Modeling the respiratory Central Pattern Generator with resonate-and-fire Izhikevich-Neurons
Computational models of the respiratory central pattern generator (rCPG) are usually based on biologically-plausible Hodgkin Huxley neuron models. Such models require numerous parameters and thus are prone to overfitting. The HH approach is motivated by the assumption that the biophysical properties of neurons determine the network dynamics. Here, we implement the rCPG using simpler Izhikevich resonate-and-fire neurons. Our rCPG model generates a 3-phase respiratory motor pattern based on established connectivities and can reproduce previous experimental and theoretical observations. Further, we demonstrate the flexibility of the model by testing whether intrinsic bursting properties are necessary for rhythmogenesis. Our simulations demonstrate that replacing predicted mandatory bursting properties of pre-inspiratory neurons with spike adapting properties yields a model that generates comparable respiratory activity patterns. The latter supports our view that the importance of the exact modeling parameters of specific respiratory neurons is overestimated.
💡 Research Summary
The respiratory central pattern generator (rCPG) is a brain‑stem circuit that produces the rhythmic motor pattern underlying breathing. Traditional computational models of the rCPG have relied almost exclusively on Hodgkin‑Huxley (HH) type neurons because HH models capture the detailed biophysics of ion channels and are therefore considered biologically realistic. However, HH models require dozens of parameters, are computationally expensive, and are prone to over‑fitting when calibrated against limited experimental data. The present study asks whether such detailed biophysical fidelity is truly necessary for reproducing the essential dynamics of the rCPG.
To address this question, the authors replace HH neurons with Izhikevich resonate‑and‑fire (R‑F) units. An Izhikevich neuron is defined by two differential equations for membrane potential (v) and a recovery variable (u) and a small set of parameters (a, b, c, d). The “resonate” extension adds a subthreshold oscillatory component that can be tuned to preferentially respond to a particular frequency band, making it well suited for generating rhythmic activity. Importantly, the R‑F formulation can emulate both bursting and spike‑frequency adaptation by simple adjustments of a and b, allowing the same mathematical framework to represent diverse neuronal phenotypes with far fewer parameters than an HH model.
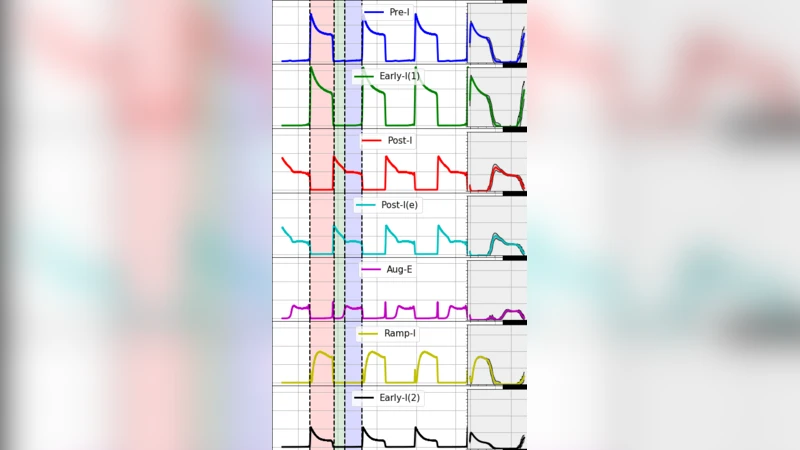
The network architecture mirrors that of well‑established rCPG models: distinct neuronal populations (pre‑inspiratory, early‑inspiratory, post‑inspiratory, augmenting expiratory, etc.) are connected by excitatory and inhibitory synapses according to experimentally derived connectivity matrices. In the canonical configuration, pre‑inspiratory (Pre‑I) neurons are modeled as intrinsic bursters, a feature that has been argued to be essential for initiating the inspiratory phase. The authors test this assumption by constructing two versions of the model. In the first, Pre‑I neurons retain their bursting parameters (a≈0.02, b≈0.2, c≈‑55 mV, d≈2). In the second, the same cells are given spike‑adapting parameters (a≈0.1, b≈0.2, c≈‑65 mV, d≈8), eliminating intrinsic bursting while preserving the ability to fire a transient high‑frequency burst at the onset of each cycle.
Simulation results show that both configurations generate a robust three‑phase respiratory pattern (inspiration, post‑inspiratory pause, expiration) that matches key temporal features reported in physiological recordings. The burst‑based Pre‑I model reproduces the classic high‑frequency burst that triggers the inspiratory population, whereas the adapting Pre‑I model produces a brief high‑frequency train that quickly adapts, yet still succeeds in recruiting the inspiratory ensemble through the same excitatory pathways. Quantitative analysis of phase durations, motor output amplitudes, and inter‑burst intervals reveals no statistically significant differences between the two versions, indicating that intrinsic bursting is not a strict prerequisite for rhythmogenesis in this network.
A systematic sensitivity analysis further demonstrates that the overall rhythm is far more sensitive to the strength of inhibitory connections and the balance of excitation versus inhibition than to the precise values of a, b, c, or d for any single neuron type. Small reductions in inhibitory conductance lengthen the expiratory phase and can even destabilize the three‑phase pattern, mimicking pathological conditions such as hypoventilation. Conversely, moderate variations in the adaptation parameters of Pre‑I neurons produce only minor changes in burst frequency and amplitude, underscoring the robustness of the network architecture.
The authors argue that these findings shift the focus from detailed ion‑channel kinetics to the topology of synaptic connectivity when modeling central pattern generators. By using a compact R‑F neuron model, they achieve a dramatic reduction in computational load (simulation times are an order of magnitude faster than comparable HH models) while preserving the ability to explore large parameter spaces, perform extensive bifurcation analyses, and integrate additional physiological inputs such as chemosensory drive (CO₂/pH) or descending cortical modulation.
In the discussion, the paper highlights several implications. First, the flexibility of the Izhikevich framework makes it straightforward to incorporate sensory feedback loops, allowing future models to simulate how changes in blood gases or lung stretch receptors reshape the respiratory rhythm. Second, because the model relies on a small set of interpretable parameters, it is well suited for data‑driven fitting to patient‑specific recordings, opening avenues for personalized simulation of breathing disorders. Third, the demonstration that intrinsic bursting is not essential challenges a long‑standing assumption in the field and suggests that experimental efforts should prioritize mapping synaptic connectivity and neuromodulatory influences over exhaustive characterization of single‑cell electrophysiology.
In conclusion, this study presents a parsimonious yet powerful computational model of the rCPG based on Izhikevich resonate‑and‑fire neurons. The model faithfully reproduces the canonical three‑phase respiratory pattern, remains stable under substantial variations of neuronal intrinsic properties, and highlights the dominant role of network connectivity in rhythm generation. By reducing the number of free parameters and computational complexity, the approach offers a scalable platform for investigating normal and pathological breathing, testing pharmacological interventions, and ultimately bridging the gap between detailed biophysical modeling and functional systems neuroscience.
Comments & Academic Discussion
Loading comments...
Leave a Comment