A Clinical and Finite Elements Study of Stress Urinary Incontinence in Women Using Fluid-Structure Interactions
Stress Urinary Incontinence (SUI) or urine leakage from urethra occurs due to an increase in abdominal pressure resulting from stress like a cough or jumping height. SUI is more frequent among post-menopausal women. In the absence of bladder contract…
Authors: ** - Mojtaba Barzegari¹ - Bahman Vahidi¹* (교신 저자) – bahman.vahidi@ut.ac.ir - Mohammad Reza Safarinejad² ¹ Division of Biomedical Engineering, Department of Life Science Engineering, Faculty of New Sciences
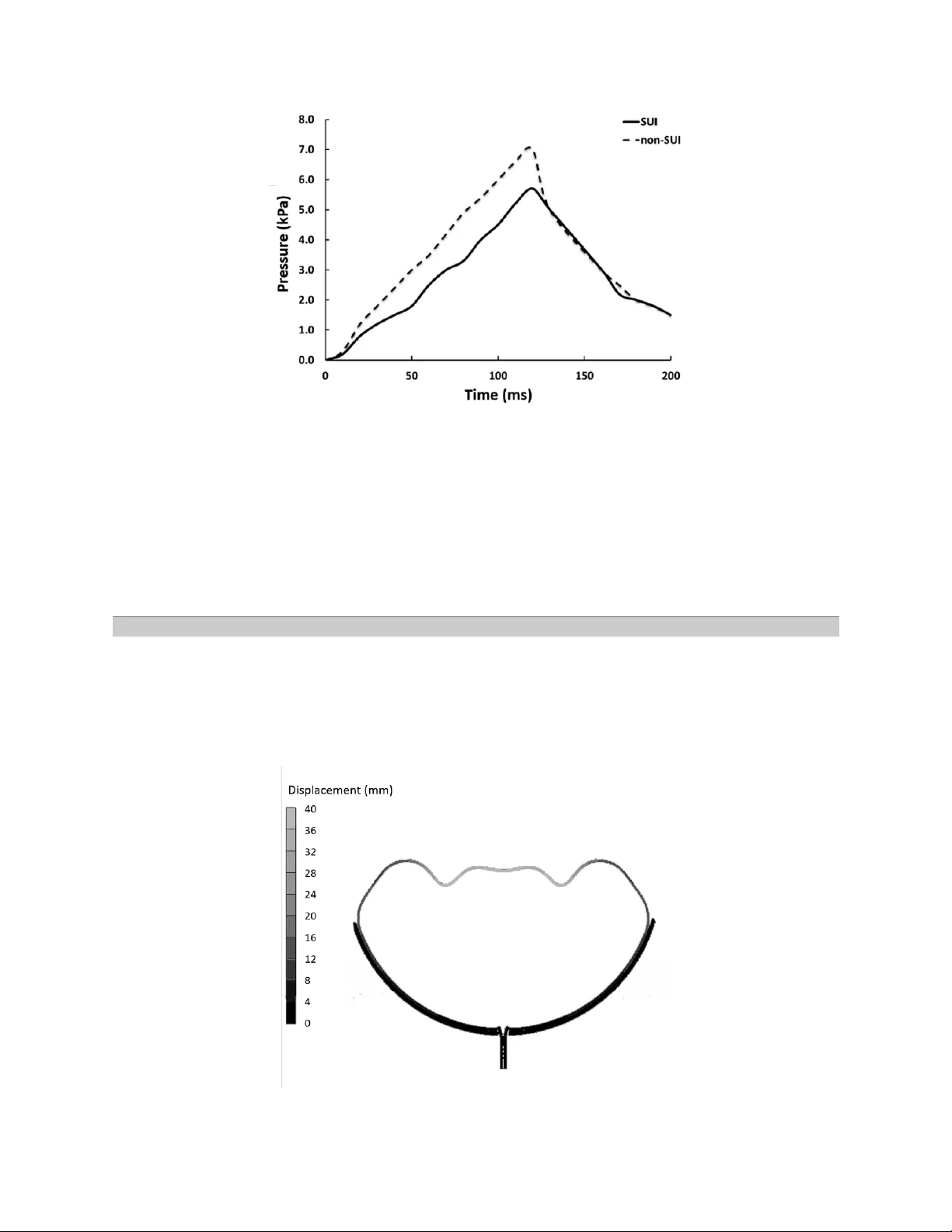
A Clinical and Finite Elements Study of Stress Urinary Inconti nence in Women Usin g Fluid-Structure Int eractions Mojtaba Barzegari 1 , Bahman Vahidi 1 *, Mohammad Rez a Safarinejad 2 1- Division of Biom edical En gineering , Departm ent of Life Science E ngineering, Faculty of New Sciences and Techno logies, U niversity of Teh ran, Tehran, I ran 2- Clinical Center fo r Urolog ical Disease D iagnosis and Private Clinic S pecialized in Urological and Andrological Ge netics, Tehran, I ran * P.O.B. 1439957131, Tehran, Iran, bahman.vahidi@ut.ac.ir Abstract Stress U rin ary I ncontinence (SUI ) or ur ine leak age from urethra occu rs due to an increase in abdominal pressure resulting from st ress l ike a cough or j umping hei ght. SUI i s m ore f requent among post- menopausal wom en. In t he absence of bladder contraction, vesical pressure exc eeds from urethral pressure leading t o urine leakag e. Despit e a lar ge number of patients diagnosed with this problem, few studies have investigated its function and mechanics. T he main goal of this study is to model bladder and urethra com putationally under an e xternal pr essure like sneezing. Finite Elem ent Method and Fluid- Structure I nteractions are utiliz ed for simulation. Lin ear mechanical p roperties as signed to the bladder and urethra and pressure boundary conditions are indispensable in this model. T he results show good accordance between the clinical data and predicted values of the computational models , such as the pressure at the center of the bladder. This indicates t hat numerical methods and simplified physics of biological systems like inferior urinary tract are helpful to achieve the results similar t o clinical res ults, in order to investig ate patholog ical conditions. Key words : Computational Fluid Dynamics, Urinary Tract, Stress Urinary Incontinence, Finite Element Method, Fluid- Structure Interacti on Introduction Recently, diseases associated w ith ur ine and g enital tract, with the general term urolog y, are more prevalent among both men and women older than 40 years old. The most critical subset of these problem s is urinary incontinence [1]. Stress Urinary Incontinence is very common among women, which occurs due to a mechanica l pressure like sneezing or jumping height. In this type of incontinence, any ac tivity that increases abdominal pressure (i ncluding laughing, coughing, sne ezing, and straining) l eads to urine leakage as a result of urethral sphincter weakness. R especting to the published reports on 2001, costs of urinary incontinence treatments exceeded 16.3 billion US dollar. The reports indicate 75% of total costs were spent on diag nosis and treatm ent for wom en [1]. SUI is m ore prevalent in women [1]. An incr ease in abdom inal pressure i n t he absence of bladder contraction raises the vesical pressure to a level that exceeds urethral pressure, leading t o involuntary loss of urine which mainly characterizes SUI. Abdo minal pressure increases due to a mechanical incident like