Arxiv 2512.21652
📝 Original Info
- Title: Arxiv 2512.21652
- ArXiv ID: 2512.21652
- Date: 2025-12-25
- Authors: Zi Wang, Mingkai Huang, Zhang Shi, Hongjie Hu, Lan Lan, Hui Zhang, Yan Li, Xi Hu, Qing Lu, Zongming Zhu, Qiong Yao, Yuxiang Dai, Fanwen Wang, Yinzhe Wu, Jun Lyu, Qianqian Gao, Guangming Xu, Zhenxuan Zhang, Haosen Zhang, Qing Li, Guangming Wang, Tianxing He, Lizhen Lan, Siyue Li, Le Xue, Mengting Sun, Yuntong Lyu, Junpu Hu, Jiayu Zhu, Rizwan Ahmad, Zhengyu Bu, Xianling Qian, Guanke Cai, Ruiyu Cao, Weirui Cai, Chang Xu, Yuyang Ren, Feidan Yu, Siying Ma, Ziqiang Xu, Xinran Chen, Sha Hua, Daniel Kim, Yajing Zhang, Chen Ouyang, Wenjia Bai, Jing Qin, Yucheng Yang, Daniel Rueckert, He Wang, Qian Tao, Claudia Prieto, Michael Markl, Alistair Young, Lianming Wu, Shuo Wang, Chen Qin, Mengsu Zeng, Xihong Hu, Haibo Xu, Xiaobo Qu, Hao Li, Guang Yang, Chengyan Wang
📝 Abstract
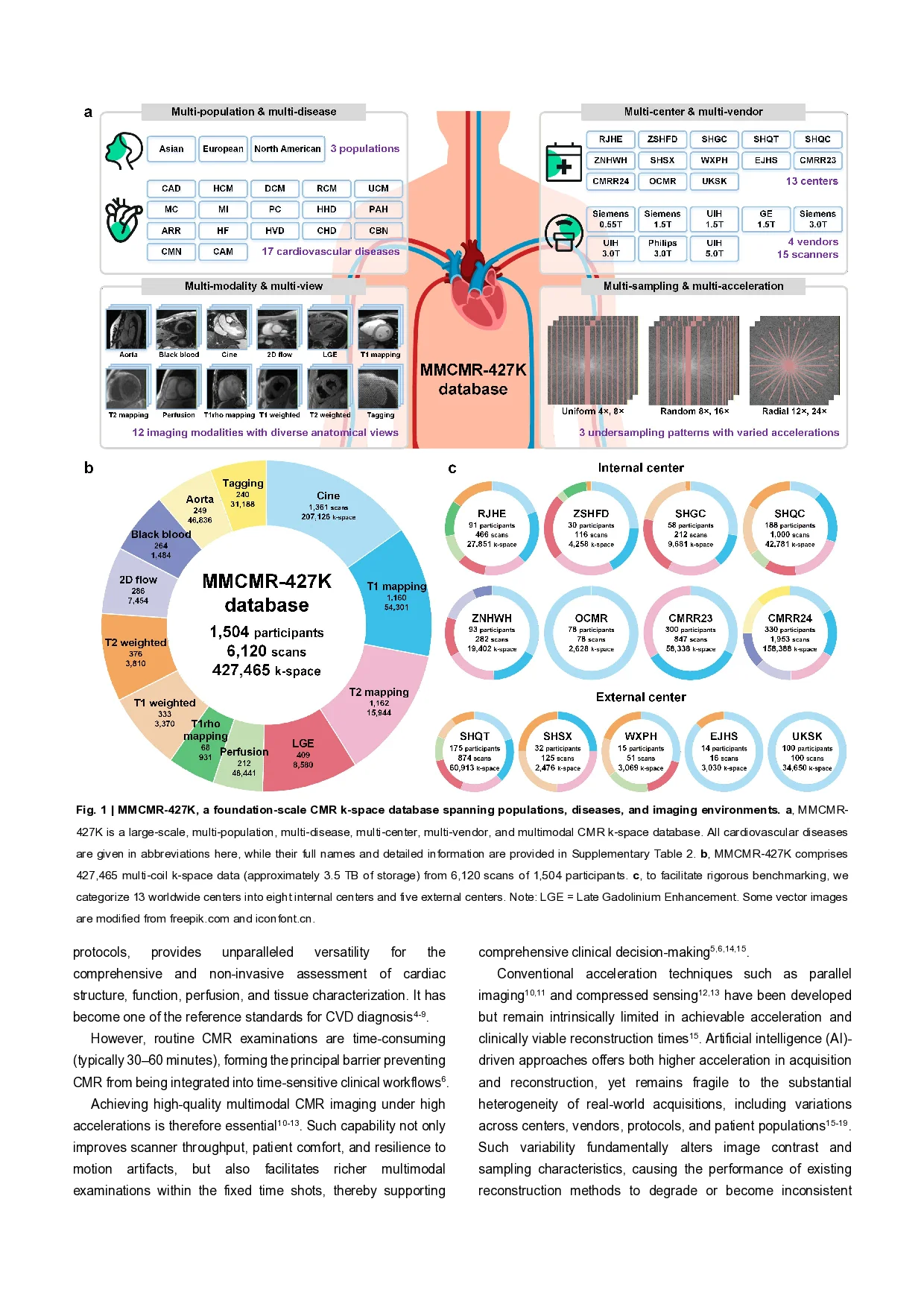
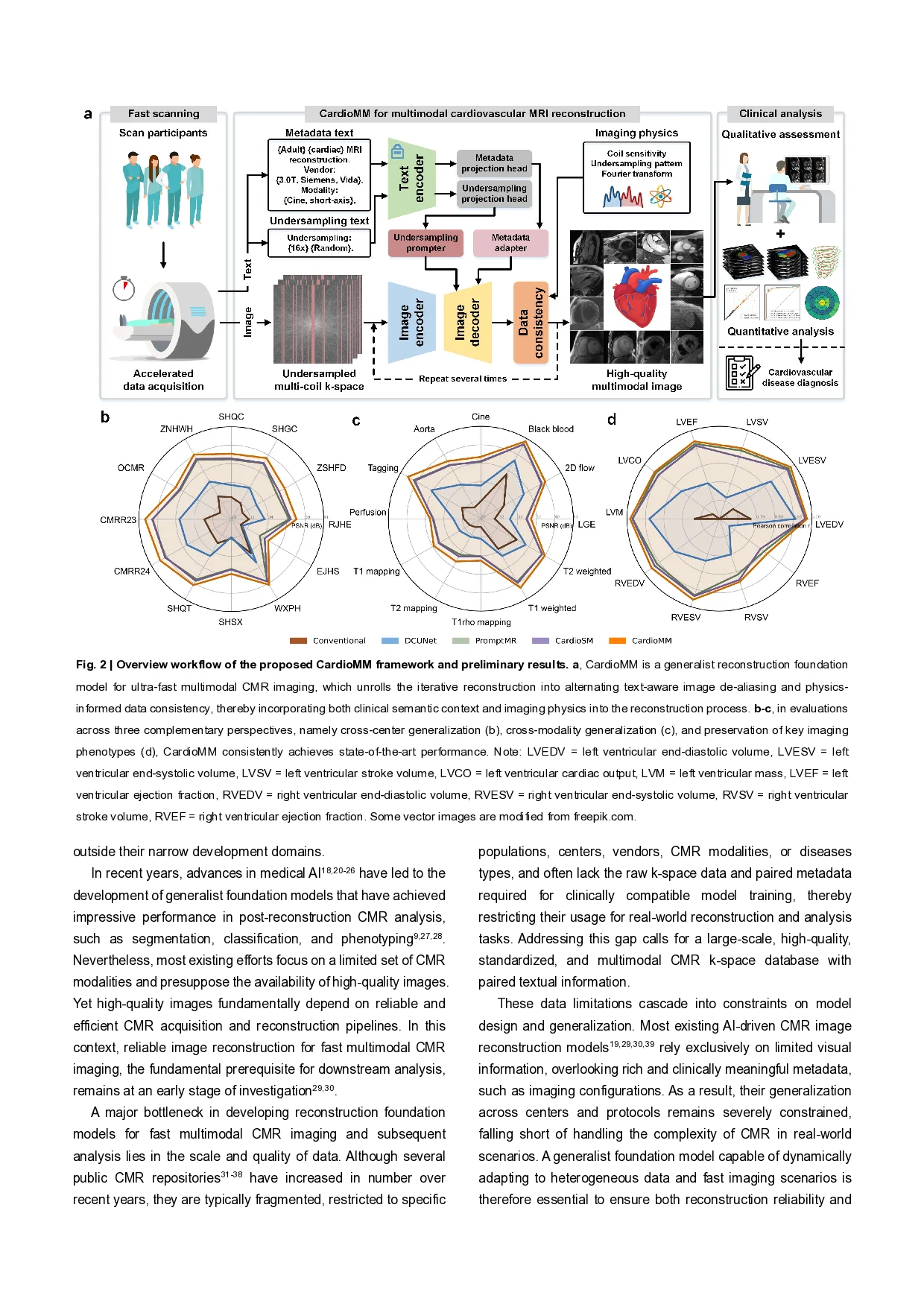
Multimodal cardiovascular magnetic resonance (CMR) imaging provides comprehensive and non-invasive insights into cardiovascular disease (CVD) diagnosis and underlying mechanisms. Despite decades of advancements, its widespread clinical adoption remains constrained by prolonged scan times and heterogeneity across medical environments. This underscores the urgent need for a generalist reconstruction foundation model for ultra-fast CMR imaging-one capable of adapting across diverse imaging