Metabolic fluxes and value production
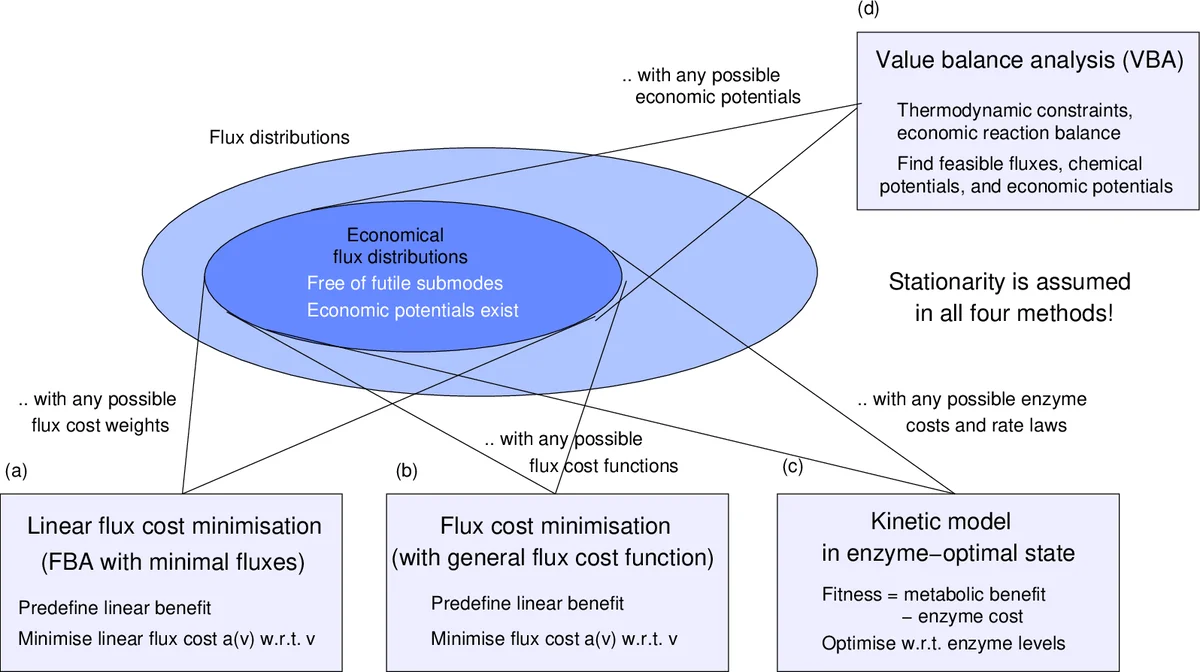
Metabolic fluxes in cells are governed by physical, physiological, and economic principles. Here I assume an optimal allocation of enzyme resources and postulate a general principle for metabolism: each enzyme must convert less valuable into more valuable metabolites to justify its own cost. The “values”, called economic potentials, describe the individual contributions of metabolites to cell fitness. Local value production implies that the cost of an enzyme must be balanced by a benefit, given by the economic potential difference the catalysed reaction multiplied by the flux. Flux profiles that satisfy this principle - i.e. for which consistent potentials can be found - are called economical. Economical fluxes must lead from lower to higher economic potentials, so certain flux cycles are incompatible with any choice of economic potentials and can be excluded. To obtain economical flux profiles, non-beneficial local patterns, called futile motifs, can be systematically removed from a given flux distribution. The principle of local value production resembles thermodynamic principles and complements them in models. Here I describe a modelling framework called Value Balance Analysis (VBA) that uses the two principles and yields the same solution as enzyme cost minimisation (in kinetic models) and flux cost minimisation (in FBA). Given an economical flux distribution, kinetic models in enzyme-optimal states and with these fluxes can be constructed systematically. VBA justifies the principle of minimal fluxes and the exclusion of futile cycles, predicts enzymes that could be plausible targets for regulation, provides criteria for the usage of enzymes and pathways, and explains the choice between high-yield and low-yield flux modes.
💡 Research Summary
The paper introduces Value Balance Analysis (VBA), a modeling framework that integrates physical, physiological, and economic principles to predict metabolic flux distributions and associated enzyme allocations. Building on the assumption that cells allocate their limited protein (enzyme) resources optimally, the author postulates a “value production” principle: each enzyme must convert metabolites of lower economic value into metabolites of higher economic value, thereby justifying its own cost. Economic value is quantified by “economic potentials” assigned to each metabolite, representing its contribution to cellular fitness (e.g., biomass formation).
In an enzymatic reaction, the product‑reactant economic potential difference (Δw) multiplied by the flux (v) must equal the enzyme investment (z > 0). This balance equation (Eq. 1) ensures that the benefit generated by the reaction (value production) exactly offsets the cost of the catalyst. A flux distribution satisfying this condition for every active reaction is termed “economical.” Consequently, economical fluxes must proceed from lower to higher economic potentials, mirroring the directionality imposed by thermodynamic potentials in classical metabolic modeling.
The framework identifies non‑beneficial patterns—so‑called futile motifs or cycles—whose potential differences sum to zero or negative values, making it impossible to satisfy Eq. 1 regardless of the chosen potentials. VBA systematically removes such motifs from any given flux distribution, thereby guaranteeing loop‑free, biologically plausible solutions.
Methodologically, VBA augments standard Flux Balance Analysis (FBA) with the economic balance constraint. The model retains the usual mass‑balance, steady‑state, and thermodynamic feasibility constraints, but adds Eq. 1 for every reaction. The objective function can be a traditional flux‑minimisation or enzyme‑cost minimisation; the author demonstrates mathematically that VBA yields the same optimal solution as Enzyme Cost Minimisation (ECM) in kinetic models and Flux Cost Minimisation (FCM) in constraint‑based models. Thus, the widely used “minimum‑flux” heuristic is not merely empirical—it is a direct consequence of the underlying economic balance.
VBA also accommodates empirical data: enzyme catalytic rates (kcat), measured protein abundances, and overall protein‑mass limits can be incorporated as cost weights (z). This makes the approach adaptable to real‑world proteomics and kinetic information, improving its predictive power.
The paper applies VBA to glycolytic pathways, illustrating how the model distinguishes between high‑yield (e.g., full oxidation to biomass) and low‑yield (e.g., fermentative) strategies. High‑yield routes generate larger economic potential differences but require higher enzyme investment; low‑yield routes may be favoured when protein resources are scarce. VBA predicts which pathway will be optimal under varying environmental or resource constraints.
Furthermore, the framework identifies candidate enzymes for regulation: reactions that violate the economic balance (Δw·v < z) must be down‑regulated or silenced, providing a systematic way to propose transcriptional or post‑translational control points.
The author supplies MATLAB code and a GitHub repository, facilitating adoption and further development. In summary, VBA offers a unified theory that couples thermodynamics, enzyme economics, and flux balance, explains the exclusion of futile cycles, justifies flux‑minimisation principles, predicts enzyme usage, and clarifies the trade‑offs between yield and enzyme cost in metabolic networks.
Comments & Academic Discussion
Loading comments...
Leave a Comment