📝 Original Info
- Title: q3-MuPa: Quick, Quiet, Quantitative Multi-Parametric MRI using Physics-Informed Diffusion Models
- ArXiv ID: 2512.23726
- Date: 2025-12-19
- Authors: Researchers from original ArXiv paper
📝 Abstract
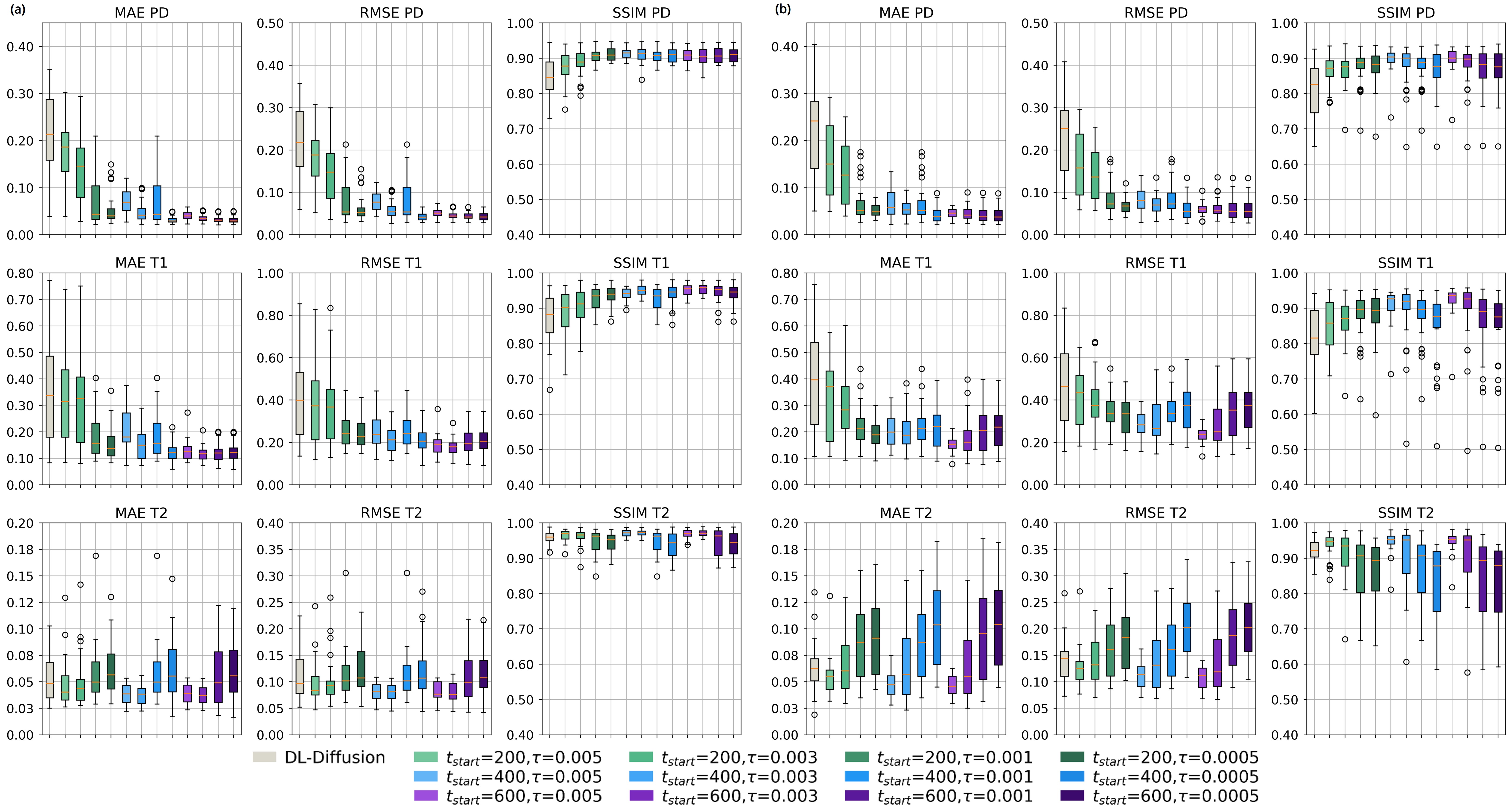
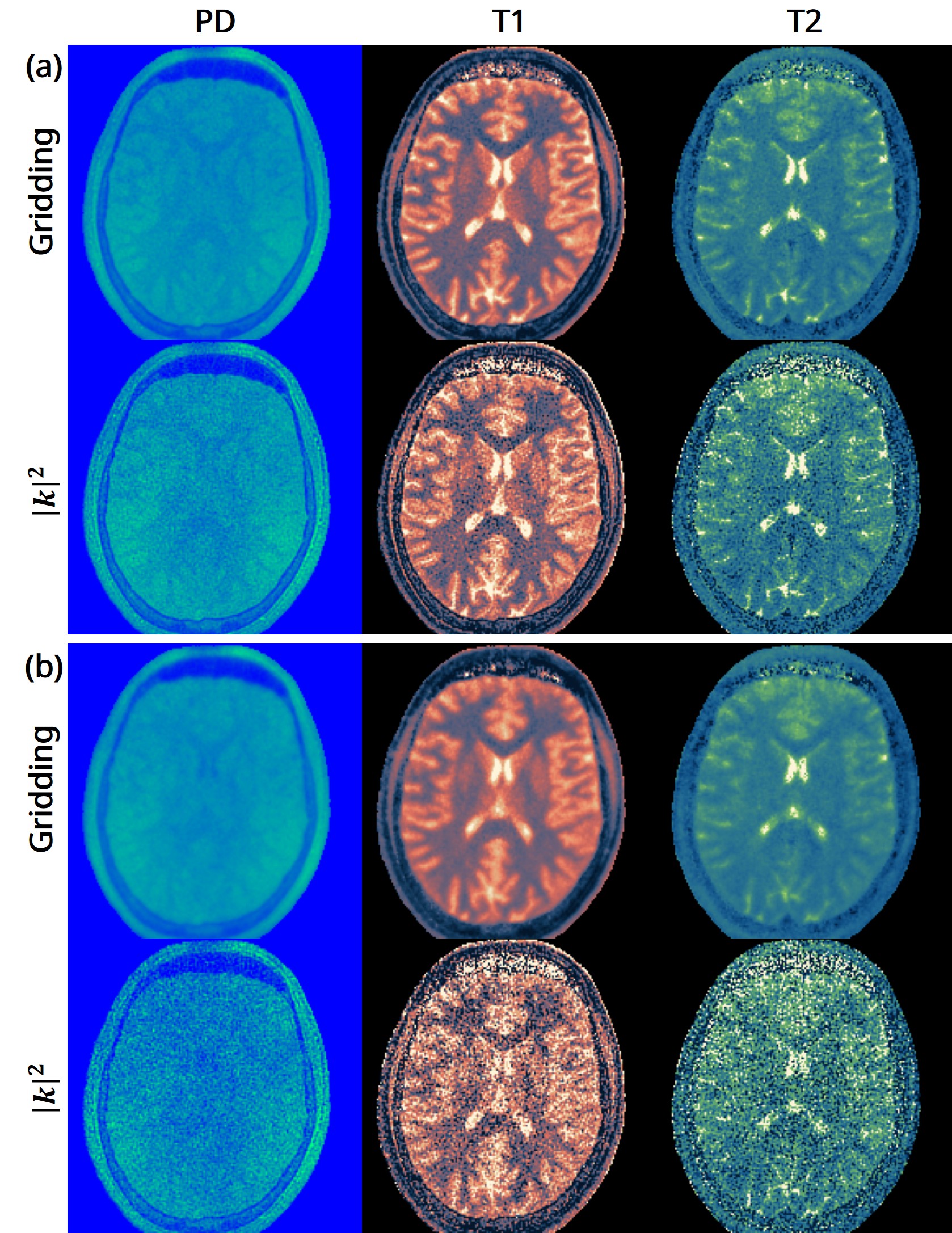
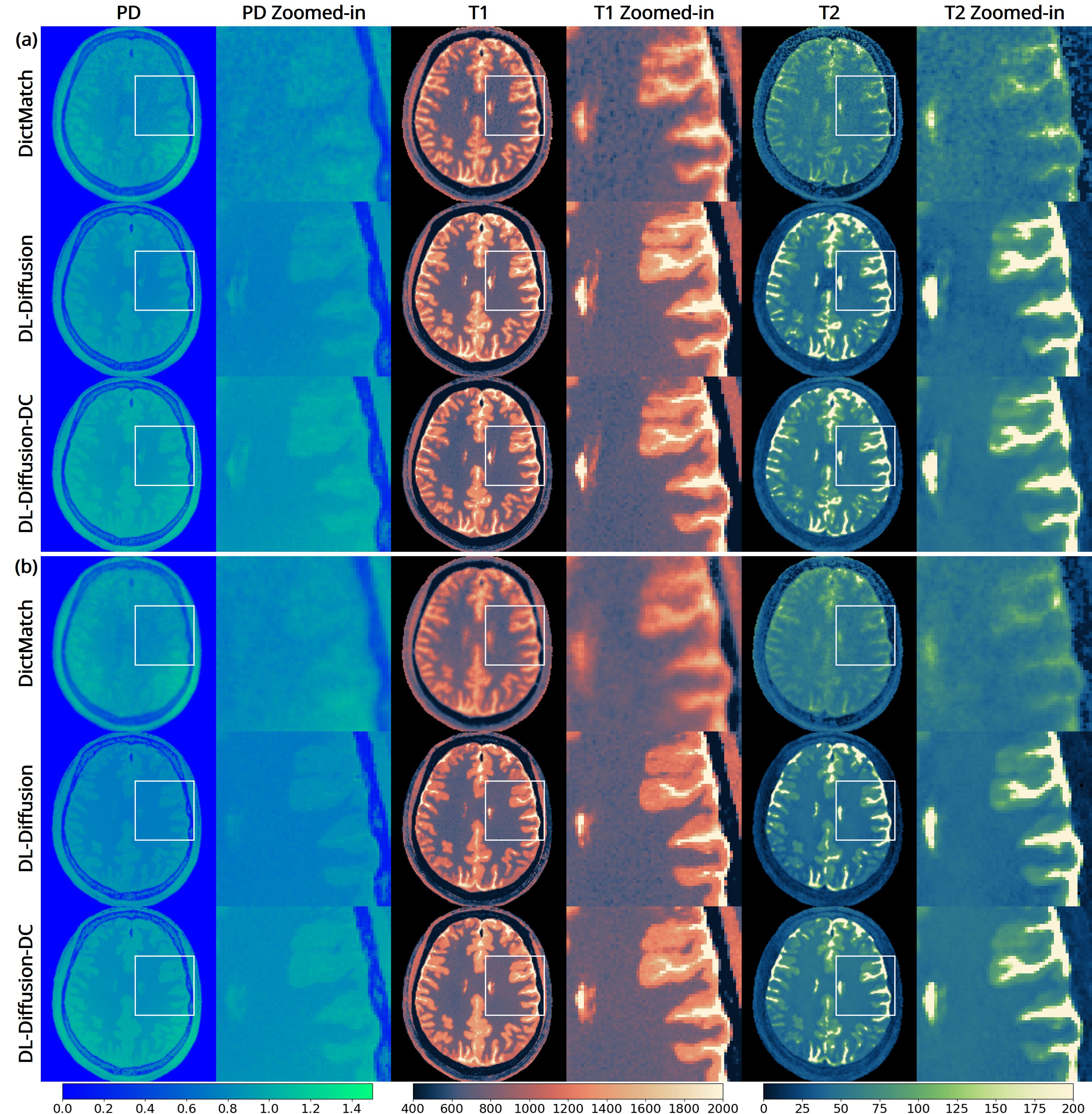
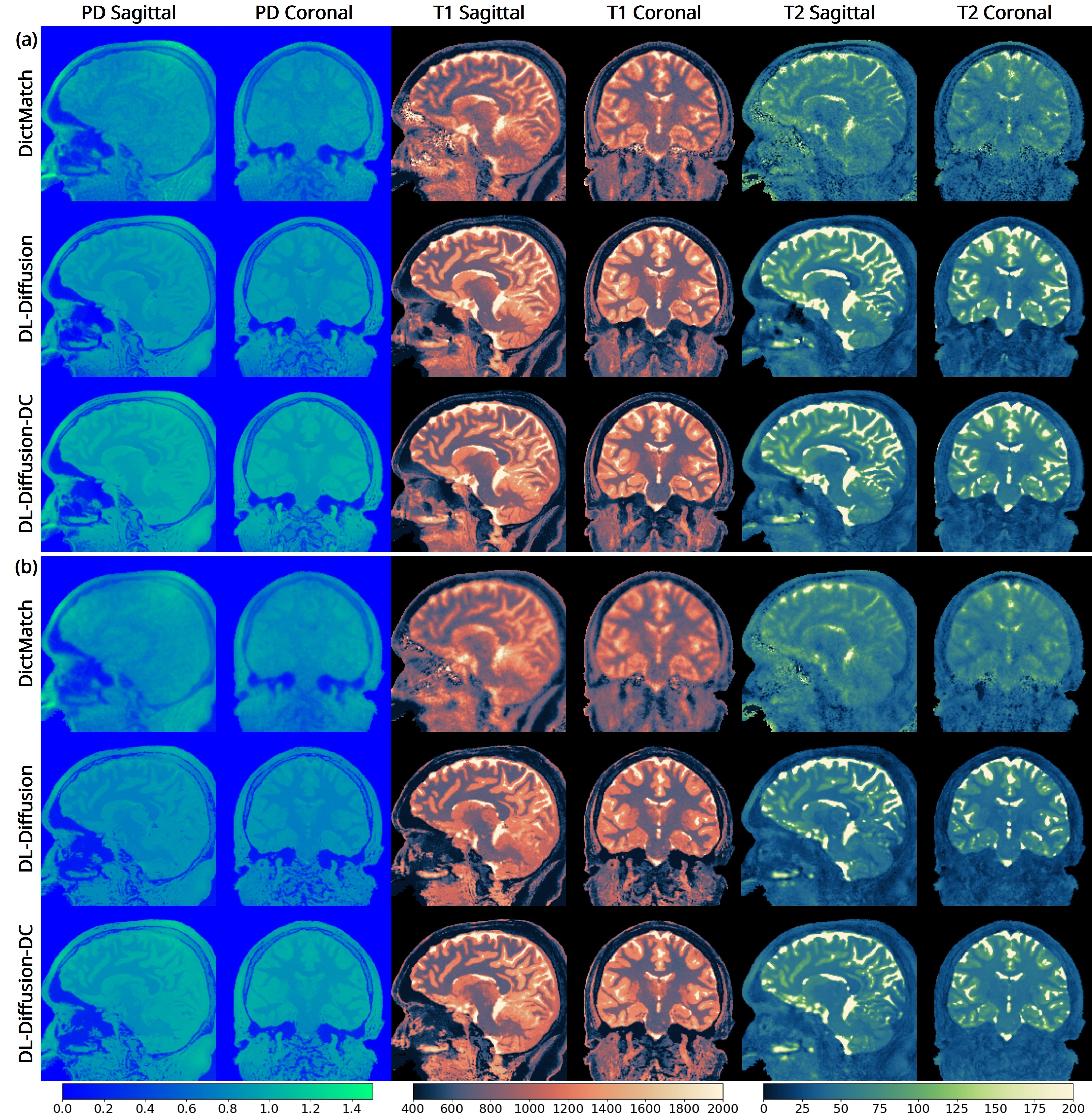
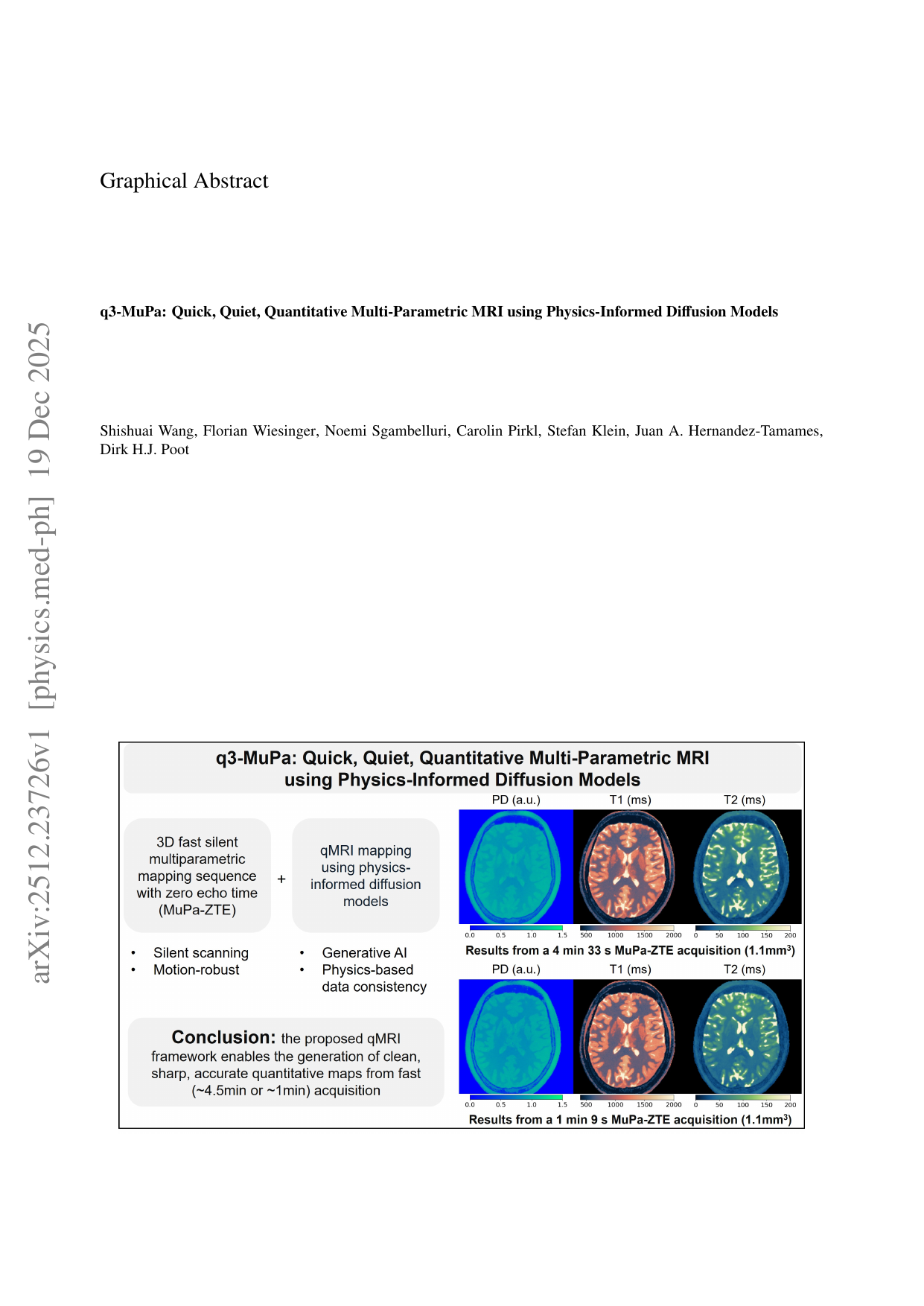
The 3D fast silent multi-parametric mapping sequence with zero echo time (MuPa-ZTE) is a novel quantitative MRI (qMRI) acquisition that enables nearly silent scanning by using a 3D phyllotaxis sampling scheme. MuPa-ZTE improves patient comfort and motion robustness, and generates quantitative maps of T1, T2, and proton