laughing, sneezing, jumping height, or any other t ension i n the body [1] . This explains why the SUI is considered as a m echanical force. Although the main reason of SUI remains unknown, a large num ber of physicians believe that SUI is caused by injuries to the pelvic fl oor neuro-m usculature, which m ainly happen among those who have given birth vaginally [1]. While not a life thr eatening condi tion, SUI can detrimentally impact the quality of life. Up to now , biomechanical studies over female incontinen ce are dominated by three theories ; including pressure transmission theory pr oposed by Enhoring [ 2], the integral theory proposed by Petrus and Ulmestan [3], and the hammock theory pr oposed by Delancy and Ashton -Miller [4]. Moreover, all these three theories seem par adoxical and within themselv es, ther e is no consistency about involved structures and tissues. On the other hand, analytical studies and organic structures’ modeling is a pract ical way to study biom echanica l phenom ena. In the cur rent study, complications of the numerical model could challenge the researcher. In the current paper , t he urine flow in the urinary tract is investigated. It is more common to i nvestigate a part of urinary tract since scrutinizing all parts is very complex and time -consum ing. Here, the m odeling is mainly focused on bladder and urethra. The preferred computational parameter is t he fluid (urine) pressure which is com pared to clin ical data. Pressu re is t he considered parameter sinc e it is available i n urodynamic measurem ent and it has varied m easuring m ethods. There are few reports of fi nite element analysis ab out SUI. Kim proposed a 2-dim ensional model to study urethral closure during stress [5]. Kim pr oposed an axisymm etric model of the urethra and pelvic floor to find proper dimensions for his model. In Kim’s model, a catheter was placed in the urethra. This model was developed to analyze active (muscle contraction) or passive (pressure transm ission) contributions to urethral closure. The results showed t hat an active contraction of sphincter muscle and pelvic floor play an important r ole in urin e continence [5]. T he findings showed levator ani’s con traction and its connectivity to endo pelvi c fasc ia contribute in urine continence [5]. Zhang et al. developed a rather complex finite elem ent model of t he urin ary tract and the pelvic floor in athlete women while j umping height to examine feasibility and repeatability of finite element model for urinary i ncontinence [6]. This model included an ideal modeling of the intestine, vagina, rectum, pelvic diaphragm , uri nary diaphragm, bladder and abdominal m uscles. Although urine leakage was not clearly defined, it was proved that a finite element m odel of SUI in women, while they are jumping high, could reprod uce the clinical dat a. Haridas et al. developed a finite element model to study biom echanical characteristics of the inferior urinary tract and the pelvic floor [7]. Their premier investigations were related to the finite elem ent modeling of the vag ina, bladder, ute rus, bladder, levator an i, endo pelvic fasc ia and rectum derived from a specific MRI. A good coherence was observed between the modeling results and t he ultrasound imag es. In the latest studies, Spirka et al. developed a finite element model of stress urinary i ncontinence [ 8]. The most cr itical limitation of this model was employing a quasi-solid m odel f or the fluid. To verify the model, they used limited urody namic data. In this study, bladder reaction to an external force ( abdom inal pressure resulting f rom a cough) is studie