Energy landscape analysis of cardiac fibrillation wave dynamics using pairwise maximum entropy model
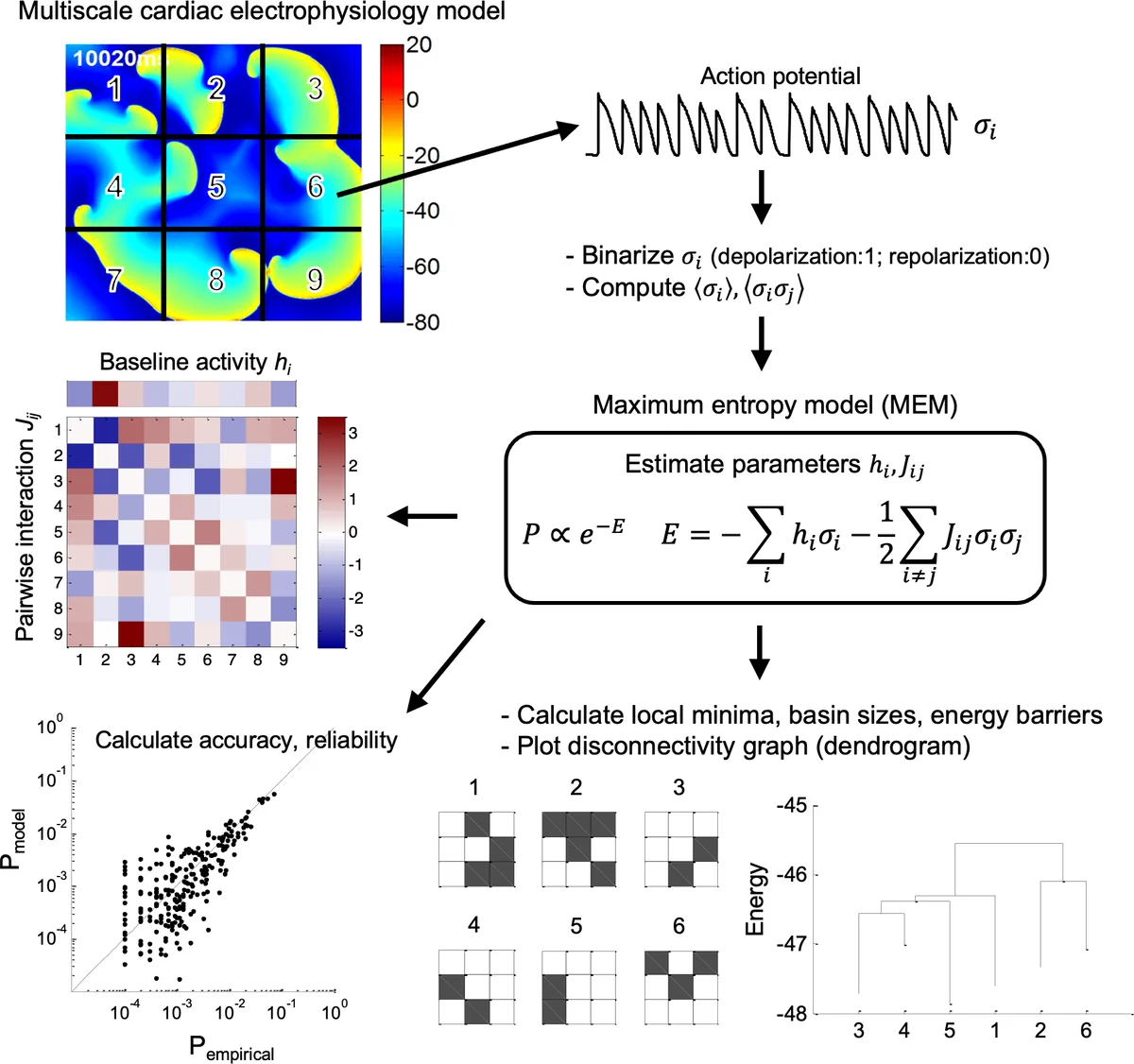
Cardiac fibrillation is characterized by chaotic and disintegrated spiral wave dynamics patterns, whereas sinus rhythm shows synchronized excitation patterns. To determine functional correlations among cardiomyocytes during complex fibrillation states, we applied a pairwise maximum entropy model (MEM) to the 2D numerical simulation data of human atrial fibrillation. We then constructed an energy landscape and estimated a hierarchical structure among the different local minima (attractors) to explain the dynamic properties of cardiac fibrillation. The MEM could describe the wave dynamics of sinus rhythm, single stable rotor, and single rotor with wavebreak (both accuracy and reliability>0.9), but not the multiple random wavelet case. The energy landscapes exhibited unique profiles of local minima and energy barriers, characterizing the spatiotemporal patterns of cardiac fibrillation dynamics. The pairwise MEM-based energy landscape analysis provides reliable explanations of complex nonlinear dynamics of cardiac fibrillation, which might be determined by the presence of a ‘driver’ such as a sinus node or rotor.