density using the acquired weighted image series. In this work, we propose a diffusion model-based qMRI mapping method that leverages both a deep generative model and physics-based data consistency to further improve the mapping performance. Furthermore, our method enables additional acquisition acceleration, allowing high-quality qMRI mapping from a fourfold-accelerated MuPa-ZTE scan (approximately 1 minute). Specifically, we trained a denoising diffusion probabilistic model (DDPM) to map MuPa-ZTE image series to qMRI maps, and we incorporated the MuPa-ZTE forward signal model as an explicit data consistency (DC) constraint during inference. We compared our mapping method against a baseline dictionary matching approach and a purely data-driven diffusion model. The diffusion models were trained entirely on synthetic data generated from digital brain phantoms, eliminating the need for large real-scan datasets. We evaluated on synthetic data, a NISM/ISMRM phantom, healthy volunteers, and a patient with brain metastases. The results demonstrated that our method produces 3D qMRI maps with high accuracy, reduced noise and better preservation of structural details. Notably, it generalised well to real scans despite training on synthetic data alone. The combination of the MuPa-ZTE acquisition and our physics-informed diffusion model is termed q3-MuPa, a quick, quiet, and quantitative multi-parametric mapping framework, and our findings highlight its strong clinical potential.
💡 Deep Analysis
Deep Dive into q3-MuPa: Quick, Quiet, Quantitative Multi-Parametric MRI using Physics-Informed Diffusion Models.
The 3D fast silent multi-parametric mapping sequence with zero echo time (MuPa-ZTE) is a novel quantitative MRI (qMRI) acquisition that enables nearly silent scanning by using a 3D phyllotaxis sampling scheme. MuPa-ZTE improves patient comfort and motion robustness, and generates quantitative maps of T1, T2, and proton density using the acquired weighted image series. In this work, we propose a diffusion model-based qMRI mapping method that leverages both a deep generative model and physics-based data consistency to further improve the mapping performance. Furthermore, our method enables additional acquisition