Computational modeling of degradation process of biodegradable magnesium biomaterials
Despite the advantages of using biodegradable metals in implant design, their uncontrolled degradation and release remain a challenge in practical applications. A validated computational model of the degradation process can facilitate the tuning of implant biodegradation by changing design properties. In this study, a physicochemical model was developed by deriving a mathematical description of the chemistry of magnesium biodegradation and implementing it in a 3D computational model. The model parameters were calibrated using the experimental data of hydrogen evolution by performing a Bayesian optimization routine. The model was validated by comparing the predicted change of pH in saline and buffered solutions with the experimentally obtained values from corrosion tests, showing maximum 5% of difference, demonstrating the model’s validity to be used for practical cases.
💡 Research Summary
**
This paper presents a comprehensive three‑dimensional computational framework for predicting the degradation of commercially pure (CP) magnesium (Mg) biomaterials used in biodegradable implants. Recognizing that uncontrolled corrosion can compromise mechanical integrity and generate excess hydrogen gas, the authors aim to develop a physics‑based model that captures the coupled chemical reactions, ion transport, and evolving geometry of the metal‑solution interface.
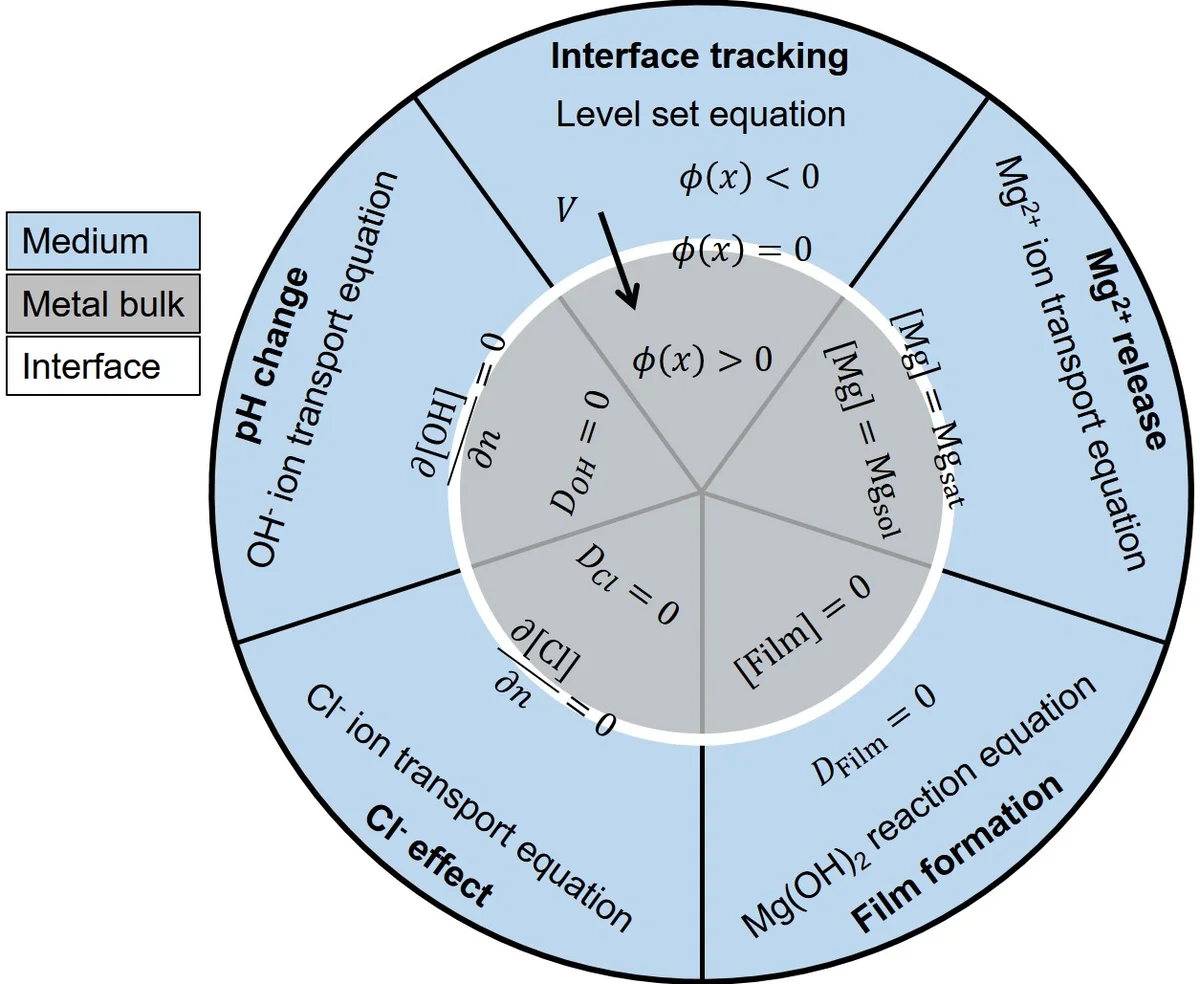
The degradation process is formulated as a reaction‑diffusion system involving four scalar fields: Mg²⁺ concentration, a protective Mg(OH)₂ film, chloride (Cl⁻) concentration, and hydroxide (OH⁻) concentration. The governing partial differential equations (PDEs) (10‑13) describe diffusion of each species together with source terms derived from the underlying electrochemical reactions (Mg oxidation, water reduction, and Cl⁻‑induced film dissolution). The protective film’s porosity (ϕ) and tortuosity (τ) are incorporated to compute an effective diffusion coefficient Dₑ that varies with the local film thickness, thereby reproducing the experimentally observed rapid initial corrosion followed by a slower, diffusion‑limited regime.
To track the moving metal‑solution boundary, the authors employ the level‑set method. An implicit signed‑distance function φ(x,t) defines the interface as the zero‑isosurface. The normal interface velocity V_N is obtained from the Rankine–Hugoniot condition (mass balance across the moving front) and inserted into the simplified level‑set equation (4). For low‑diffusion scenarios, the initial interface advance is accelerated using the analytical Stefan solution (Eqs. 1‑2) to mimic the experimentally observed fast start‑up of corrosion. This hybrid approach enables robust simulation of complex 3‑D geometries without mesh remeshing.
