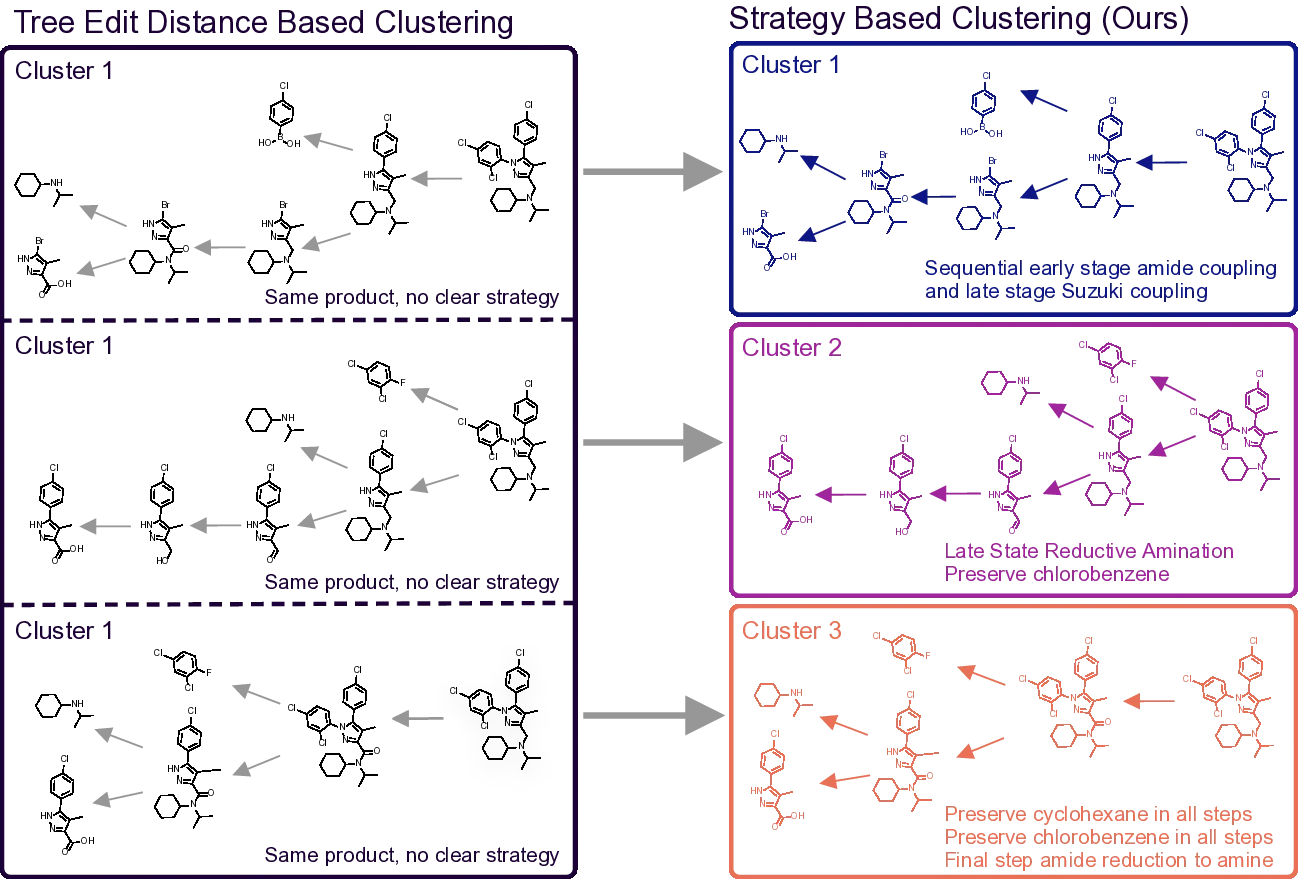
Modern computer-assisted synthesis planning (CASP) systems show promises at generating chemically valid reaction steps but struggle to incorporate strategic considerations such as convergent assembly, protecting group minimization, and optimal ring-forming sequences. We introduce a methodology that leverages Large Language Models to distill synthetic knowledge into code. Our system analyzes synthesis routes and translates strategic principles into Python functions representing diverse strategic and tactical rules, such as strategic functional group interconversions and ring construction strategies. By formalizing this knowledge as verifiable code rather than simple heuristics, we create testable, interpretable representations of synthetic strategy. We release the complete codebase and the USPTO-ST dataset -synthesis routes annotated with strategic tags. This framework unlocks a novel capability for CASP: natural language-based route retrieval, achieving 75% Top-3 accuracy on our benchmark. We further validate our library through temporal analysis of historical trends and chemically intuitive route clustering that offers more granular partitioning than common previous
Deep Dive into 대형 언어 모델을 활용한 합성 전략 코딩 및 CASP 향상.