acceleration, allowing high-quality qMRI mapping from a fourfold-accelerated MuPa-ZTE scan (approximately 1 minute). Specifically, we trained a denoising diffusion probabilistic model (DDPM) to map MuPa-ZTE image series to qMRI maps, and we incorporated the MuPa-ZTE forward signal model as an explicit data consistency (DC) constraint during inference. We compared our mapping meth
📄 Full Content
Graphical Abstract
q3-MuPa: Quick, Quiet, Quantitative Multi-Parametric MRI using Physics-Informed Diffusion Models
Shishuai Wang, Florian Wiesinger, Noemi Sgambelluri, Carolin Pirkl, Stefan Klein, Juan A. Hernandez-Tamames,
Dirk H.J. Poot
arXiv:2512.23726v1 [physics.med-ph] 19 Dec 2025
Highlights
q3-MuPa: Quick, Quiet, Quantitative Multi-Parametric MRI using Physics-Informed Diffusion Models
Shishuai Wang, Florian Wiesinger, Noemi Sgambelluri, Carolin Pirkl, Stefan Klein, Juan A. Hernandez-Tamames,
Dirk H.J. Poot
• The proposed qMRI mapping method leverages
diffusion model and physics information.
• We tailored our method for a novel 3D silent qMRI
sequence (MuPa-ZTE)
• Our method is trained on synthetic data and gener-
alises well to real data.
• Our method provides high accuracy and superior
visual performance.
• It is feasible to get high-quality 3D qMRI maps
from approximate 1-minute scan.
q3-MuPa: Quick, Quiet, Quantitative Multi-Parametric MRI using
Physics-Informed Diffusion Models
Shishuai Wanga, Florian Wiesingerb,c, Noemi Sgambelluria, Carolin Pirklb, Stefan Kleina, Juan A.
