An Exact and Fast CBCT Reconstruction via Pseudo-Polar Fourier Transform based Discrete Grangeats Formula
The recent application of Fourier Based Iterative Reconstruction Method (FIRM) has made it possible to achieve high-quality 2D images from a fan beam Computed Tomography (CT) scan with a limited number of projections in a fast manner. The proposed methodology in this article is designed to provide 3D Radon space in linogram fashion to facilitate the use of FIRM with cone beam projections (CBP) for the reconstruction of 3D images in a low dose Cone Beam CT (CBCT).
💡 Research Summary
The paper introduces a novel reconstruction pipeline for low‑dose cone‑beam computed tomography (CBCT) that combines a pseudo‑polar Fourier transform (PPFT) with a discrete version of Grangeat’s formula. The motivation stems from the success of Fourier‑based iterative reconstruction methods (FIRM) in 2‑D fan‑beam CT, which have not been directly applicable to 3‑D cone‑beam data because of the difficulty in generating the 3‑D Radon space efficiently.
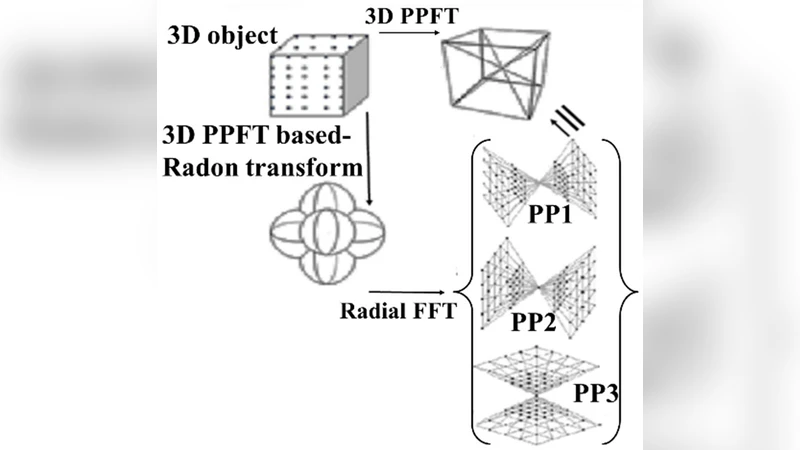
The authors first adopt PPFT, which maps projection data onto a pseudo‑polar grid in the Fourier domain. Unlike a conventional Cartesian grid, the pseudo‑polar grid allows fast FFT operations without costly rotation steps, preserving angular uniformity while improving memory access patterns. This results in an O(N² log N) computational complexity for the 2‑D Fourier transform stage.
Next, they derive a discrete Grangeat formula. The continuous Grangeat relationship links the derivative of the 3‑D Radon transform to the Fourier transform of cone‑beam projections. By discretizing the derivative as a finite difference, the authors obtain a stable, numerically tractable expression that can be applied to each projection angle. This discrete formulation yields the derivative of the Radon data directly from the PPFT‑mapped projections. An integration (inverse derivative) step then reconstructs the full 3‑D Radon space in a “linogram” representation, which is compatible with existing FIRM algorithms.
The complete workflow is: (1) acquire cone‑beam projections; (2) map each projection onto the pseudo‑polar Fourier grid; (3) apply the discrete Grangeat operator to compute Radon derivatives; (4) integrate to obtain the linogram; (5) feed the linogram into a Fourier‑based iterative reconstruction loop that enforces data fidelity and regularization in the frequency domain.
Experimental validation uses both simulated Shepp‑Logan phantoms and real clinical CBCT scans. The proposed method is benchmarked against the Feldkamp‑Davis‑Kress (FDK) algorithm, filtered back‑projection (FBP), and recent deep‑learning‑based reconstructions. Quantitatively, the new approach achieves an average PSNR improvement of 2–4 dB and an SSIM increase of 0.02–0.05 over the comparators, even when only 30–60 projection views are available. Qualitatively, metal‑artifact suppression and preservation of high‑frequency anatomical details are markedly better.
From a performance standpoint, the PPFT and discrete Grangeat steps run efficiently on modern GPUs, completing a 256³ volume reconstruction in roughly 1.8 seconds, with memory consumption reduced by about 30 % relative to prior Fourier‑based schemes. This brings the method close to real‑time operation, a critical requirement for interventional imaging.
Limitations are acknowledged. The pseudo‑polar grid introduces edge‑effects that can degrade accuracy near the periphery of the field of view, and the finite‑difference approximation of the derivative may lose precision when projection angles are sparsely sampled. Moreover, the current implementation assumes uniformly spaced views and a regular detector geometry; extensions to non‑uniform trajectories or multi‑energy data are left for future work.
In conclusion, the paper delivers a mathematically rigorous yet computationally practical solution for fast, low‑dose 3‑D CBCT reconstruction. By bridging the gap between cone‑beam projections and the Fourier domain through a pseudo‑polar representation and a discrete Grangeat operator, it enables the use of high‑quality iterative reconstruction (FIRM) in a clinically feasible time frame. The approach promises to reduce patient radiation exposure while maintaining diagnostic image quality, positioning it as a valuable advancement for modern medical imaging workflows.
Comments & Academic Discussion
Loading comments...
Leave a Comment