💡 Research Summary
This study introduces a novel application of the pairwise maximum entropy model (MEM) to characterize the complex spatiotemporal dynamics of atrial fibrillation (AF) using two‑dimensional numerical simulations of human atrial tissue. The authors first generate four canonical wave patterns—normal sinus rhythm, a single stable rotor, a rotor with wavebreak, and a multiple‑wavelet state—by perturbing ionic conductances in a Courtemanche‑Ramirez‑Nattel cellular model implemented on a 512 × 512 isotropic grid. Electrical activity is sampled at 1 ms intervals, and nine representative sites are binarized to form a nine‑dimensional state vector for each time point.
The MEM framework seeks a probability distribution over the 2⁹ possible binary configurations that maximizes entropy while reproducing the empirically observed mean activation rates and pairwise correlations. Assuming an Ising‑type Hamiltonian E = −∑ h_iσ_i − ½∑ J_ijσ_iσ_j, the authors iteratively update the fields h_i and couplings J_ij using a scaling rule with learning rate α = 0.1. Model fit is quantified by Kullback‑Leibler divergence–based accuracy and an entropy‑based reliability metric.
Results show that the MEM accurately captures the sinus rhythm, stable rotor, and rotor‑with‑wavebreak cases, achieving accuracy > 0.90 and reliability > 0.99. In contrast, the multiple‑wavelet scenario yields a markedly lower accuracy of 0.508, indicating that pairwise correlations alone are insufficient to describe the high‑order, chaotic interactions characteristic of this state.
Using the fitted Hamiltonian, the authors construct an energy landscape and identify local minima (attractors), their basin sizes, and the energy barriers separating them. Sinus rhythm and the stable rotor exhibit few deep minima with large basins and high barriers, reflecting strong “driver” dominance (sinus node or rotor) that stabilizes the system. The rotor‑with‑wavebreak case shows a greater number of minima and reduced barriers, consistent with intermittent breakup and re‑formation of spiral waves. The multiple‑wavelet landscape is populated by hundreds of shallow minima separated by low barriers, illustrating a highly unstable, rapidly transitioning dynamical regime.
The discussion links these findings to analogous energy‑landscape analyses in neuroscience, where MEM has successfully described resting‑state brain networks. The authors argue that, like the brain, cardiac tissue’s functional connectivity extends beyond anatomical coupling and can be inferred from pairwise statistics. However, they acknowledge limitations: the current model ignores higher‑order interactions, anisotropic conduction, and structural heterogeneity. They propose future extensions incorporating higher‑order MEM, non‑Ising Hamiltonians, and realistic 3‑D atrial geometries, as well as application to clinical intracardiac electrogram datasets for driver localization and targeted ablation.
In conclusion, the pairwise MEM provides a powerful statistical‑physics framework for dissecting AF dynamics, revealing that the presence of a dominant driver (sinus node or rotor) shapes the energy landscape and thereby the stability of cardiac wave patterns. This approach offers a quantitative basis for understanding AF mechanisms and may inform more precise therapeutic interventions.
Comments & Academic Discussion
Loading comments...
Leave a Comment