Numerical investigation of Differential Biological Models via RBF collocation Method with Genetic Strategy
In this paper, we use Kansa method for solving the system of differential equations in the area of biology. One of the challenges in Kansa method is picking out an optimum value for Shape parameter in Radial Basis Function to achieve the best result of the method because there are not any available analytical approaches for obtaining optimum Shape parameter. For this reason, we design a genetic algorithm to detect a close optimum Shape parameter. The experimental results show that this strategy is efficient in the systems of differential models in biology such as HIV and Influenza. Furthermore, we prove that using Pseudo-Combination formula for crossover in genetic strategy leads to convergence in the nearly best selection of Shape parameter.
💡 Research Summary
The paper addresses the longstanding difficulty of selecting an optimal shape parameter (c) in radial basis function (RBF) collocation methods, specifically the Kansa approach, for solving systems of ordinary differential equations that arise in biological modeling. Two representative models are examined: an HIV infection model describing the dynamics of susceptible CD4⁺ T cells, infected cells, and free virus particles, and an influenza SIRC (Susceptible‑Infected‑Recovered‑Cross‑immune) model. Both models lack closed‑form solutions, making high‑accuracy numerical methods essential.
The authors first outline the RBF collocation framework. By representing each unknown function as a linear combination of radial basis functions ψ(‖t−t_i‖) with coefficients ξ_i, the differential equations are transformed into a set of algebraic residual equations. The shape parameter c controls the width of ψ and directly influences both the approximation accuracy and the conditioning of the resulting matrix. Existing strategies for choosing c (e.g., empirical formulas, LOOCV, PRESS) are either problem‑specific or computationally expensive, and no universal analytical rule exists.
To automate the selection of a near‑optimal c, the authors develop a continuous genetic algorithm (CGA). An initial population of candidate c values is generated uniformly within a user‑defined interval. The fitness of each individual is evaluated using one of two problem‑specific metrics: (i) the average 2‑norm of the residuals (ASN2R) for the HIV model, or (ii) the average relative error (ARE) compared with a reference solution for the influenza model. The fitness function is defined as exp(1/(1+Θ)), turning a minimization problem into a maximization one.
Selection is performed via roulette‑wheel sampling, giving higher‑fitness chromosomes a larger probability of becoming parents. The key novelty lies in the crossover operator, termed “Pseudo‑Combination Formula” (PCF). For two parents a and b with fitness values f(a) and f(b), the offspring d is computed as:
d = a + b/2 + sign(b−a)·(|b−a + b/2 + sign(b−a)·ε|) / |f(a)|,
where ε is a small random perturbation. This formula simultaneously exploits the numerical values of the parents and their fitness, encouraging larger jumps when parents differ significantly while biasing the offspring toward the fitter parent. Mutation adds a Gaussian noise term to maintain diversity.
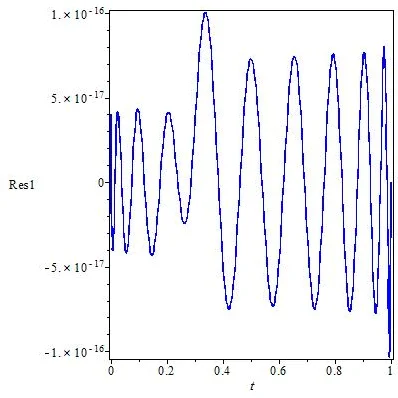
The CGA runs for a preset number of generations (e.g., 100) with a population size of 50, crossover probability 0.8, and mutation probability 0.1. After each generation, the current best c is used in the RBF collocation to solve the algebraic system via Newton‑Raphson, and the fitness is recomputed. The process converges to a c that yields minimal residuals or relative errors.
Experimental results are presented for both models using 30–50 equally spaced collocation points in the interval
Comments & Academic Discussion
Loading comments...
Leave a Comment