Normal and pathological dynamics of platelets in humans
We develop a comprehensive mathematical model of platelet, megakaryocyte, and thrombopoietin dynamics in humans. We show that there is a single stationary solution that can undergo a Hopf bifurcation, and use this information to investigate both normal and pathological platelet production, specifically cyclic thrombocytopenia. Carefully estimating model parameters from laboratory and clinical data, we then argue that a subset of parameters are involved in the genesis of cyclic thrombocytopenia based on clinical information. We provide excellent model fits to the existing data for both platelet counts and thrombopoietin levels by changing six parameters that have physiological correlates. Our results indicate that the primary change in cyclic thrombocytopenia is a major interference with or destruction of the thrombopoietin receptor with secondary changes in other processes, including immune-mediated destruction of platelets and megakaryocyte deficiency and failure in platelet production. This study makes a major contribution to the understanding of the origin of cyclic thrombopoietin as well as significantly extending the modeling of thrombopoiesis.
💡 Research Summary
This paper presents a comprehensive mathematical framework for the regulation of human platelet production, incorporating the dynamics of megakaryocytes, circulating platelets, and the thrombopoietin (TPO) feedback loop. The authors construct an age‑structured partial differential equation model for megakaryocyte development, separating the mitotic phase (cell division) from the endomitosis phase (nuclear replication without cytokinesis). Both phases are driven by TPO‑dependent proliferation rates modeled as Hill functions, allowing a basal proliferation rate in the absence of TPO and a saturating maximal rate at high TPO concentrations.
Platelet dynamics are described by a delay differential equation that accounts for platelet release from mature megakaryocytes after a fixed maturation delay, random platelet clearance, and a platelet‑dependent removal term representing macrophage‑mediated destruction. TPO is produced constitutively by the liver, degrades randomly, and is cleared through binding to receptors on both platelets and megakaryocytes; this receptor‑mediated clearance creates a negative feedback loop whereby low platelet counts raise circulating TPO levels.
Mathematical analysis demonstrates that the system possesses a unique positive equilibrium for any biologically realistic parameter set. Linearization around this equilibrium yields a characteristic equation whose roots determine stability. By varying key parameters, the authors identify a Hopf bifurcation: a critical set of parameter values at which a pair of complex conjugate eigenvalues cross the imaginary axis, giving rise to sustained oscillations. Sensitivity analysis highlights six parameters with the greatest impact on system stability: the maximal megakaryocyte proliferation rates (mitosis and endomitosis), the platelet‑dependent clearance rate, the random platelet degradation rate, the TPO production rate, and, most importantly, the rate of TPO‑receptor loss (κ_T).
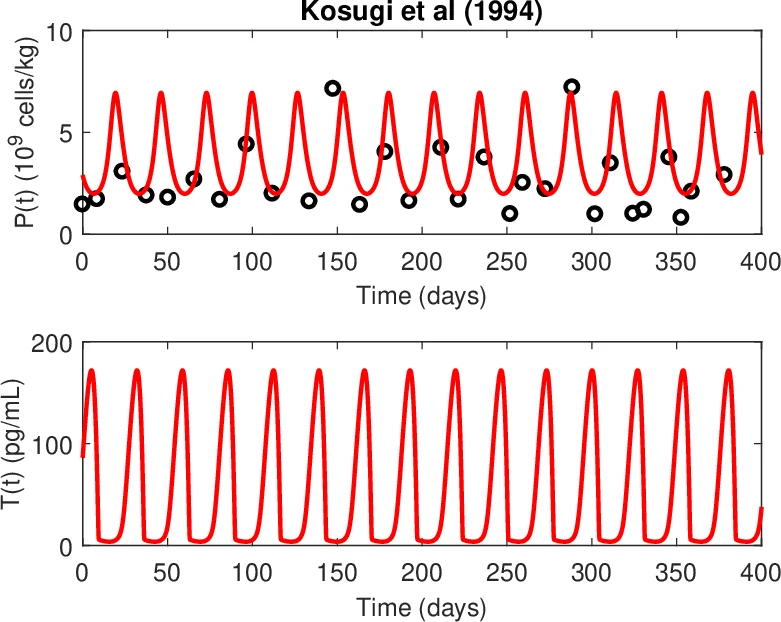
To validate the model, the authors fit it to clinical time‑series data from healthy volunteers and from three patients with cyclic thrombocytopenia (CT). Parameter estimation for the healthy cohort uses literature values and Bayesian inference, achieving excellent agreement with observed platelet counts (≈150–300 × 10⁹ L⁻¹) and TPO concentrations (≈50–150 pg mL⁻¹). For CT patients, the model reproduces the characteristic 30‑day oscillations in platelet counts (ranging from 1 × 10⁹ to 2 000 × 10⁹ L⁻¹) and the accompanying TPO fluctuations by increasing κ_T roughly five‑fold, modestly reducing the maximal megakaryocyte proliferation rates, and elevating the platelet‑dependent clearance term. No additional structural changes to the model are required; the same set of six altered parameters suffices to capture the pathological dynamics across all three patients.
The results support a mechanistic hypothesis in which primary interference with or destruction of the TPO receptor leads to elevated circulating TPO, which in turn suppresses megakaryocyte maturation and platelet production while simultaneously exposing platelets to heightened immune‑mediated destruction. Secondary effects—such as modest megakaryocyte deficiency and increased platelet clearance—amplify the oscillatory behavior but are not the root cause. The identification of a Hopf bifurcation provides a theoretical explanation for why relatively small perturbations in receptor function can shift the hematopoietic system from a stable steady state to a limit‑cycle regime, offering a quantitative basis for therapeutic strategies aimed at restoring receptor function or dampening immune‑mediated platelet loss.
In conclusion, this study delivers a biologically grounded, mathematically rigorous model that unifies normal platelet homeostasis and the pathology of cyclic thrombocytopenia within a single dynamical system. By linking specific physiological parameters to the emergence of oscillations via a Hopf bifurcation, it opens avenues for patient‑specific parameter inference and for the rational design of interventions targeting the TPO‑receptor axis. Future work may extend the framework to incorporate stochastic fluctuations, multi‑lineage interactions, or drug pharmacodynamics, thereby enhancing its clinical applicability in the management of platelet‑related disorders.
Comments & Academic Discussion
Loading comments...
Leave a Comment