Study of dose-dependent combination immunotherapy using engineered T cells and IL-2 in cervical cancer
Adoptive T cell based immunotherapy is gaining significant traction in cancer treatment. Despite its limited success, so far, in treating solid cancers, it is increasingly successful, demonstrating to have a broader therapeutic potential. In this paper we develop a mathematical model to study the efficacy of engineered T cell receptor (TCR) T cell therapy targeting the E7 antigen in cervical cancer cell lines. We consider a dynamical system that follows the population of cancer cells, TCR T cells, and IL-2. We demonstrate that there exists a TCR T cell dosage window for a successful cancer elimination that can be expressed in terms of the initial tumor size. We obtain the TCR T cell dose for two cervical cancer cell lines: 4050 and CaSki. Finally, a combination therapy of TCR T cell and IL-2 treatment is studied. We show that certain treatment protocols can improve therapy responses in the 4050 cell line, but not in the CaSki cell line.
💡 Research Summary
This paper presents a quantitative, mechanistic framework for evaluating dose‑dependent combination immunotherapy that couples engineered T‑cell receptor (TCR) T‑cell therapy with interleukin‑2 (IL‑2) administration in cervical cancer. The authors focus on two HPV‑16 + cell lines—4050 (oropharyngeal) and CaSki (cervical)—which have previously been shown to respond differently to TCR‑engineered T‑cells targeting the viral E7 antigen.
Model formulation
A three‑dimensional ordinary differential equation (ODE) system is constructed for the populations of cancer cells C(t), engineered TCR T‑cells T(t), and cytokine IL‑2 I(t):
- Cancer dynamics – logistic growth with intrinsic rate a and carrying‑capacity parameter b, minus T‑cell mediated killing at rate n·T·C.
- T‑cell dynamics – a source term s_T(t) representing a single bolus infusion at t = 0, natural death at rate d, antigen‑driven proliferation modeled by a Michaelis‑Menten term p·T·C/(g + C), a negative interaction term m·C·T (representing tumor‑induced T‑cell inactivation), and a positive feedback from IL‑2 via p₁·T·I/(g₁ + I).
- IL‑2 dynamics – a source term s_I(t) consisting of d discrete injections, natural decay at rate k, and production stimulated by the interaction of T‑cells and tumor cells via p₂·I·T/(g₂ + T).
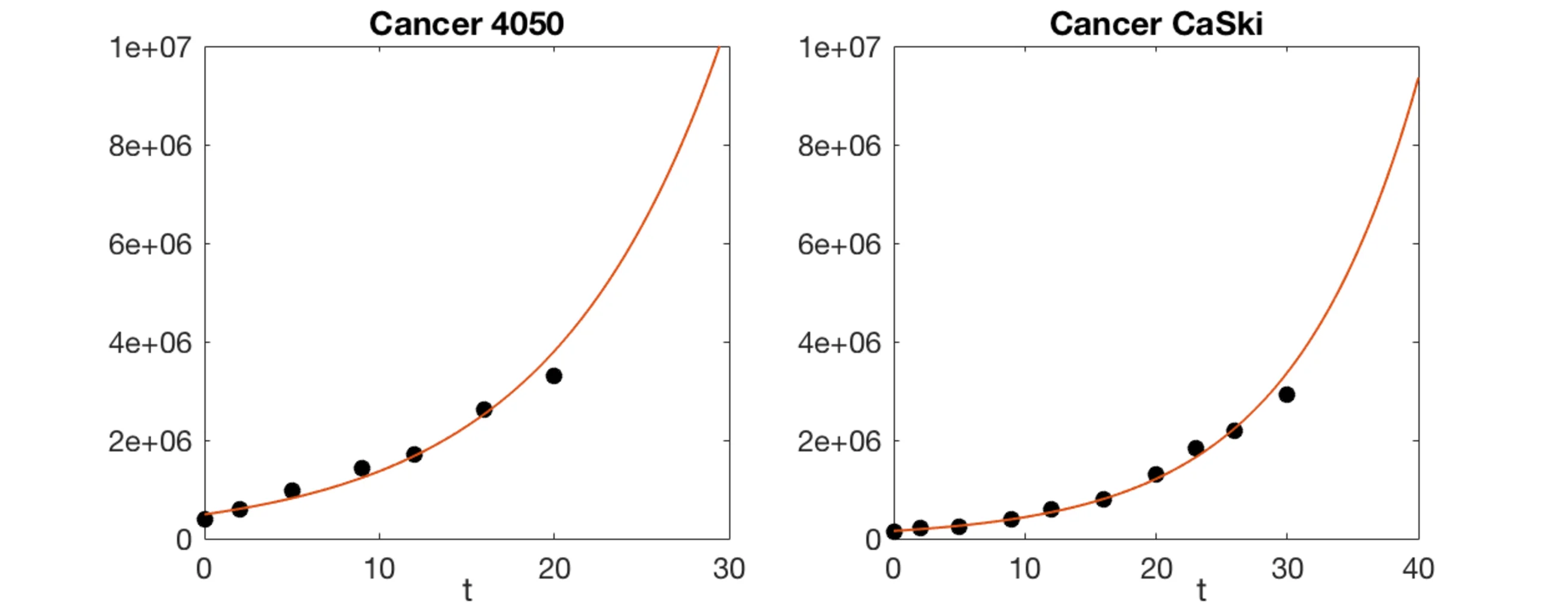
All parameters are biologically motivated and bounded using literature values (Table 2). The authors employ a delayed‑rejection adaptive Metropolis (DRAM) MCMC algorithm to fit the model to three experimental data sets from Jin et al. (2017): (i) untreated tumor growth, (ii) TCR T‑cell monotherapy at three doses (10⁵, 10⁶, 10⁷ cells), and (iii) combination therapy with IL‑2. Parameter estimates for each cell line are reported in Table 3.