d in both physiological and pathological conditions. To achieve t his goal, two different computational models are c onsidered to investigate urine dynamics in the bladder. Fluid-Structure Interaction s ( FSI) method is utilized in the com putational m odels. The required data for the a ssumption of the sim ulation are extracted from urodynam ic measurements. In comparison to the importance of the considered material properties for tissues, the geometry of urinary tract i s less i mportant. So simplified models are considered in this study . The current study works on extending previous studies to model mechanically t he inferior urinary tract, besides validating model through comparing to clinical and urodynam ic data. It i s mentioned before t hat the preceding models we re focused on one mechan ism of urinary continence or they modeled urine leakage. This study is focused on the parameters contributed t o leakage and compares physiological and pathological case s. Another fea ture is em ploying FSI method to improve the reliability of results. Methods Here, t he Stress Urinary Incontinence is studied ; collected clinical data ar e di vided into pathological and physiological groups. Delibera te sneez ing during urody namic m easurements is a com parison criterion between clinical and analytical data. In this study, a cough and its i mpact on the i nferior urinary tract in both physiologic al and pathological groups are studied. To achieve this goal, two computational model s are develop ed : one without support structure as a pathological one and the other with a support structure which prevents leakage from the bladder outlet resembling physiological groups. The external force is sneezing which occurs in 200 ms. Both models use linearly elastic material properties. The measured parameter for verification is pressure. Total time of the simulation is 200 ms. The i nitial external pressure is zero at t he i nitial state and remains zero f or 10 ms . T hen, the pressure increases to its maximum level (which corresponds to urody namic measurem ents) and gradually decreases to the i nitial l evel. During the process, the pressu re in the center of the bladder c hanges with tim e. To model both pathological and physiological conditions, two different geometries are considered. In the physiological model, pelvic diaphragm closes urethra and p revent urine leakage. In t he pathological model, the suppo rt structure does not exist, leading to bladder d islocation which prevents ure thral closur e. For geometry and boundary conditions, data were collected from the continent case for the physiolog ical condition and the incontinent case for the pathological condition . For geom etry construction, bladd er capacity is considered and data for both boundary conditions and results validation include abdominal pressure and bladder pressure. Also, t he sensitivity of the model to different param eters is considered to verify analytical da ta. Inferior urinary tract g eometry includes the bladder, urethr a and the support structure. I n the physiological model, support structure exists, wh ile in the pathological model, i t is dislocated and it is upper than th e bladder outle t so it has no impact on urine continence. So , it is eliminated from the pathological model. Figure 1 and 2 sh ow patholog ical and physiolog ical models, respect ively. Fig. 1 Computational geometry for pathological mode Fig. 2 Computational geometry for physiologica l model Pathological and physiolog ical vesical capa cities are 346cc and 410cc, respectively. Since b ladder geometry is presumed as a sphere, its diameter changes