A dialog between cell adhesion and topology at the core of morphogenesis
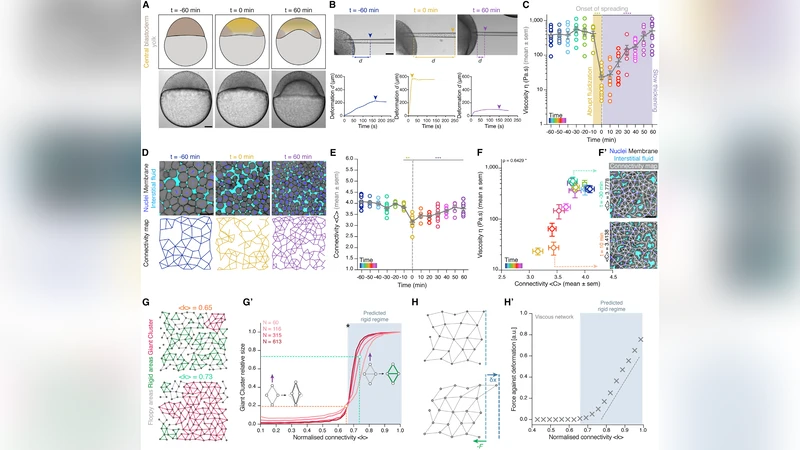
During the development of an organism, cells must coordinate and organize to generate the correct shape, structure, and spatial patterns of tissues and organs, a process known as morphogenesis. The morphogenesis of embryonic tissues is supported by multiple processes that induce the precise physical deformations required for tissues to ultimately form organs with complex geometries. Among the most active players shaping the morphogenetic path are fine-tuned changes in cell adhesion. We review here recent advances showing that changes of a local, pair-wise property defined at the cell-cell contact level has important global consequences for embryonic tissue topology, being determinant in defining both the geometric and material properties of early embryo tissues.
💡 Research Summary
The paper provides a comprehensive review of how subtle, locally‑controlled changes in cell‑cell adhesion drive global alterations in tissue topology and material properties during embryonic morphogenesis. Beginning with an overview of morphogenetic processes, the authors emphasize that tissue shaping is not merely a matter of cellular movement but also of dynamic regulation of adhesive interactions at the level of individual cell contacts. Molecular mechanisms are detailed, highlighting the roles of cadherin‑catenin complexes, integrins, and other adhesion proteins whose expression levels and membrane distribution dictate contact area, angle, and duration. These local adhesion variations modulate actomyosin contractility, thereby reshaping the internal stress network and influencing how cells transmit forces to one another.
The review then turns to quantitative modeling, describing how vertex and cellular Potts models incorporate adhesion energy as a tunable parameter. Simulations reveal that crossing a critical adhesion threshold can trigger a rapid topological phase transition: regular polygonal arrays become irregular, average cell area and perimeter shift dramatically, and overall tissue curvature changes. This demonstrates that minute pair‑wise adhesion adjustments can precipitate system‑wide reorganization.
Experimental evidence from several model organisms supports the theoretical framework. In mouse blastocyst formation, reduced basal‑lamina adhesion leads to a transition from spherical to cylindrical geometry. Drosophila gastrulation shows that spatial redistribution of E‑cadherin reorients cell edges, generating complex curvature in the epithelial sheet. Zebrafish neural tube closure involves localized up‑regulation of N‑cadherin, which pulls neighboring cells together to produce the characteristic tube shape. Across these systems, the same principle holds: local adhesion modulation determines both the geometric layout and the mechanical stiffness of the emerging tissue.
The authors also discuss implications for tissue engineering and regenerative medicine. By fine‑tuning cadherin expression or mimicking adhesion gradients in vitro, researchers can steer the self‑assembly of engineered tissues toward desired shapes and mechanical strengths. For instance, suppressing E‑cadherin yields more compliant cardiac patches, whereas overexpressing N‑cadherin produces thicker, stiffer constructs.
In conclusion, the review argues that cell adhesion functions as a dynamic “tuning knob” that simultaneously governs tissue topology and material properties. A full understanding of morphogenesis therefore requires an integrated approach that links molecular adhesion pathways, cellular mechanical responses, and emergent tissue‑level topology. Future work should focus on real‑time imaging of adhesion dynamics, development of high‑resolution quantitative models, and translation of these insights into controllable strategies for building functional, shape‑specific tissues.
Comments & Academic Discussion
Loading comments...
Leave a Comment