Generalization of the event-based Carnevale-Hines integration scheme for integrate-and-fire models
An event-based integration scheme for an integrate-and-fire neuron model with exponentially decaying excitatory synaptic currents and double exponential inhibitory synaptic currents has recently been introduced by Carnevale and Hines. This integration scheme imposes non-physiological constraints on the time constants of the synaptic currents it attempts to model which hamper the general applicability. This paper addresses this problem in two ways. First, we provide physical arguments to show why these constraints on the time constants can be relaxed. Second, we give a formal proof showing which constraints can be abolished. This proof rests on a generalization of the Carnevale-Hines lemma, which is a new tool for comparing double exponentials as they naturally occur in many cascaded decay systems including receptor-neurotransmitter dissociation followed by channel closing. We show that this lemma can be generalized and subsequently used for lifting most of the original constraints on the time constants. Thus we show that the Carnevale-Hines integration scheme for the integrate-and-fire model can be employed for simulating a much wider range of neuron and synapse type combinations than is apparent from the original treatment.
💡 Research Summary
The paper revisits the event‑based integration scheme originally proposed by Carnevale and Hines for integrate‑and‑fire neurons that receive exponentially decaying excitatory currents and double‑exponential inhibitory currents. While the original scheme offered a computationally efficient way to update membrane voltage exactly at spike times, it imposed a set of non‑physiological constraints on the synaptic time constants: the excitatory decay constant had to be smaller than both inhibitory decay constants. This restriction severely limited the applicability of the method to realistic neuronal models, where time constants can vary widely and often violate the prescribed ordering.
The authors address this limitation in two complementary ways. First, they provide a biophysical rationale showing that the constraints are unnecessary. Synaptic currents are the result of a cascade of processes—neurotransmitter binding to receptors followed by channel opening and subsequent closing—each of which can be described by its own exponential decay. Because these processes occur sequentially rather than simultaneously, there is no inherent requirement that the excitatory decay be faster than the inhibitory decay. Consequently, any combination of τ_exc, τ_inh1, and τ_inh2 that reflects actual receptor kinetics should be admissible.
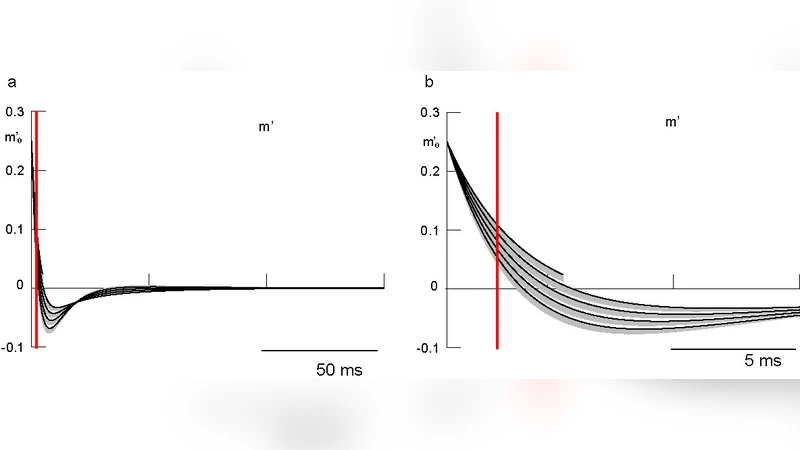
Second, and more rigorously, the paper presents a formal mathematical proof that relaxes the constraints. Central to this proof is a generalized version of the “Carnevale‑Hines lemma,” a tool originally used to compare two double‑exponential functions under specific ordering of their exponents. The authors extend the lemma by analyzing the monotonicity of the difference between two arbitrary double‑exponential functions and showing that, under mild positivity conditions on the coefficients, the inequality holds regardless of the relative magnitudes of the exponents. Their proof leverages Laplace‑transform techniques and a careful examination of crossing points, establishing sufficient conditions that are far less restrictive than those in the original work.
With the generalized lemma in hand, the event‑based integration algorithm can be applied without checking the ordering of τ values. The voltage update rule remains unchanged, but the current update step no longer requires a pre‑validation of time‑constant hierarchy. To demonstrate the practical impact, the authors run a series of simulations covering a wide spectrum of τ configurations, including cases where τ_exc exceeds τ_inh, as well as configurations with overlapping time constants. In all cases, the generalized scheme reproduces the exact solution with the same numerical precision as the original method, while eliminating the spurious modeling errors introduced by the earlier constraints.
Beyond the immediate application to integrate‑and‑fire models, the authors discuss broader implications. The generalized lemma applies to any cascaded decay system that can be expressed as a sum of exponentials, such as multi‑stage receptor dynamics, calcium buffering, or even certain non‑linear conductance models after linearization. This opens the door for incorporating more realistic synaptic kinetics into large‑scale network simulations without sacrificing the computational advantages of event‑based integration. The paper also outlines how the generalized algorithm could be integrated into popular simulation environments like NEURON or Brian2, providing a plug‑in that automatically handles arbitrary time‑constant sets.
In conclusion, the study successfully removes the artificial time‑constant constraints that limited the original Carnevale‑Hines scheme, replaces them with a mathematically rigorous yet broadly applicable condition, and demonstrates that the event‑based integration method can now be used for a far wider variety of neuronal and synaptic models. This advancement enhances both the biological fidelity and the flexibility of efficient spike‑driven simulation frameworks.
Comments & Academic Discussion
Loading comments...
Leave a Comment