from 43 mm to 46 mm f or the patho logical and physiological model. Sean et al. studied urodynam ic measurement of 42 women and concluded that average thickness of bladde r wall with 200cc of capa city would be 1.7 mm [9]. Similar findings presum ed that the t hickness of bladder wall is approximately 0.9 in 56cc of capacity [5, 10]. With respect to the previous findings [10], to simplify meshing process and fluid and solid net coherence, the bladder wall thickness is presum ed 1.5 m m in both conditions . The urethra is modeled as a cylindrical tube [11, 12]. In both models, the urethra is relatively cl osed and fluid flow would increase its diameter. Since the purpose of this study is t o calc ulate the vesical central pressure, the length of the urethra is not important. T he model i s axisymm etric ; so the bladder outlet does not have a cruciate form, but boundary conditions are applied in a way that accurately models bladder outlet during urin ation. In the physiological m odel, urethra passes through a support structure and this struc ture sup ports the bladder. The support structure is modeled like a bowl- shape based on its anatomy [13, 14] . Janda et al. reported that pelvic diaphragm supporting bladder is an oval-shaped with 140mm of log axis dim ension and 122 mm of short axis dimension [15]. T o further simplify calculat ions, a circular bar was utilized with the radius taking the average of the 2 mentioned values, 131mm and with 2mm of thickness similar to levator ani’s thick ness [ 16 ]. The Eulerian kinem atic description is proposed for fluid flow. A large enough f luid region is considered which encompasses all Lag rangian details. The bladder is filled with u rine. Since each case is asked to sneeze deliberately duri ng urination, the bladder is not completely f ull at the time of sneezing. Figure 3 shows the final com putational m odel of the phy siological blad der . Fig. 3 Final co mputational model for p hysiological condition The abdominal pressure in urodynamic measurements resulti ng from sne ezing i s t he most important boundary condition which is applied to the top hemisphere of the bladder. It is better to apply pressure gradually. For the first 10 seconds, the pressure remains zero. Then in the f ollowing 200 seconds, the pressure reaches its maximum level and decreases to zero l evel again. Rising and falling i n pr essure level is to model sneezing. The maxim um pressur e is derived from urodynam ic measurem ents. In bot h pathological and physiological conditions, the abdomina l pressure during deliberate sneezing is 50 cm H 2 O (4.9 k Pa ). In order to discretize the model, a structured mesh is employed. The Lagrangian mesh including bladder, urethra and support structure (exc lusively for the physiological model) is unstructured quadrilateral, whil e thanks to sweep method, they are homog eneously dispersed in this region. The Eulerian mesh description includes 568 elem ents and the Lag rangian mesh de scription con tains 32400 e lements. In continuum m echanics, material properties dictate mechanical behavior. An interpolated function describes the field variabl e in each element. Usually, the relation be tween deformation and ex ternal force s are experimenta lly derived and is expressed as str ess -st rain equation [17, 18 ]. If genital and urinary tissues presumed linearly elastic, their elastic modulus can be extracted from Table 1 [19]. Table 1. Elastic prop erties of genital and lower urinary tract tissues (P oisson ratio = 0.45) [19] Tissue Elastic modulus [MPa] Density [kg/m 3 ] Bladder 0.05 1030 Urethra 3 1030 Uterus 0.05 1030 Vagina 0.005 1030 