scenarios and serving as the essential substrate for all downstream analyses. To enable this goal, we curate MMCMR-427K, the largest and most comprehensive multimodal CMR k-space database to date, comprising 427,465 multi-coil k-space data paired with structured metadata across 13 international centers, 12 CMR modalities, 15 scanners spanning four field strengths, and 17 CVD categories in populations across three continents. Building on this unprecedented resource, we introduce CardioMM, a generalist reconstruction foundation model capable of dynamically adapting to heterogeneous fast CMR imaging scenarios. CardioMM unifies semantic contextual understanding with physics-informed data consistency to deliver robust reconstructions across varied scanners, protocols, and patient presentations. Comprehensive evaluations demonstrate that CardioMM achieves state-of-the-art performance in the internal centers and exhibits strong zero-shot generalization to unseen external settings. Even at imaging acceleration up to 24×, CardioMM reliably preserves key cardiac phenotypes, quantitative myocardial biomarkers, and diagnostic image quality, enabling a substantial increase in CMR examination throughput without compromising clinical integrity. Together, our open-access MMCMR-427K database and CardioMM framework establish a scalable pathway toward high-throughput, high-quality, and clinically accessible multimodal CMR imaging, overcoming the long-standing barriers of slow acquisitions and real-world heterogeneity that have hindered broad clinical adoption of cardiovascular imaging. Cardiovascular diseases (CVDs) remain the leading cause of death worldwide and continue to impose a substantial burden on healthcare systems 1-3 . Multimodal cardiovascular magnetic resonance (CMR) imaging, encompassing diverse imaging💡 Deep Analysis
Deep Dive into Arxiv 2512.21652.Multimodal cardiovascular magnetic resonance (CMR) imaging provides comprehensive and non-invasive insights into cardiovascular disease (CVD) diagnosis and underlying mechanisms. Despite decades of advancements, its widespread clinical adoption remains constrained by prolonged scan times and heterogeneity across medical environments. This underscores the urgent need for a generalist reconstruction foundation model for ultra-fast CMR imaging-one capable of adapting across diverse imaging scenarios and serving as the essential substrate for all downstream analyses. To enable this goal, we curate MMCMR-427K, the largest and most comprehensive multimodal CMR k-space database to date, comprising 427,465 multi-coil k-space data paired with structured metadata across 13 international centers, 12 CMR modalities, 15 scanners spanning four field strengths, and 17 CVD categories in populations across three continents. Building on this unprecedented resource, we introduce CardioMM, a generalist re
📄 Full Content
📸 Image Gallery