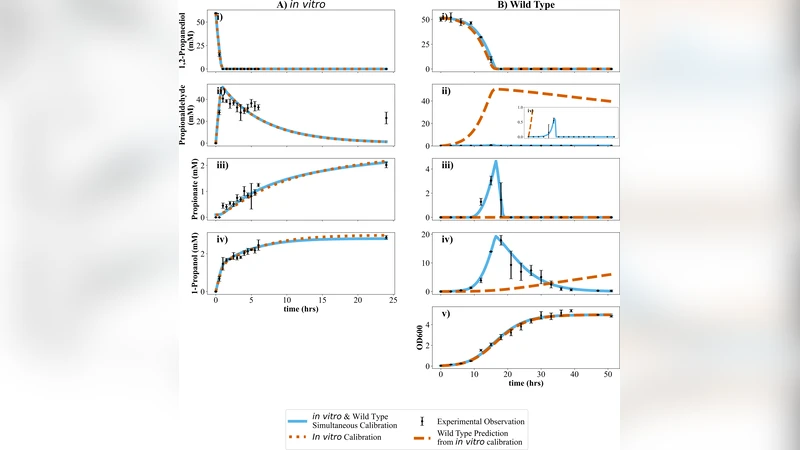
On exposure to 1,2-propanediol (1,2-PD), Salmonella enterica serovar Typhimurium LT2 produces 1,2-PD utilization (Pdu) microcompartments (MCPs), nanoscale protein-bound shells that encapsulate metabolic enzymes. MCPs serve as a bioengineering platform to study reaction organization and enhance flux through specific pathways. However, a recently published assay of purified wild-type (WT) MCPs reported metabolic activity that differed markedly from that observed in vivo. Using kinetic modeling, we attribute these discrepancies to in vivo cell growth and to the cytosolic presence of MCP-associated enzymes and promiscuous alcohol dehydrogenases, which are not present in the purified MCPs. Assays of purified MCPs in E. coli lysate, together with an LT2 growth assay in which the native Pdu MCP-associated alcohol dehydrogenase, PduQ, was knocked out, support the conclusion that exogenous Pdu cytosolic enzyme activity can narrow the gap between in vitro and in vivo experiments. Our modeling further suggests that MCP-localized enzymes contribute little to in vivo metabolic flux downstream of PduCDE. We therefore propose a revised in vivo model of WT growth on 1,2-PD in which PduCDE is fully encapsulated, while much of the downstream Pdu activity occurs in the cytosol.
💡 Research Summary
The paper revisits the metabolic activity of the 1,2‑propanediol (1,2‑PD) utilization microcompartments (Pdu MCPs) produced by Salmonella enterica serovar Typhimurium LT2 when the bacterium is grown on 1,2‑PD. Earlier work had reported a striking discrepancy between the kinetic parameters measured in purified wild‑type (WT) MCPs in vitro and the growth‑associated fluxes observed in vivo. The authors set out to explain this gap using kinetic modeling, targeted genetic manipulation, and hybrid in‑vitro assays.
First, they argue that living cells continuously grow and exchange metabolites with the environment, which inherently raises the effective substrate concentration at the enzyme sites compared with a static in‑vitro assay. Second, they point out that several Pdu‑associated enzymes, most notably the alcohol dehydrogenase PduQ, are present in the cytosol in addition to being encapsulated within the MCP shell. These cytosolic enzymes can act on 1,2‑PD or its intermediates, providing an auxiliary pathway that is absent from purified MCP preparations.
To test these ideas, the authors built a kinetic model that incorporates three key variables: (i) cell growth rate (affecting volume and substrate dilution), (ii) the activity of cytosolic Pdu‑related enzymes, and (iii) the degree of encapsulation of each enzyme. Simulations showed that the initial dehydratase complex PduCDE must be fully encapsulated to prevent leakage of the toxic aldehyde intermediate, but downstream enzymes (PduP, PduL, PduQ) can function efficiently in the cytosol without compromising overall flux.
Experimental validation involved two complementary approaches. In one, purified WT MCPs were mixed with an Escherichia coli lysate that contains endogenous, non‑specific alcohol dehydrogenases. The lysate restored a substantial portion of the activity lost in the pure MCP assay, indicating that cytosolic dehydrogenases can complement the encapsulated pathway. In a second approach, the authors constructed an LT2 strain lacking the native MCP‑associated alcohol dehydrogenase PduQ. This knockout displayed a markedly slower growth rate on 1,2‑PD, confirming that PduQ’s cytosolic activity contributes significantly to the in‑vivo flux.
The combined modeling and experimental data lead the authors to propose a revised in‑vivo picture: PduCDE is sequestered inside the MCP shell, acting as a protective “reaction chamber,” while the majority of downstream Pdu enzymatic steps occur in the cytosol. This hybrid organization reconciles the previously observed kinetic discrepancy and suggests that MCPs serve more as a selective barrier for the most hazardous step rather than a complete encapsulation device for the entire pathway.
From a synthetic biology perspective, the findings have important implications. Engineering MCPs to improve metabolic flux should consider not only the encapsulation of target enzymes but also the deliberate placement of downstream activities in the cytosol to exploit native cellular co‑factor pools and transport mechanisms. Over‑encapsulation may unnecessarily limit substrate access and reduce overall productivity, whereas a balanced distribution—protecting only the toxic intermediates while allowing downstream steps to remain cytosolic—could maximize both safety and efficiency. The study thus provides a nuanced framework for future design of MCP‑based biocatalytic systems.
Comments & Academic Discussion
Loading comments...
Leave a Comment