Rectum 0.1 1030 Intestine 0.1 1030 Muscle 2.4 1040 Pelvic Floor 1.2 1030 Ligam ent 1.2 1030 The current st udy presumed the bladder, the urethra and the support structure as linearly elastic materials. Table 1 reports the constants of the linear equat ions. The urine flow was modeled l inearly at first, but there was no accordanc e with the clinical results. So , a polynom ial equation was em ployed to describe the ur ine f low. Urin e densi ty i s 1g/cm 3 [8]. I n l inear modeling w ith neglecting thermal effects, the o nly incorporated p arameter is shear stress m odulus is 6×10 6 k Pa for urine. In the representa tive polynom ial equation of fluid flo w, the pressure is derived f rom equation (1 ): Eq. (1) µ stands f or compression, ρ 0 is density at P 0 , e internal force per mass unit and other constants are water constants which a re present ed in Tabl e (2). Table 2. Urine poly nom ial model constant s [8] Parameter Value In order to model SUI, i t is not sufficient to model only bladder and urethra. It is important to consider urine fl ow. Abdominal pressure increases flow pressure and as a result, urine exerts pressure on the walls of the bladder and the urethra. Fluid-structure Interaction s is indispens able in computations. The shear stress at t he i nterface leads to solid deform ation o r dislocation which impact s fluid flow [20-22]. To analyze this condition, the A rbitrary-Lag rangian-Eulerian equation is used, s ince it is simple and accu rate . In this method, Euler equation is employed for fixed boundaries (fluids), Lagrang e equation for moving boundaries (solids) and the Euler-Lag range equation for the interactions [ 23 ]. Here, since sneezing is a quick dynam ic incident, explici t method is em ployed. ANSYS AUTODYN is used to analyze the model and solve its equations. Initial pressure in Euler region is zero. T o solve fluid and sol id equations together, FSI and Arbitrary- Lagrangian-Eulerian equat ion is employed. Penalty method is employed after re -m eshing the def ormed or dislocated r egion to sa ve t ime. Therefore, the coupling of two Lagrangian and Eulerian methods automatically start. Self-interaction is activated in Lagrange region and its tolerance is 0.2. Maximum time of sol ving is 200 ms and the allowe d tolerance of energy error is 4. Safety factor i n time steps is 0.65. Strain rate in the Eulerian region i s calculated based on the we ight method, and Eulerian pressure is calcula ted based on the mean method. Also, mass scaling method is deact ivated. The s imulations were performed on a q uad-core CPU with 6GB RAM com puter. Results The comparison between pressure-time diagram s with respect to quantitative results is t he most important part. Figure 4 shows the sim ulated pressure changes for patho logical and phy siological conditions. Fig. 4 Pressure in the ce nter of bladder in ter ms of time compared to physiologic al and pathological models Table 3 presents the computed maximum exter nal pressure during sneezing and urodynamic measurem ents and compares the re sults. Table 3. Com parison of clinica l bladder pressur e with sim ulation result s during a cough Model Real pressure (Pa) Simulated pressur e (MPa) Error percentage Physiological 6962 7070 1.3 Pathological 5785 5712 1.2 Bladder wall dislocation contour is drawn to study vesical deform ations and is compared to clinical data. Figure 7 and 8 represents bladder wall deformation contour for the physiological and pathological model, respectively. Fig. 5 2 D Postprocessing of deformation contour i n physiological model simulation (displace ments in mm) Fig. 6 2 D Postprocessing of