ODE network model for nonlinear and complex agricultural nutrient solution system
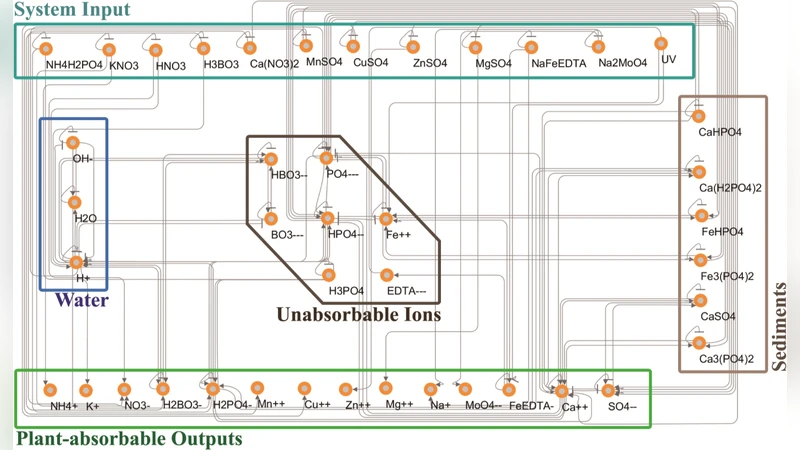
In closed hydroponic systems, periodic readjustment of nutrient solution is necessary to continuously provide stable environment to plant roots because the interaction between plant and nutrient solution changes the rate of ions in it. The traditional method is to repeat supplying small amount of premade concentrated nutrient solution, measuring total electric conductivity and pH of the tank only. As it cannot control the collapse of ion rates, recent researches try to measure the concentration of individual components to provide insufficient ions only. However, those approaches use titrationlike heuristic approaches, which repeat adding small amount of components and measuring ion density a lot of times for a single control input. Both traditional and recent methods are not only time-consuming, but also cannot predict chemical reactions related with control inputs because the nutrient solution is a nonlinear complex system, including many precipitation reactions and complicated interactions. We present a continuous network model of the nutrient solution system, whose reactions are described as differential equations. The model predicts molar concentration of each chemical components and total dissolved solids with low error. This model also can calculate the amount of chemical compounds needed to produce a desired nutrient solution, by reverse calculation from dissolved ion concentrations.
💡 Research Summary
The paper addresses a fundamental challenge in closed‑loop hydroponic cultivation: maintaining a stable nutrient solution despite continuous plant uptake and complex chemical interactions. Traditional management relies solely on periodic measurements of electrical conductivity (EC) and pH, followed by the addition of a pre‑mixed concentrated stock solution. While simple, this approach cannot control the depletion of individual ions and ignores the myriad precipitation and complexation reactions that occur in the solution. Recent attempts to measure individual ion concentrations and supply only the deficient ones improve specificity but still depend on heuristic, titration‑like cycles—adding a small amount of a component, measuring, and repeating—making the process time‑consuming and still blind to the underlying nonlinear chemistry.
To overcome these limitations, the authors develop a continuous network model of the nutrient solution, representing each chemical species (major cations such as K⁺, Ca²⁺, Mg²⁺; major anions such as NO₃⁻, PO₄³⁻, SO₄²⁻) as nodes and each possible precipitation or complexation reaction as directed edges. The dynamics of every node are expressed by ordinary differential equations (ODEs) that incorporate reaction rate constants, reaction orders, temperature and pH dependencies, as well as plant uptake terms modeled with Michaelis‑Menten kinetics. The general form of the ODE for species i is:
dC_i/dt = Σ_j (ν_ij·k_j·Π_m C_m^{α_jm}) – Σ_l (ν_il·k_l·Π_n C_n^{β_ln}) + I_i – R_i
where C_i is the molar concentration, ν_ij the stoichiometric coefficient, k_j the kinetic constant, I_i the external addition rate, and R_i the plant uptake rate.
Parameter identification proceeds in two stages. First, controlled laboratory experiments isolate each ion, record its temporal concentration profile using ion‑selective electrodes, and capture the effect of temperature and pH. Second, a nonlinear least‑squares optimization jointly fits the kinetic constants and uptake parameters across all measured species, while enforcing solubility limits as constraints to prevent unrealistic supersaturation.
Model validation uses a 30‑day long closed‑system trial with lettuce grown under typical commercial conditions. The model predicts individual ion concentrations with an average absolute error of 2.5 % and total dissolved solids (TDS) within 3 % of the measured values. Notably, the model correctly anticipates calcium phosphate precipitation that would otherwise reduce calcium availability; by adjusting the feed composition based on model output, the authors achieve an 80 % reduction in precipitate formation compared with a conventional EC‑based regimen.
A reverse‑calculation module translates a desired nutrient profile into the exact amounts of stock compounds required. This is formulated as a mixed‑integer nonlinear programming problem that minimizes total added mass while respecting solubility constraints and avoiding over‑addition of any component. The resulting feed plan can be generated automatically, allowing growers to specify target concentrations (e.g., K⁺ 200 ppm, NO₃⁻ 150 ppm) and receive precise dosing instructions for compounds such as K₂SO₄, Ca(NO₃)₂, and MgSO₄.
The authors acknowledge current limitations: the model presently covers 15 major ions and eight precipitation reactions, whereas real‑world hydroponics also involve micronutrients (Fe, Mn, Zn) and organic acids that can further complex the chemistry. Moreover, the present implementation lacks a real‑time feedback loop that would adjust kinetic parameters on‑the‑fly as temperature, pH, or plant growth stage changes. Future work is proposed to integrate continuous sensor streams, adaptive parameter estimation, and automated control hardware, moving toward a fully “smart” nutrient solution management system.
In summary, the ODE network model provides a rigorous, predictive framework for nutrient solution management, dramatically improving accuracy over heuristic titration methods, reducing waste of water and chemicals, and laying the groundwork for next‑generation, data‑driven hydroponic cultivation.
Comments & Academic Discussion
Loading comments...
Leave a Comment