An orthotropic electro-viscoelastic model for the heart with stress-assisted diffusion
We propose and analyse the properties of a new class of models for the electromechanics of cardiac tissue. The set of governing equations consists of nonlinear elasticity using a viscoelastic and orthotropic exponential constitutive law (this is so for both active stress and active strain formulations of active mechanics) coupled with a four-variable phenomenological model for human cardiac cell electrophysiology, which produces an accurate description of the action potential. The conductivities in the model of electric propagation are modified according to stress, inducing an additional degree of nonlinearity and anisotropy in the coupling mechanisms; and the activation model assumes a simplified stretch-calcium interaction generating active tension or active strain. The influence of the new terms in the electromechanical model is evaluated through a sensitivity analysis, and we provide numerical validation through a set of computational tests using a novel mixed-primal finite element scheme.
💡 Research Summary
This paper introduces a comprehensive electromechanical model for human cardiac tissue that integrates orthotropic nonlinear elasticity, viscoelasticity, active contraction mechanisms (both active stress and active strain formulations), and a novel stress‑assisted diffusion (SAD) feedback on electrical conductivity. The mechanical part is built on an orthotropic exponential strain‑energy function that distinguishes fiber, sheet, and cross‑fiber directions, with anisotropic contributions activated only in tension (positive part of the exponential terms) to reflect the asymmetric response of myocardial fibers under compression versus extension. Viscous effects are incorporated via a Maxwell‑type parallel arrangement, yielding a finite‑strain viscoelastic formulation that respects near‑incompressibility through a hydrostatic pressure field.
Active contraction is modeled in two complementary ways. In the active‑stress approach, the total first Piola‑Kirchhoff stress is decomposed into passive and active parts, with the active stress tensor expressed as a combination of fiber‑aligned, sheet‑aligned, and cross‑fiber components scaled by an active tension scalar Tₐ and direction‑specific coefficients (κ_sn, κ_nn). This formulation captures the directional dependence of myocardial activation and allows for different magnitudes of stress in the off‑diagonal sheet‑cross‑fiber coupling. In the active‑strain approach, the deformation gradient is multiplicatively split into an elastic part and an active part F_A that introduces prescribed stretches γ_f, γ_s, γ_n along the three material directions. The elastic response then depends only on the elastic deformation, preserving strong ellipticity of the energy functional. Both formulations are embedded within the same orthotropic framework, enabling direct comparison of their impact on deformation and electrophysiology.
The electrical subsystem employs a four‑variable phenomenological ionic model calibrated for human action potentials, capable of reproducing normal pacing, sustained fibrillation, and spiral‑wave breakup. Crucially, the diffusion tensor governing the monodomain equation is modified by the Kirchhoff stress: D = D₀ (I + α σ_K), where α is a coupling parameter. This stress‑assisted diffusion introduces a mechano‑electrical feedback loop: mechanical loading alters conductivity, which in turn influences wave propagation speed and direction. Such a feedback is supported by experimental observations in cardiac tissue and polymer gels, but its systematic inclusion together with orthotropy and viscoelasticity is novel.
To solve the coupled system, the authors formulate a three‑field mixed‑primal variational problem with primary variables displacement u, pressure p, and Kirchhoff stress σ. This formulation, derived from a modified Hu‑Washizu principle, avoids volumetric locking on low‑order triangular/tetrahedral meshes by adding a pressure‑stabilisation term. It also provides σ directly, which is required for the SAD term, eliminating the need for post‑processing. A consistent Newton linearisation is presented, and a mixed‑primal finite element discretisation using continuous piecewise‑linear displacement, discontinuous piecewise‑constant pressure, and appropriate stress spaces is implemented.
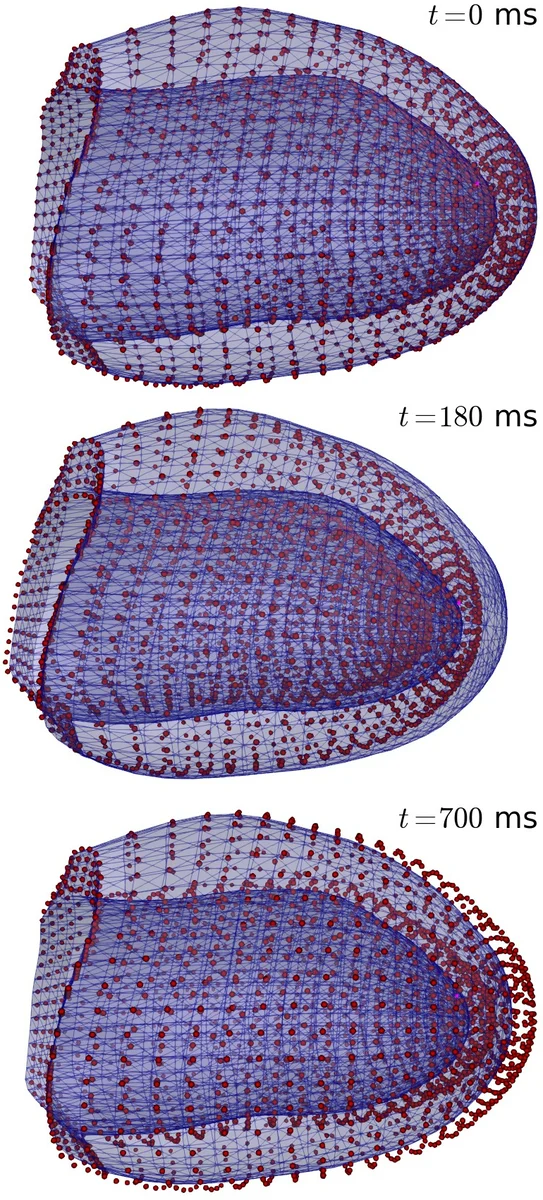
Numerical experiments are performed in both two‑dimensional idealised domains and realistic three‑dimensional ventricular geometries. A sensitivity analysis explores the influence of material parameters (a_f, b_f, κ coefficients), the SAD coupling α, and the active‑contraction parameters on electrophysiological outcomes such as conduction velocity, activation time, and propensity for re‑entry. Results show that regions of high tensile stress experience reduced conductivity, leading to delayed wavefronts and, in some scenarios, the formation of functional blocks that can trigger arrhythmogenic patterns. Comparisons between active‑stress and active‑strain formulations reveal that the active‑strain model better reproduces physiologically realistic deformation fields, while the active‑stress model is computationally simpler and integrates more readily with existing passive‑elastic frameworks.
Overall, the paper delivers a mathematically rigorous, physically detailed, and numerically robust framework for cardiac electromechanics that captures the interplay of anisotropic elasticity, viscoelastic damping, active contraction, and stress‑dependent electrical diffusion. The mixed‑primal finite element method ensures stable and accurate simulations on complex geometries, paving the way for patient‑specific studies, drug‑effect simulations, and the design of therapeutic interventions that exploit mechano‑electrical feedback.
Comments & Academic Discussion
Loading comments...
Leave a Comment