deformation contour i n pathological model simulation (displace ments in mm) Discussion Although several studies have investigated bladder and uret hra mechanical behavi or, t heir results focused on bladder filling or emptying [10, 12, 24-32]. Few studies focused on external forces and their impact on bladder condition. Zhang et al. published the most reliable results in the field of str ess impact on th e bladder [6]. Th at study m odeled the i nvolved organs during the j umping of height and presum ed urine leakage occur as t he urine enters the urethra. In their reported r esults, only the upper part of the urethra was filled with urine, while i n the current study in the patholog ical condition, the support structure is missed and the urine fills the urethra com pletely which indicates that this criterion is not an appropriate criterion for urine leakage, even though urine leakage is not the pr eferred parameter in the present study. Similar to Zhang model [6 ], the current developed model did not simulate urine transmission t hrough the urethra. Limitations in hardware r esources and time-consuming processes l ead to these defects. Although Zh ang et al. studied 2 different stress incidents, our qualitative i mag es of modeled bladder deform ation match per fectly with Zhang et al. results [6]. Images show that elem ents of th e upper hemisphe re are deformed and di slocated m ore than other e lements. The le ast dislocation o ccurs in the inferior par ts. Kim et al. inves tigated the cough impact on a 2-dimensional model of the bl adder and studied pressure transmission t heory. The main features of the c urrent study ar e analytical methods and computa tional techniques which differentiate it from other studies. On the other words, abdominal pr essure is modeled as a dynamic impact inserted on the superior part of bladder and bladder central pressure is calculated in the model. Kim added up abdominal pressure to bladder central pressure and bladder inner walls [5]. Kim also considered li near el astic properti es. Kim’s validation cri terion is bladder and support struc ture deformation. The abdominal pressure i n K im’s study is relatively equal ed to clinical ly measured pressure for the pathologica l conditions in the cur rent study. Maxim um deform ation of the curre nt study is reported 2cm (Figure 6 ) wh ich acco rds with K im results [5]. Respecting to the diagram shown in Figure 4 , bladder i nternal pressure in the physiological condition is more than the pathological one, which match es perfectly with uro dynam ic measurements. The patient diagnosed with SUI has no cont rol on urethral pressure, s o any external stress to bladder leads to urine leakage and bladder central pr essure reduction. Whil e in physiological conditions, the pressure inside t he bladder increases, since sup port structure prev ents urine leak age. Table 3 verified quantitative simulations in the current study. I t determines that the model could be verified through comparison of simulated and clinically measured data. Noise i n clinical data because of environmenta l conditions and sen sor accuracy is inevit able but sim ulation results are free from any noise. For further st udies, it is proposed to apply more rec ent FSI techniques and to define variable meshing in the Eulerian r egion to increase accuracy. Boundary conditions should be defined in a way that do es not limit the natural activity of bladder and is more similar to the real situation. It is also proposed to utilize MRI i mages for geometry construction to im prove the reliability of the results, in order to predict and differentiate path