A mechanism for sarcomere breathing: volume change and advective flow within the myofilament lattice
During muscle contraction, myosin motors anchored to thick filaments bind to and slide actin thin filaments. These motors rely on energy derived from ATP, supplied, in part, by diffusion from the sarcoplasm to the interior of the lattice of actin and myosin filaments. The radial spacing of filaments in this lattice may change or remain constant during contraction. If the lattice is isovolumetric, it must expand when the muscle shortens. If, however, the spacing is constant or has a different pattern of axial and radial motion, then the lattice changes volume during contraction, driving fluid motion and assisting in the transport of molecules between the contractile lattice and the surrounding intracellular space. We first create an advective-diffusive-reaction flow model and show that the flow into and out of the sarcomere lattice would be significant in the absence of lattice expansion. Advective transport coupled to diffusion has the potential to substantially enhance metabolite exchange within the crowded sarcomere. Using time-resolved x-ray diffraction of contracting muscle, we next show that the contractile lattice is neither isovolumetric nor constant in spacing. Instead, lattice spacing is time-varying, depends on activation, and can manifest as an effective time-varying Poisson ratio. The resulting fluid flow in the sarcomere lattice of synchronous insect flight muscles is greater than expected for constant lattice spacing conditions. Lattice spacing depends on a variety of factors that produce radial force, including crossbridges, titin-like molecules, and other structural proteins. Volume change and advective transport varies with the phase of muscle stimulation but remains significant at all conditions. Akin to “breathing,” advective-diffusive transport in sarcomeres is sufficient to promote metabolite exchange and may play a role in the regulation of contraction itself.
💡 Research Summary
The paper proposes and validates a novel mechanism by which the contractile lattice of a sarcomere undergoes volume changes during contraction, generating fluid flow that augments diffusive transport of metabolites such as ATP. The authors combine a theoretical advective‑diffusive‑reaction model with high‑speed time‑resolved X‑ray diffraction measurements on intact insect flight muscle to demonstrate that the lattice is neither isovolumetric nor of constant spacing, but instead exhibits a time‑varying “effective Poisson ratio” that drives a breathing‑like fluid exchange.
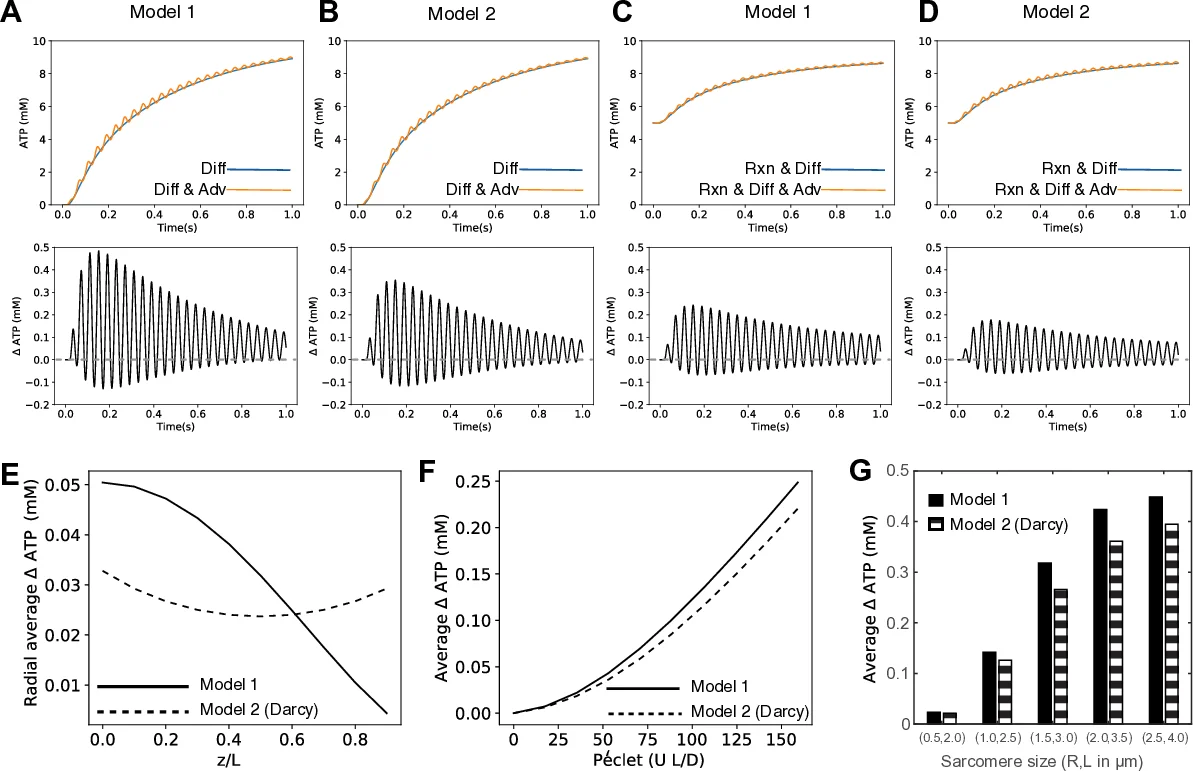
The modeling component starts from the one‑dimensional advection‑diffusion‑reaction equation (∂c/∂t = D∇²c – u·∇c – χ) where c is ATP concentration, D the diffusion coefficient, u the velocity field inside the sarcomere, and χ the Michaelis‑Menten consumption term. Two distinct flow fields are constructed. Model 1 assumes a plug flow with a no‑slip condition at the Z‑disk, yielding an axial velocity u_z that varies linearly with axial position and a radial velocity u_r proportional to radius. Model 2 treats the filament lattice as a porous medium, applying Darcy’s law with separate axial (k_l) and radial (k_r) permeabilities; pressure gradients generate both axial and radial components, with the radial outflow maximal at the sarcomere perimeter. Both models satisfy continuity in cylindrical coordinates.
Numerical simulations use a half‑sarcomere of radius 1.5 µm and length 3 µm, driven at 25 Hz with a 5 % length oscillation (0.15 µm). Diffusion coefficient D = 8 × 10⁻⁹ cm² s⁻¹, external ATP concentration 10 mM, V_max = 29.3 mM s⁻¹, and K_m = 101 mM are employed. When only diffusion is considered, internal ATP builds up very slowly; however, inclusion of the advective velocity fields dramatically accelerates ATP delivery, producing periodic spikes in concentration that can be 2–3 times higher than diffusion alone. Model 1 shows the strongest radial flux at the M‑line, while Model 2 predicts maximal flux near the sarcomere wall due to pressure‑driven Darcy flow.
Experimentally, the authors record lattice spacing (d₁₀) changes in Manduca sexta dorsal longitudinal flight muscle using synchrotron X‑ray diffraction at kilohertz rates. The spacing oscillates by ~0.02 nm during each contraction cycle, contradicting the assumption of constant spacing. The effective Poisson ratio, derived from simultaneous axial strain and radial spacing measurements, varies between –0.2 and +0.1 throughout the cycle, indicating that the lattice expands radially when the sarcomere shortens and contracts radially when it lengthens, but not in a simple linear fashion. This time‑varying geometry necessarily creates volume changes that drive fluid in and out of the lattice.
The combined modeling and experimental evidence supports the concept of “sarcomere breathing”: periodic volume fluctuations generate advective flow that, together with diffusion, ensures rapid replenishment of ATP and other metabolites in the crowded filament lattice. This mechanism is especially relevant for muscles that operate at high frequency and power, such as insect flight muscles, sound‑producing muscles, and cardiac tissue. The authors argue that sarcomere breathing could be a fundamental, yet underappreciated, contributor to muscle energetics, force production, and possibly to the regulation of contraction itself. The findings open new avenues for investigating muscle pathophysiology, designing bio‑inspired actuators, and improving models of intracellular transport in highly ordered, anisotropic structures.
Comments & Academic Discussion
Loading comments...
Leave a Comment