The syncytial Drosophila embryo as a mechanically excitable medium
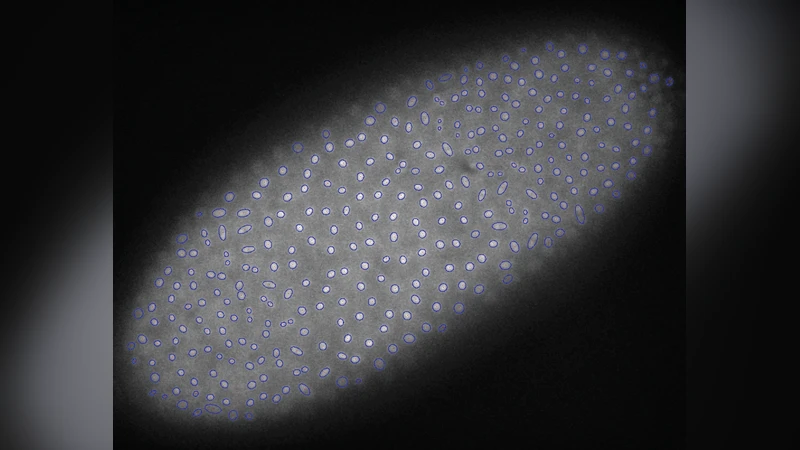
Mitosis in the early syncytial Drosophila embryo is highly correlated in space and time, as manifested in mitotic wavefronts that propagate across the embryo. In this paper we investigate the idea that the embryo can be considered a mechanically-excitable medium, and that mitotic wavefronts can be understood as nonlinear wavefronts that propagate through this medium. We study the wavefronts via both image analysis of confocal microscopy videos and theoretical models. We find that the mitotic waves travel across the embryo at a well-defined speed that decreases with replication cycle. We find two markers of the wavefront in each cycle, corresponding to the onsets of metaphase and anaphase. Each of these onsets is followed by displacements of the nuclei that obey the same wavefront pattern. To understand the mitotic wavefronts theoretically we analyze wavefront propagation in excitable media. We study two classes of models, one with biochemical signaling and one with mechanical signaling. We find that the dependence of wavefront speed on cycle number is most naturally explained by mechanical signaling, and that the entire process suggests a scenario in which biochemical and mechanical signaling are coupled.
💡 Research Summary
In this study the authors investigate mitotic wavefronts in the early syncytial Drosophila embryo and propose that the embryo functions as a mechanically excitable medium rather than solely a chemically excitable one. Using high‑resolution confocal microscopy of GFP‑labeled histones, they automatically track nuclear positions, shapes, and the ratio of the major to minor axes of the chromatin mass throughout cycles 9–13. They identify two distinct markers of mitosis— the onset of metaphase (chromosome condensation into an ellipsoidal plate) and the onset of anaphase (a rapid 90° rotation and change in aspect ratio). Both markers appear as coherent wavefronts that originate near the embryonic poles and travel toward the equator at a constant speed for a given cycle. Importantly, the measured wave speed decreases systematically from one nuclear division cycle to the next, despite the fact that nuclear density (and thus inter‑nuclear spacing) increases.
In addition to the shape changes, the authors observe a collective “breathing” motion of nuclei along the embryo’s long axis. After each metaphase or anaphase transition, nuclei shift in the same direction as the wavefront, producing a global displacement pattern that mirrors the timing of the shape‑based markers. The interval between metaphase and anaphase onsets remains constant within a single embryo but varies between experimental batches, while the overall duration of each nuclear cycle lengthens slightly with cycle number.
To explain these observations the authors construct two theoretical frameworks. The first is a classic chemically excitable medium model: each nucleus releases a diffusible activator A when the local concentration exceeds a threshold α. The activator diffuses with coefficient D, and a refractory period equal to interphase prevents re‑activation within the same cycle. In this model the wave speed v depends on D, the nuclear spacing a, and α, and it predicts that v should increase as a decreases (i.e., as nuclear density rises). This prediction contradicts the experimental finding of decreasing speed with increasing density, indicating that a purely chemical diffusion mechanism cannot account for the data.
The second framework treats the embryo as a mechanically excitable medium. The authors posit that the mechanical forces generated at the onset of metaphase or anaphase produce a stress field σ in the surrounding cytoskeleton. Stress propagates diffusively with a mechanical diffusion constant D_mech, and when σ exceeds a critical value σ_c, neighboring nuclei are mechanically triggered to enter mitosis. In this model the wave speed scales inversely with the inter‑nuclear distance (v ∝ σ_c/(a√D_mech)), naturally yielding slower propagation as nuclei become more closely packed. By inserting the experimentally measured spacing a≈2^(-β(n‑n0)) with β≈0.46, the model reproduces the observed exponential decay of wave speed across cycles.
The authors further discuss evidence for concurrent biochemical signals, such as calcium spikes that travel at the same speed as the mitotic wavefronts, suggesting that mechanical and chemical cues may be coupled. They argue that nuclei likely sense both stress and biochemical cues, forming a hybrid excitable medium in which mechanical signaling dominates the observed speed trends while biochemical signaling could modulate timing or robustness.
Overall, the paper provides (1) quantitative imaging evidence for two synchronized mitotic wavefronts, (2) a systematic decrease in wave speed with successive nuclear cycles, (3) a global nuclear displacement pattern that follows the same wave dynamics, (4) a rigorous comparison of chemical diffusion versus mechanical stress diffusion models, and (5) the conclusion that mechanical signaling, possibly in concert with biochemical pathways, best explains the data. This work introduces the concept of a mechanically excitable embryonic tissue and opens avenues for exploring similar mechano‑chemical wave phenomena in other developmental and physiological contexts.
Comments & Academic Discussion
Loading comments...
Leave a Comment