Modern computer-assisted synthesis planning (CASP) systems show promises at generating chemically valid reaction steps but struggle to incorporate strategic considerations such as convergent assembly, protecting group minimization, and optimal ring-forming sequences. We introduce a methodology that leverages Large Language Models to distill synthetic knowledge into code. Our system analyzes synthesis routes and translates strategic principles into Python functions representing diverse strategic and tactical rules, such as strategic functional group interconversions and ring construction strategies. By formalizing this knowledge as verifiable code rather than simple heuristics, we create testable, interpretable representations of synthetic strategy. We release the complete codebase and the USPTO-ST dataset -synthesis routes annotated with strategic tags. This framework unlocks a novel capability for CASP: natural language-based route retrieval, achieving 75% Top-3 accuracy on our benchm
SynthStrategy: Extracting and Formalizing
Latent Strategic Insights from LLMs in
Organic Chemistry
Daniel Armstrong,† Zlatko Jončev,† Andres Bran,†,‡ and Philippe Schwaller∗,†
†École Polytechnique Fédérale de Lausanne (EPFL)
‡National Centre of Competence in Research (NCCR) Catalysis
E-mail: {daniel.armstrong,philippe.schwaller}@epfl.ch
Abstract
Modern computer-assisted synthesis planning (CASP) systems show promises at
generating chemically valid reaction steps but struggle to incorporate strategic con-
siderations such as convergent assembly, protecting group minimization, and optimal
ring-forming sequences. We introduce a methodology that leverages Large Language
Models to distill synthetic knowledge into code. Our system analyzes synthesis routes
and translates strategic principles into Python functions representing diverse strategic
and tactical rules, such as strategic functional group interconversions and ring construc-
tion strategies. By formalizing this knowledge as verifiable code rather than simple
heuristics, we create testable, interpretable representations of synthetic strategy. We
release the complete codebase and the USPTO-ST dataset – synthesis routes annotated
with strategic tags. This framework unlocks a novel capability for CASP: natural
language-based route retrieval, achieving 75% Top-3 accuracy on our benchmark. We
further validate our library through temporal analysis of historical trends and chemically
intuitive route clustering that offers more granular partitioning than common previous
1
arXiv:2512.01507v1 [cs.AI] 1 Dec 2025
methods. This work bridges the tactical-strategic divide in CASP, enabling specification,
search, and evaluation of routes by strategic criteria rather than structure alone.
Introduction
Computer-assisted synthesis planning (CASP) has evolved from early rule-based systems
to sophisticated machine learning models capable of proposing retrosynthetic disconnec-
tions for complex molecules1–12. These systems can systematically explore vast chemical
spaces to identify pathways connecting target molecules to commercially available starting
materials13–20.
However, a critical gap remains: while these systems typically generate chemically valid
steps, they struggle to evaluate routes based on strategic considerations, such as convergent
assembly, protecting group minimization, and optimal ring-forming sequences1,21. This creates
a needle in the haystack problem. CASP can generate thousands of valid routes but cannot
identify those with sound strategic design, leading to work on route ranking by neural nets
and synthesis cost estimates9,18,22. Recent efforts have addressed this challenge with specific
synthetic constraints, such as reaction class guidance23, disconnection prompts24, bond
constraints25, and starting material constraints26,27. In addition, numerous approaches have
attempted to combine a variety of networks to assign a certainty to how likely a synthesis
pathway is to work based on single-step reaction scores8,13.
Alternative approaches to
multi-step route evaluation include statistical methods, which quantifies plausibility through
template sequence overlap with known pathways28, and composite scoring schemes that
penalize route length while incorporating step confidence and intermediate complexity29.
These approaches focus on individual tactical decisions rather than the holistic, high-level
reasoning that expert chemists employ when designing synthesis routes. In contrast, to
capture more comprehensive strategies, recent work has turned to transformer-based models
that autoregressively generate entire synthesis routes, implicitly incorporating multi-step
2
synthetic strategy30,31. Additionally, Roh et al. 32 reformulate synthesis planning to align
with organic chemistry teaching practice by predicting synthons, enabling the model to focus
on higher-level strategic reasoning.
Over the past years, substantial progress has been made in the capabilities of modern
Large Language Models (LLMs), numerous studies showing this transfers to a multitude of
chemical tasks. Initial demonstrations of chemical knowledge were completed by Jablonka
et al. 33 and Guo et al. 34, while seminal work by Bran et al. 35 and Boiko et al. 36 revealed
the promising performance of GPT-4 in chemical task planning and tool use. Since then, a
significant body of work has built up which leverage LLMs for data extraction, retrosynthesis,
chemical optimisation and a host of other tasks37–43. Recent work has demonstrated that
LLMs possess extensive strategic knowledge, with Bran et al. 44 successfully using LLMs to
re-rank CASP outputs based on strategy-specific prompts. However, this approach requires
computationally expensive real-time inference for each route. Earlier efforts using tree-LSTMs
and similarity metrics9,45–47 similarly focused on post-hoc evaluation rather than extracting
reusable strategic knowledge. This motivates our central question: if LLMs possess strategic
know
…(Full text truncated)…
This content is AI-processed based on ArXiv data.