Hernandez-Tamamesa,d, Dirk H.J. Poota
aDepartment of Radiology and Nuclear Medicine, Erasmus MC, Rotterdam, The Netherlands
bGE HealthCare, Munich, Germany
cDepartment of Neuroimaging, Institute of Psychiatry, Psychology & Neuroscience, King’s College London, London, United Kingdom
dDepartment of Imaging Physics, TU Delft, Delft, The Netherlands
Abstract
The 3D fast silent multi-parametric mapping sequence with zero echo time (MuPa-ZTE) is a novel quantitative MRI
(qMRI) acquisition that enables nearly silent scanning by using a 3D phyllotaxis sampling scheme. MuPa-ZTE im-
proves patient comfort and motion robustness, and generates quantitative maps of T1, T2, and proton density using
the acquired weighted image series. In this work, we propose a diffusion model-based qMRI mapping method that
leverages both a deep generative model and physics-based data consistency to further improve the mapping perfor-
mance. Furthermore, our method enables additional acquisition acceleration, allowing high-quality qMRI mapping
from a fourfold-accelerated MuPa-ZTE scan (approximately 1 minute). Specifically, we trained a denoising diffusion
probabilistic model (DDPM) to map MuPa-ZTE image series to qMRI maps, and we incorporated the MuPa-ZTE
forward signal model as an explicit data consistency (DC) constraint during inference. We compared our mapping
method against a baseline dictionary matching approach and a purely data-driven diffusion model. The diffusion
models were trained entirely on synthetic data generated from digital brain phantoms, eliminating the need for large
real-scan datasets. We evaluated on synthetic data, a NISM/ISMRM phantom, healthy volunteers, and a patient with
brain metastases. The results demonstrated that our method produces 3D qMRI maps with high accuracy, reduced
noise and better preservation of structural details. Notably, it generalised well to real scans despite training on syn-
thetic data alone. The combination of the MuPa-ZTE acquisition and our physics-informed diffusion model is termed
q3-MuPa, a quick, quiet, and quantitative multi-parametric mapping framework, and our findings highlight its strong
clinical potential.
Keywords: Quantitative MRI, Diffusion Models, Multi-parametric Mapping, Deep Generative Models
1. Introduction
Magnetic Resonance Imaging (MRI) is a widely used
non-invasive imaging technique that provides superior
soft-tissue contrast and detailed anatomical or func-
tional information. However, conventional MRI pro-
duces weighted images whose voxel intensities have no
standardised physical meaning, complicating compar-
isons across different scanner settings or sites. In con-
trast, quantitative MRI (qMRI) aims to measure the in-
trinsic tissue properties (e.g. T1 and T2 relaxation times
or proton density), yielding qMRI maps that are com-
parable across scans and promising for use as imaging
biomarkers.
Recent research has focused on 3D fast multi-
parametric mapping sequences [1, 2, 3, 4], aiming to in-
fer multiple relevant tissue properties within clinically
acceptable additional scan time.
However, many 3D
fast qMRI sequences generate loud acoustic noise due
to rapidly switched gradients, which can be problematic
for certain patient groups (e.g. children or individuals
with hyperacusis). In this work, we instead focus on a
novel 3D fast silent multi-parametric mapping sequence
with zero echo time (MuPa-ZTE) [5, 6, 7]. By employ-
ing a nominal zero echo time and a 3D radial phyllotaxis
readout, MuPa-ZTE minimises gradient switching noise
(enabling nearly silent scanning) and is motion-robust,
while also capturing signal from ultrashort T2 compo-
nents normally invisible at longer echo times [6].
The baseline of qMRI mapping for MuPa-ZTE re-
constructs a series of weighted images (with varying
T1, T2, proton density weightings) and then performs
dictionary matching to the underlying
…(Full text truncated)…
📸 Image Gallery
Reference
This content is AI-processed based on ArXiv data.