ological and phy siological cond itions. Conclusion The current study developed two models of stress urinary i ncontinence for physiological and pathological conditions to study urine dynamic flow in bladde r under abdominal pressure resulting f rom sneezing. Developed model and i ts validations show analytical methods and simplifying biological systems, lik e inferior urine tract, would successful ly simulate t he clinical results in a virtual environm ent. It shows that the investigation s of biolog ical systems do not necessar ily require exp erimenta l sets. Although FS I method deploys complex and time-consum ing calcul ations, it is very practical and accurate in modeling coupled systems incl uding both solid and fluid phase. Results indicate maximum deformation in bladder occurs when the internal pressure is reaching its maxim um level. For further studies, it is proposed to utilize more recent FSI t echniques, more accurate boundary conditions and geometry construction based on MRI im ages, in order to pred ict improved resul ts in patholog ical and physio logical cond itions. References [1] L. Wilson, J. S. Brown, G. P. Shin, K .-O. Luc, and L. L. Subak, "Annual Direct Cost of Urinary Incontinence," Ob stetrics & Gynecology, vol. 9 8, pp. 398- 406, 2001/09 2001. [2] G. Enhorning, E. R. Miller, and F. Hinm an, "Sim ultaneous recording of intravesi ca l and intraurethral p ressure," Acta Chirurgi ca candinavica, vol. 276, pp. 1- 68, 1961. [3] P. E. P. Petros and U. I. Ulmsten, "AN INTEGRA L THEORY OF FEMALE URINARY INCON TINENCE," Act a Obstetricia et G ynecologica Scandinavica, vol. 69, pp. 7-31, 1990/01 1990. [4] J. A. Ashton- Miller and J. O. L. Delancey, "Functional Anatomy of the Fem ale Pelvic Floo r," Annals of the New York Ac ademy of Sciences, vol. 1 101, pp. 266- 296, 2007/02/15 2007. [5] K. J. Kim, "Biomechanical Analyses of Female Stress Urinary Incontinence," Ph .D., University of Michigan, 1994. [6] Y. Zhang, S. Kim, A. G. Erdman, K. P. Roberts, and G. W. Timm, "Feasibility of Using a Computer Modeling Approach to Study SUI Induced by Landing a Jump," Annals of Biomedical Engineering, vol. 37, pp. 1425- 1433, 2009/05/05 2009 . [7] B. Haridas, H. Hong, R. Minoguchi, S. Owens, and T. Osborn, "PelvicSim - A computational experimental system for biomechanical evaluation of female p elvic floor organ disorders an d associated minimally invasive interventions," Studies in Health Technology and Informatics, vol. 119, pp. 182- 187, 2006. [8] T. Spirka, K. Kenton, L. Brubaker, and M. S. Damaser, "Effect of Material Properties on Predicted Vesical Pressure During a Cough i n a Simpl ified Computation al Model of the Bladder and Urethra," Anna ls of Biomedica l Engineering, vol. 4 1, pp. 185-194, 2012 /08/21 2012. [9] L. Chan, S. The, J . T itus, and V. Tse , " P14.02: The value of bl adder wall thickness measurement in t he assessment of overactive blad der syndrome," Ultrasound in Obstetrics and Gynecology, vol. 26, pp. 460- 460, 2005/09/08 2 005. [10] M. S. Damaser and S. L. L ehman, "The effect of urinary bladder shape on its mechanics during filling," Journal of B iomechanics, vol. 28, p p. 725-732, 1995 /06 1995. [11] K. A. Backman, "Effective urethral diameter," in Hydrodynamics of Micturition , C. C. Thomas, Ed., ed IL: Springf ield, 1971, pp. 250- 256. [12] R. A. Hosein and D. J. Griffiths, " Compute r simulation of the neural control of bladder and urethra," Neurourolog y and Urodynami cs, vo l. 9, pp. 601-618, 1990. [13] R. L. Drake, W. Vogl, and A. W. Mitchell, Gray’s Anatomy for Students . New York: Elsevier, 2005. [14] F. H. Netter, Atlas o f Huma n Anatomy . Philadelph ia: Saunde rs Elsevier, 2006 . [15] Š. Janda, F. C. T. van der Helm, and S. B. de Blok, "Measuring morphological parameters of the pelvic floor for finite element modelling purposes," Journal of Biomechanics, vol. 36, pp. 749- 757, 2003/06 2003. [16] D. d'A ulignac, J. A. C. Martins, E. B. Pires, T. Mascarenhas, and R. M. N. Jorge, "A shell finite element model of the pelvic f loor muscles," Computer Methods in Biomecha nics and Biomedical Engineering, vol. 8, p p. 339- 347, 2005/10 2005. [17] S. S. Rao, " Overv iew of Finit e Element Method," i n The Finite Element Method in Engineering , ed: Elsevier, 200 5, pp. 3-49. [18] H. Saunders, "Book Reviews : THE FI NITE ELEMENT METHOD FOR ENGINEE RS K . E. Huebner John Wiley & Sons - New York (1975)," The Shock and Vibration Digest, vol. 9, pp. 39-39, 1977/02/01 1977. [19] H. Yam ada, Strength of Biological Materia ls . Baltimore: Will iams and Wilkins, 1970. [20] E. K uhl, S. Hulshoff, and R. de Borst, " An arbitrary Lagrangian Eulerian finite -element approach for fluid-structure interaction phenomena," International Journal for Numerical Methods in En gineering, vol. 57, pp. 1 17- 142, 2003. [21] E. J . Weinberg and M. R. Kaazem pur Mofrad, "Transient, Three-dimensional, Multiscale Simulations of the Hum an A ortic Valve," Cardiovascular Engineering, vol. 7, pp. 140-155, 2007/11/16 2007. [22] M. Souli, A. Ouahsine, and L. Lewin, "ALE formulation for fluid – structure interaction problems," Computer Methods in Applied Mechanics and Engineer ing, v ol. 190, pp. 659-675, 2000/11 2000. [23] J. Donea, A. Huerta, J. P. Ponthot, and A . Rodrï¿½ï¿ ½guez-Ferran, "Arbitrary Lagrangian- Eulerian Methods," in Encyclopedia of Computational Mechanics , ed: John Wiley & Sons, Ltd, 2004. [24] D. J. Griffiths, "Urethral el asticity and m icturition hydrodynamics in females," Medi cal & Biological Engine ering, vol. 7, pp. 201- 215, 1969/03 1 969. [25] A. Spángberg, H. Terió, A. Engberg, and P. Ask, "Quantification of urethral funct ion based on Griffiths' model of flow through elastic tubes," Neurourol ogy and Urodynamics, vol. 8, pp. 29- 52, 1989. [26] E. H. C. Basti aanssen, J. L. van Leeuwen, J. Vanderschoot, and P. A. Redert, " A Myocybernetic Model of the Lower Urinary T ract," Jour nal of Theoretical Biology, vol. 178, pp. 113-133, 1996/01 1996. [27] E. H. C. Bastiaanssen, J. Vanderschoot, and J . L. v an Leeuwen, "State -space Analysis of a Myocybernetic Model of the Lower Urinary Tract," Journal of Theoretical Biology, vol. 180, pp. 215-227, 1996/06 1 996. [28] D. J. Griff iths, "Hydrodynam ics of m ale m icturition — I Theory of st eady flow through elastic- walled tubes," Med ical & Biological Engineering, vol. 9, pp. 581-588, 1971 /11 1971. [29] M. Horák and J. Křen, "Mathem atical model of the m ale urinary tract," Mathemat ics and Computers in Simu lation, vol. 61, pp. 573-581, 2003 /01 2003. [30] R. van Mastrigt and D. J . Griffiths, "An evaluation of contracti lity parameters determined from isometric contractions and micturition studies," Urological Research, vol. 14, pp. 45-52, 1986/02 1986. [31] J. R. Fielding, D. J. Griffiths, E. Versi, R. V. Mu lkern, M. L. Lee, and F. A. Jolesz, "MR imaging of pelvic floor continence mechanism s in the supine and sitting positions," American Journal of Roentgenology, vol. 1 71, pp. 1607- 1610, 1998/12 199 8. [32] H. T. Lotz, P. Remeijer, M. van Herk, J. V. Lebesque, J. A. de Bois, L. J. Zijp , et al. , "A model to predict bladder sha pes from changes in bladder and rectal filling," Medical Physics, vol. 31, pp. 1415-1423, 2004/05 /24 2004.
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment