Maintaining the stem cell niche in multicellular models of epithelia
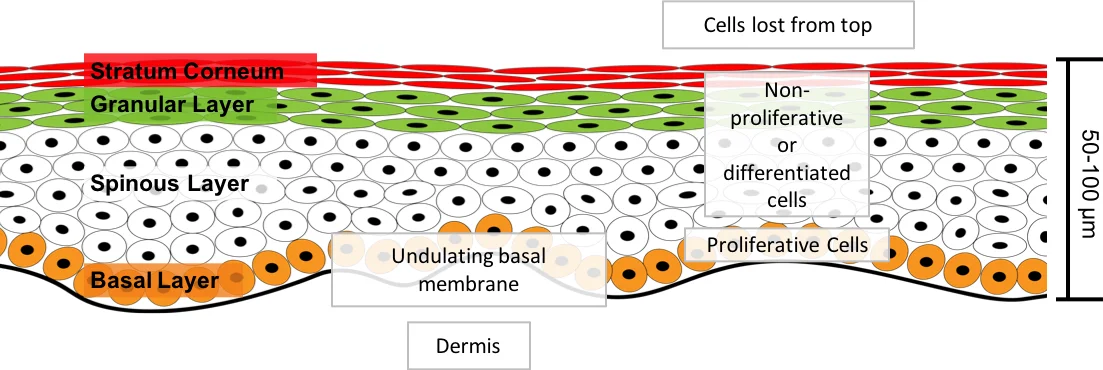
The maintenance of the proliferative cell niche is critical to epithelial tissue morphology and function. In this paper we investigate how current modelling methods can result in the erroneous loss of proliferative cells from the proliferative cell niche. Using an established model of the inter-follicular epidermis we find there is a limit to the proliferative cell densities that can be maintained in the basal layer (the niche) if we do not include additional mechanisms to stop the loss of proliferative cells from the niche. We suggest a new methodology that enables maintenance of a desired homeostatic population of proliferative cells in the niche: a rotational force is applied to the two daughter cells during the mitotic phase of division to enforce a particular division direction. We demonstrate that this new methodology achieves this goal. This methodology reflects the regulation of the orientation of cell division.
💡 Research Summary
This paper investigates a critical shortcoming of widely used off‑lattice, cell‑center agent‑based models of epithelial tissues: the inadvertent loss of proliferative cells from the basal stem‑cell niche. Using an established three‑dimensional overlapping‑spheres model of the human inter‑follicular epidermis (IFE), the authors demonstrate that the conventional method of splitting a parent cell into two daughters—each subjected to distinct intercellular forces—leads to rapid depletion of basal proliferative cells. In 25 stochastic simulations, more than 80 % of the original proliferative population disappeared within 15 days, and after 1,000 days the mean number of basal proliferative cells fell to 2.8, far below experimentally reported growth fractions of 20‑60 % in normal epidermis.
The authors review two common strategies for preserving the niche. The first involves “pinning” cells to the basal membrane or dramatically increasing membrane adhesion. While this can prevent loss, it imposes unrealistic constraints on cell mobility and fails to accommodate population‑asymmetry dynamics. The second relies on extracellular signalling gradients (e.g., Wnt, Notch) that restrict proliferation to cells near the membrane. Although biologically plausible, such signalling alone cannot regulate excessive proliferation and does not explain the coexistence of multiple proliferative cell types observed in the epidermis.
To address these limitations, the paper proposes a third, mechanistic approach: directed division through the application of a rotational force during mitosis. By imposing a torque that aligns the mitotic spindle toward a pre‑specified orientation, the model forces one daughter cell to remain in the basal layer while the other moves suprabasally. This “rotational force” mimics experimentally observed regulation of division orientation in developing epidermis and adult skin, where spindle alignment is linked to cell polarity and basal‑membrane contacts.
Simulations incorporating the rotational force show robust maintenance of the desired proliferative population. Across 1,000 days, the average proportion of basal proliferative cells remains above 95 % of the initial value, and stochastic variability is markedly reduced compared to the unmodified model. The authors argue that this method requires only a simple mechanical addition to existing off‑lattice frameworks and can be generalized to other stratified epithelia such as colonic crypts.
The study highlights that neglecting division‑orientation mechanisms can lead to substantial artefacts in tissue‑scale predictions, including altered morphology, impaired stratification, and distorted clonal dynamics. By integrating a biologically motivated rotational torque, modelers can achieve more realistic niche homeostasis without resorting to ad‑hoc pinning or overly simplistic signalling rules. The paper calls for further experimental quantification of spindle‑orientation forces (e.g., LGN‑NuMA complexes, microtubule acetylation) to calibrate the torque parameters and validate the approach in vivo. Overall, the work provides a compelling, technically feasible solution to a pervasive problem in epithelial tissue modeling, advancing both computational methodology and our understanding of stem‑cell niche maintenance.
Comments & Academic Discussion
Loading comments...
Leave a Comment