Stability and therapeutic window analysis
Focusing first on the subsystem without IL‑2 (I = 0), the authors perform a steady‑state and Jacobian analysis. Four non‑negative equilibria exist, but only two are biologically relevant: (T, C) = (0, b⁻¹) (tumor at carrying capacity, no T‑cells) and a pair (C₁, T₁), (C₂, T₂) arising from the quadratic equation governing coexistence. Analytic conditions are derived:
- The “failure” equilibrium is stable when p < (m·b + d)/(g·b + 1).
- The “success” equilibrium (small tumor, high T‑cell count) exists and is locally stable when p > (m·b + d)(g·b + 1).
- In the intermediate regime √(d + m·g)² < p < (m·b + d)(g·b + 1), the fate depends on initial tumor size C(0) and initial T‑cell dose T(0).
These results are formalized in Theorem 1 and provide a mathematically precise definition of a “therapeutic window” that links the tumor‑induced T‑cell proliferation rate p, the tumor carrying capacity b, and the administered T‑cell dose.
Dose‑response simulations
Using the calibrated parameters, the authors simulate the full three‑species system for the two cell lines. For 4050, a single infusion of 10⁶ T‑cells already drives tumor regression, while 10⁵ is insufficient and 10⁷ yields rapid eradication. For CaSki, only the highest dose (10⁷) produces a measurable decline; lower doses fail because the tumor‑induced killing term n·T·C dominates. Phase‑plane plots (Figure 3) illustrate the separatrix that partitions the (C₀,T₀) plane into “success” (S) and “failure” (F) regions, confirming the analytical predictions.
IL‑2 combination therapy
The authors explore three IL‑2 schedules: a single bolus, short‑term repeated dosing, and prolonged (daily) dosing over the simulation horizon. In 4050, prolonged IL‑2 sustains T‑cell proliferation (via the p₁ term) and markedly improves tumor control, even at intermediate T‑cell doses. In CaSki, IL‑2 provides only a transient boost; the high killing coefficient n overwhelms the IL‑2‑mediated T‑cell expansion, and tumor growth proceeds similarly to monotherapy. These findings suggest that the benefit of IL‑2 is highly context‑dependent, governed by the underlying kinetic parameters of the tumor‑immune interaction.
Conclusions and outlook
The study demonstrates that a parsimonious ODE model, calibrated with limited in‑vitro data, can capture key qualitative differences between tumor lines and predict optimal dosing strategies for engineered T‑cell therapies. The analytical derivation of a dose‑dependent therapeutic window offers a practical tool for pre‑clinical trial design, allowing researchers to narrow the experimental search space for effective T‑cell numbers based on measured tumor burden.
Future extensions proposed include: (i) incorporation of checkpoint inhibition (CTLA‑4, PD‑1) to model synergistic drug combinations, (ii) explicit modeling of T‑cell exhaustion dynamics, and (iii) integration of patient‑specific tumor microenvironment data (e.g., cytokine profiles, immune infiltrate densities) to move toward personalized dosing regimens. Overall, the work bridges mathematical oncology and immunotherapy, providing a rigorous framework for optimizing combination treatments in solid tumors.
Comments & Academic Discussion
Loading comments...
Leave a Comment