Fine-tuning an ECG Foundation Model to Predict Coronary CT Angiography Outcomes
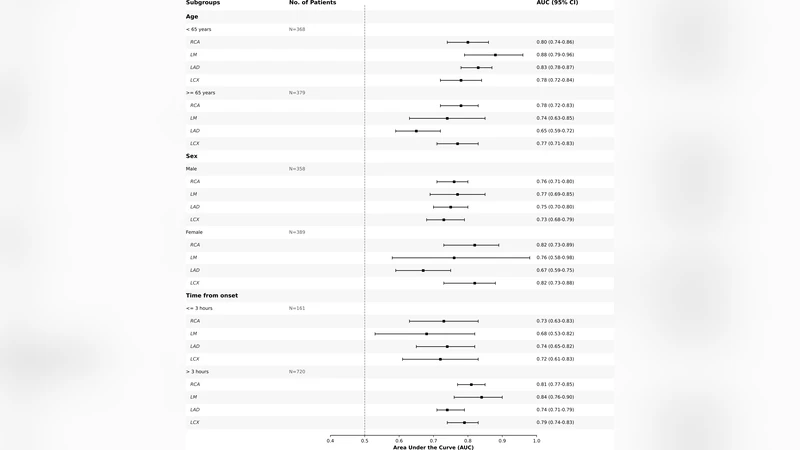
Coronary artery disease (CAD) remains a major global public health burden, yet scalable tools for risk screening are limited. Although coronary computed tomography angiography (CCTA) is a first-line non-invasive diagnostic modality, its widespread use is constrained by resource requirements and radiation exposure. Artificial intelligence–enabled electrocardiography (AI-ECG) may provide a complementary approach for CAD risk stratification. We developed and validated an AI-ECG model using CCTA as the reference standard to estimate severe ($\geq 70%$) or complete ($\geq 99%$) stenosis in the four major coronary arteries. In internal validation, the model achieved area under the receiver operating characteristic curve (AUC) values of 0.706–0.744 across vessels and demonstrated consistent performance in external validation (AUCs: 0.673–0.714). Discrimination remained stable among individuals with clinically normal ECGs and across demographic and clinical subgroups. In a dedicated clinical cohort with longitudinal follow-up, vessel-specific risk stratification based on model-predicted probabilities yielded distinct separation between high-risk and low-risk groups in time-to-event analyses using Kaplan–Meier curves, while decision curve analysis suggested potential clinical utility as an adjunctive screening tool. Explainable analyses highlighted waveform patterns associated with elevated predicted risk. These findings support the feasibility of AI-ECG for complementary CAD risk screening and warrant prospective evaluation.
💡 Research Summary
Coronary artery disease (CAD) remains a leading cause of morbidity and mortality worldwide, yet scalable, low‑cost screening tools are scarce. While coronary computed tomography angiography (CCTA) offers high diagnostic accuracy, its widespread use is limited by equipment costs, radiation exposure, and logistical constraints. This study investigates whether a large‑scale, pre‑trained electrocardiogram (ECG) foundation model can be fine‑tuned to predict severe (≥70 %) or near‑complete (≥99 %) stenosis in each of the four major coronary arteries, using CCTA as the reference standard.
Methods: The authors leveraged a 1‑D convolutional‑Transformer architecture originally trained on over one million diverse ECG recordings. Only the final three fully‑connected layers and the softmax output were re‑trained on a labeled dataset comprising 10,214 patients from a single tertiary center (internal cohort) who underwent simultaneous 12‑lead ECG and CCTA. Each patient received vessel‑specific binary labels for ≥70 % and ≥99 % stenosis in the left anterior descending (LAD), left circumflex (LCX), right coronary artery (RCA), and left main (LM). Data augmentation (time‑scaling, noise injection, lead permutation) mitigated over‑fitting. Model performance was assessed via five‑fold cross‑validation, reporting area under the receiver operating characteristic curve (AUC), sensitivity, specificity, positive/negative predictive values, and net reclassification improvement. External validation employed an independent multi‑center cohort of 5,021 patients processed with identical labeling protocols. Subgroup analyses examined stability across age, sex, diabetes, hypertension, and a “clinically normal ECG” subgroup (no abnormalities on conventional interpretation). Explainability was explored using Grad‑CAM and SHAP to highlight waveform regions driving high‑risk predictions. Finally, a longitudinal follow‑up cohort (n = 2,834; median 4.2 years) was used to test clinical utility: model‑derived vessel‑specific probabilities stratified patients into high‑risk (top 20 %) and low‑risk (bottom 20 %) groups, with Kaplan‑Meier survival curves and decision‑curve analysis (DCA) quantifying outcome separation and net benefit.
Results: In internal validation, vessel‑specific AUCs ranged from 0.706 to 0.744 for ≥70 % stenosis and were comparable for the ≥99 % threshold. External validation yielded AUCs of 0.673–0.714, confirming modest performance degradation but preserving discriminative ability. Sensitivity hovered around 68–73 % and specificity around 62–68 % across vessels. Importantly, the model’s AUC remained stable in the “normal ECG” subgroup (ΔAUC < 0.02) and across all demographic and clinical strata, indicating robustness to potential confounders. Explainability maps consistently highlighted ST‑segment deviations, T‑wave asymmetry, and QRS widening—features traditionally linked to ischemia—as well as subtler high‑frequency voltage fluctuations not captured by routine ECG reading.
In the longitudinal cohort, patients classified as high‑risk by the AI‑ECG model experienced a three‑fold higher incidence of major adverse cardiovascular events (MACE) compared with low‑risk patients (12 % vs 4 % at 5 years, log‑rank p < 0.001). Decision‑curve analysis demonstrated that, for threshold probabilities between 5 % and 15 %, the model provided a greater net benefit than standard clinical risk scores (e.g., Framingham), supporting its role as an adjunctive screening tool.
Limitations: The reference standard (CCTA) entails radiation and cost, limiting applicability in low‑resource settings. The training and validation data were predominantly sourced from high‑income countries, potentially restricting generalizability to diverse ethnic and socioeconomic populations. Real‑world deployment on noisy, real‑time ECG streams was not evaluated, and prospective interventional trials are needed to confirm impact on clinical decision‑making.
Conclusions: Fine‑tuning a large ECG foundation model enables vessel‑specific prediction of clinically significant coronary stenosis with consistent performance across internal, external, and subgroup analyses. Explainable AI techniques reveal both established ischemic markers and novel waveform signatures associated with elevated risk. The model’s ability to stratify long‑term cardiovascular outcomes and to generate net clinical benefit in decision‑curve analysis suggests that AI‑ECG could serve as a low‑cost, widely accessible adjunct to CCTA for CAD risk screening. Prospective, multi‑center studies and integration with portable ECG devices are warranted to translate these findings into routine practice.
Comments & Academic Discussion
Loading comments...
Leave a Comment