Parameter calibration is performed via Bayesian optimization, using experimentally measured hydrogen evolution data from immersion tests. This probabilistic approach not only identifies optimal values for reaction rate constants (k₁, k₂), the film saturation parameter (β), and diffusion coefficients but also quantifies their uncertainties. The calibrated model is then validated against pH measurements obtained from Mg samples immersed in two media: a simple NaCl saline solution and a simulated body fluid (SBF) that contains additional inorganic ions and buffering capacity. The simulated pH trajectories match the experimental curves with an average error below 5 %, demonstrating that the model accurately captures ion release, buffering, and protective film dynamics.
Key strengths of the work include: (1) a physically consistent coupling of reaction‑diffusion chemistry with moving‑boundary mechanics, (2) the use of level‑sets to handle arbitrary 3‑D shapes, (3) Bayesian calibration that reduces the need for extensive trial‑and‑error experiments, and (4) validation against independent experimental observables (pH, hydrogen evolution). These features collectively advance the state‑of‑the‑art beyond earlier continuous‑damage or purely phenomenological models, which often neglect diffusion or interface motion.
Nevertheless, the study has limitations. Oxygen reduction reactions, which can contribute up to 20 % of the cathodic current in oxygen‑rich environments, are omitted, potentially under‑predicting corrosion in vivo. The protective layer is simplified to pure Mg(OH)₂, ignoring the formation of mixed MgO/Mg(OH)₂ phases and localized pitting induced by chloride. Temperature effects, fluid flow, and shear stresses are not considered, restricting applicability to static immersion conditions. Finally, validation is limited to two test solutions; extrapolation to real physiological conditions (blood flow, tissue fluids) will require additional experiments.
In conclusion, the authors deliver a validated, physics‑based 3‑D model that can predict both the rate and morphological evolution of Mg implant degradation. By integrating reaction‑diffusion PDEs, level‑set interface tracking, and Bayesian parameter estimation, the framework provides a powerful tool for pre‑clinical implant design, enabling systematic exploration of alloy composition, surface treatments, and geometric configurations to achieve desired biodegradation profiles. Future work should extend the model to incorporate temperature, convection, multi‑species corrosion products, and in vivo validation, thereby paving the way for fully predictive, patient‑specific biodegradable implant design.
Comments & Academic Discussion
Loading comments...
Leave a Comment