Balance of microtubule stiffness and cortical tension determines the size of blood cells with marginal band across species
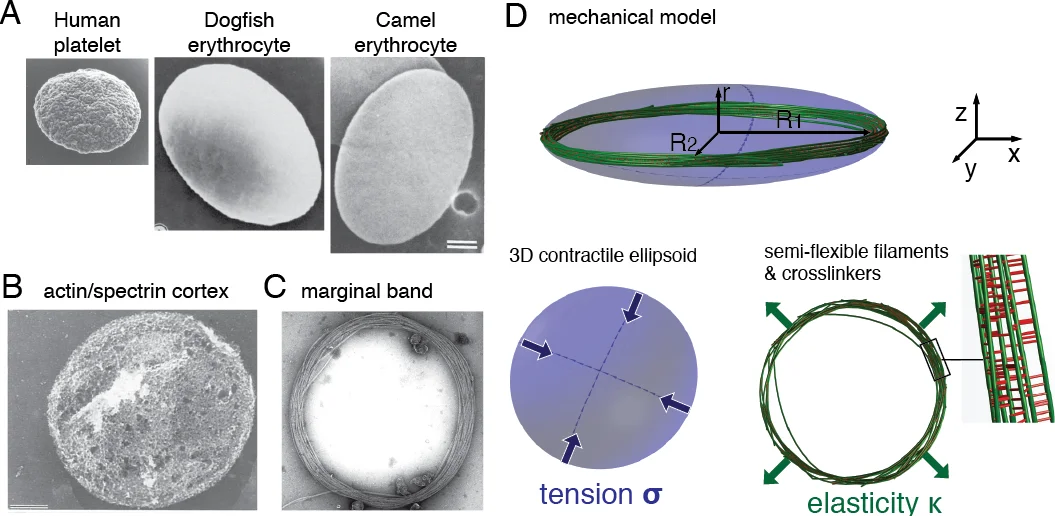
The fast blood stream of animals is associated with large shear stresses. Consequently, blood cells have evolved a special morphology and a specific internal architecture allowing them to maintain their integrity over several weeks. For instance, non-mammalian red blood cells, mammalian erythroblasts and platelets have a peripheral ring of microtubules, called the marginal band, that flattens the overall cell morphology by pushing on the cell cortex. In this article, we model how the shape of these cells stems from the balance between marginal band elasticity and cortical tension. We predict that the diameter of the cell scales with the total microtubule polymer, and verify the predicted law across a wide range of species. Our analysis also shows that the combination of the marginal band rigidity and cortical tension increases the ability of the cell to withstand forces without deformation. Finally, we model the marginal band coiling that occurs during the disc-to-sphere transition observed for instance at the onset of blood platelet activation. We show that when cortical tension increases faster than crosslinkers can unbind, the marginal band will coil, whereas if the tension increases slower, the marginal band may shorten as microtubules slide relative to each other.
💡 Research Summary
The manuscript investigates why circulating blood cells—particularly non‑mammalian red blood cells, mammalian platelets, and erythroblasts—adopt a flattened, disc‑like shape despite being exposed to high shear stresses. The authors focus on the marginal band (MB), a peripheral ring of bundled microtubules (MTs), and the cell cortex, a thin contractile surface that exerts a surface tension σ. They propose that the equilibrium shape results from a simple mechanical balance between the bending rigidity κ of the MTs and the cortical tension.
Theoretical framework
Treating the MB as a closed elastic ring of total length L, the bending energy is EB = κ L/(2R²), where R is the major cell radius (approximated as the MB radius). The cortex contributes a surface energy ET = 2πσR². Minimizing the total energy with respect to R yields the scaling law 4πR⁴ = κL/σ, or equivalently R ∝ L¹⁄⁴ σ⁻¹⁄⁴. This predicts that cell size grows with the total polymerized MT length and shrinks with increasing cortical tension.
Empirical validation
The authors compiled measurements from 25 species (published electron‑microscopy data on MT number per cross‑section, MB length, and cell radius). Plotting R versus L on log‑log axes gave a slope of ~0.25, confirming the predicted scaling over two orders of magnitude. Using the fitted line they estimated σ ≈ 0.1 pN µm⁻¹ for red blood cells, a value comparable to the negative membrane tension measured in human erythrocytes (≈‑0.65 pN µm⁻¹). For human platelets the model predicts σ ≈ 40 pN µm⁻¹, close to independent measurements of platelet cortical tension (~35 pN µm⁻¹).
Computational modeling
To test whether dynamic cross‑linkers affect the long‑term equilibrium, the authors extended the Cytosim simulation engine. MTs were linked by stochastic Hookean springs representing cross‑linkers, and the cortex was modeled as a contractile surface with tension σ. Simulations were run with either zero or 10 000 cross‑linkers, varying κ, σ, and L. In all cases the steady‑state cell radius obeyed the same R⁴ ∝ L/σ relationship, indicating that cross‑linkers do not alter the static elastic balance when they can freely reorganize.
Short‑time dynamics and viscoelasticity
Platelet activation provides a rapid increase in cortical tension (within seconds). Live imaging of mouse platelets stained with SiR‑tubulin showed the MB coiling into a “baseball‑seam” shape as the cell rounded. Simulations reproducing a stepwise increase in σ reproduced two regimes: (i) a fast tension jump leads to MB buckling because cross‑linkers cannot unbind quickly, and (ii) a slow tension increase allows MT sliding, shortening the MB without coiling. Thus the MB behaves as an incompressible elastic ring on short timescales (elastic regime) but as a viscoelastic Kelvin‑Voigt element on longer timescales when cross‑linker turnover permits relative MT motion.
Buckling of a confined ring
The authors analytically derived the critical buckling force for a closed elastic ring of rigidity κr confined within a sphere of radius R: fB = 8πκr /R². In simulations, a confinement stiffness k exceeding a critical value kc = (n R γ)⁻¹ fB (where n is the number of discretization points and γ a geometric factor) caused the MB to coil. When the cell is flattened (oblate ellipsoid with minor axis r < R), the critical stiffness grows exponentially with 1 – r/R, making the flat configuration metastable. Consequently, a platelet in its discoid state can resist buckling forces up to ~50‑fold higher than a spherical cell of the same volume.
Phase diagram of tension versus MB length
By varying the nondimensional tension σR₀³/κr and the normalized MB length L/R₀ (with constant cell volume 4πR₀³/3), simulations revealed three regions: flat MB, permanently buckled MB, and a bistable zone where both states are possible. Hysteresis was observed: a cell initially flat remains flat for tensions below a forward critical value σf, while a cell initially buckled stays buckled until a lower backward critical value σb (σb < σf). This bistability explains how platelets can maintain a flattened shape under substantial mechanical stress yet rapidly transition to a rounded, coiled state during activation.
Implications and conclusions
The work provides a unified physical picture linking microtubule polymer content, cortical tension, and cross‑linker dynamics to the morphology and mechanical robustness of blood cells with marginal bands. The simple scaling law R⁴ ∝ L/σ is remarkably supported across diverse species, suggesting evolutionary tuning of MT polymer length and cortex tension to meet species‑specific circulatory demands. The viscoelastic transition of the MB explains the rapid shape changes observed during platelet activation, while the enhanced buckling resistance of a confined MB rationalizes how circulating cells survive extreme shear in narrow capillaries. Overall, the study bridges cell biology, biomechanics, and evolutionary physiology, offering quantitative tools for interpreting pathological alterations in MB composition or cortical tension (e.g., in thrombocytopenia or hereditary RBC disorders).
Comments & Academic Discussion
Loading comments...
Leave a Comment