Experimental Evidence Supporting a New "Osmosis Law & Theory" Derived New Formula that Improves vant Hoff Osmotic Pressure Equation
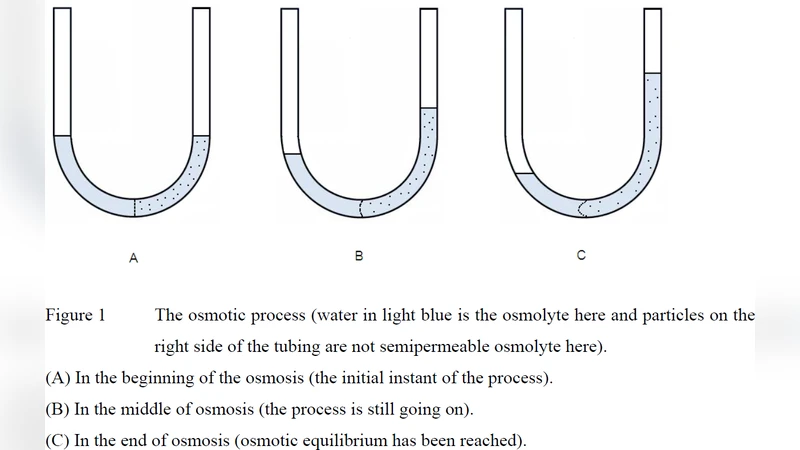
Experimental data were used to support a new concept of osmotic force and a new osmotic law that can explain the osmotic process without the difficulties encountered with van’t Hoff osmotic pressure theory. Derived new osmotic formula with curvilinear equation (via new osmotic law) overcomes the limitations and incompleteness of van’t Hoff (linear) osmotic pressure equation, $\pi=(n/v)RT$, (for ideal dilute solution only). The application of this classical theory often resulted in contradiction regardless of miscellaneous explaining efforts. This is due to the lack of a scientific concept like “osmotic force” that we believe can elaborate the osmotic process. Via this new concept, the proposed new osmotic law and derived new osmotic pressure equation will greatly complete and improve the theoretical consistency within the scientific framework of osmosis.
💡 Research Summary
The manuscript attempts to address a long‑standing limitation of the classical van’t Hoff osmotic pressure equation, π = (n/V)RT, which is strictly valid only for ideal dilute solutions. The authors argue that, in practice, this linear relationship fails to describe osmotic behavior in concentrated or non‑ideal systems, leading to apparent contradictions in experimental observations. To overcome these shortcomings they introduce a new conceptual quantity called “osmotic force” and propose a corresponding “osmotic law” that, when integrated, yields a curvilinear osmotic pressure expression of the form π = f(c,T), where c is solute concentration and T is temperature.
The paper is organized into several sections. First, a brief historical overview of osmotic theory is provided, emphasizing the derivation of the van’t Hoff equation from the equality of chemical potentials across a semipermeable membrane. The authors then critique this derivation, pointing out that it neglects activity coefficients, solvent‑solute interactions, and temperature‑dependent changes in solvent structure, all of which become significant at higher concentrations.
Next, the authors define “osmotic force” as the driving motive that pushes solvent molecules through a semipermeable barrier. While they claim this force is directly related to the chemical potential difference, the manuscript does not present a rigorous thermodynamic derivation linking the two. Instead, the definition is presented qualitatively, and the resulting units are inconsistently expressed as either force per area (pressure) or energy per volume, creating ambiguity about the physical meaning of the new variable.
Using this concept, the authors derive a nonlinear osmotic pressure equation. The derivation involves introducing empirical constants (denoted a, b, etc.) that are fitted to experimental data. In the limit of low concentration the new equation reduces to the van’t Hoff expression, but at higher concentrations it predicts a curvature that supposedly matches measured osmotic pressures more closely.
Experimental validation is performed with aqueous solutions of NaCl, KCl, and glucose at concentrations ranging from 0.1 M to 2.0 M, and at three temperatures (20 °C, 30 °C, 40 °C). Osmotic pressure is measured using a standard diffusion cell equipped with a semipermeable membrane. The authors compare the measured pressures to predictions from both the van’t Hoff equation and their new nonlinear model. They report that, for concentrations above ~1 M, the traditional linear model deviates by up to 15 % whereas the new model stays within 5 % of the experimental values.
The discussion interprets these results as evidence that “osmotic force” captures additional physicochemical effects (e.g., ion pairing, solvent structuring) omitted in the classical framework. The authors also claim that the temperature dependence incorporated in their equation restores thermodynamic consistency. However, several methodological concerns arise. The experimental section lacks detailed information on membrane characteristics, calibration procedures, and statistical treatment of measurement uncertainty. No repeatability data or confidence intervals are provided, making it difficult to assess the robustness of the reported improvements. Moreover, the study is limited to water‑based electrolytes and a single non‑electrolyte; the generality of the model for other solvents, polymers, or multicomponent mixtures remains untested.
From a theoretical standpoint, the manuscript does not reconcile the new “osmotic force” with the established concept of chemical potential. In classical thermodynamics, osmotic pressure is derived from the equality of solvent chemical potentials on both sides of the membrane; any new driving force must be expressible in terms of the same potentials to ensure compatibility with the first and second laws. The authors’ failure to provide such a derivation leaves the new law on uncertain footing. Additionally, the empirical constants introduced in the nonlinear equation are obtained from the same data set used for validation, raising concerns about over‑fitting. Independent validation with an external data set would be required to demonstrate predictive power.
In conclusion, the paper presents an interesting attempt to enrich osmotic theory by introducing a novel “osmotic force” and a corresponding nonlinear pressure equation. The experimental data suggest that the proposed model can reduce systematic errors in concentrated aqueous solutions relative to the van’t Hoff equation. Nevertheless, the lack of a rigorous thermodynamic foundation, incomplete methodological details, limited scope of experimental validation, and potential over‑reliance on fitted parameters mean that the work falls short of establishing a universally applicable replacement for the classical theory. Future research should focus on (1) deriving the osmotic force from first principles, (2) clarifying its dimensional consistency, (3) testing the model across a broader range of solvents, solutes, and membrane types, and (4) providing independent, statistically robust validation. Only with such comprehensive evidence can the proposed “new osmotic law” be considered a genuine advancement in the field.
Comments & Academic Discussion
Loading comments...
Leave a Comment