Respiratory and cardiac monitoring at night using a wrist wearable optical system
Sleep monitoring provides valuable insights into the general health of an individual and helps in the diagnostic of sleep-derived illnesses. Polysomnography, is considered the gold standard for such task. However, it is very unwieldy and therefore not suitable for long-term analysis. Here, we present a non-intrusive wearable system that, by using photoplethysmography, it can estimate beat-to-beat intervals, pulse rate, and breathing rate reliably during the night. The performance of the proposed approach was evaluated empirically in the Department of Psychology at the University of Fribourg. Each participant was wearing two smart-bracelets from Ava as well as a complete polysomnographic setup as reference. The resulting mean absolute errors are 17.4 ms (MAPE 1.8%) for the beat-to-beat intervals, 0.13 beats-per-minute (MAPE 0.20%) for the pulse rate, and 0.9 breaths-per-minute (MAPE 6.7%) for the breath rate.
💡 Research Summary
This paper presents a non‑intrusive wrist‑worn optical system for night‑time monitoring of cardiac and respiratory activity. Using a commercial Ava smart‑bracelet equipped with dual‑wavelength (green and infrared) photoplethysmography (PPG) and a 3‑axis accelerometer sampled at 25 Hz, the authors extract beat‑to‑beat intervals (RR), heart rate (HR), and breathing rate (BR) during sleep. The study involved seven healthy young women (average age 21.1 years, BMI 20.0 kg/m²) who each spent four nights in a sleep laboratory, wearing two bracelets (one on each wrist) and a full polysomnographic (PSG) setup as reference. After quality control, 31 full‑night recordings were retained for analysis.
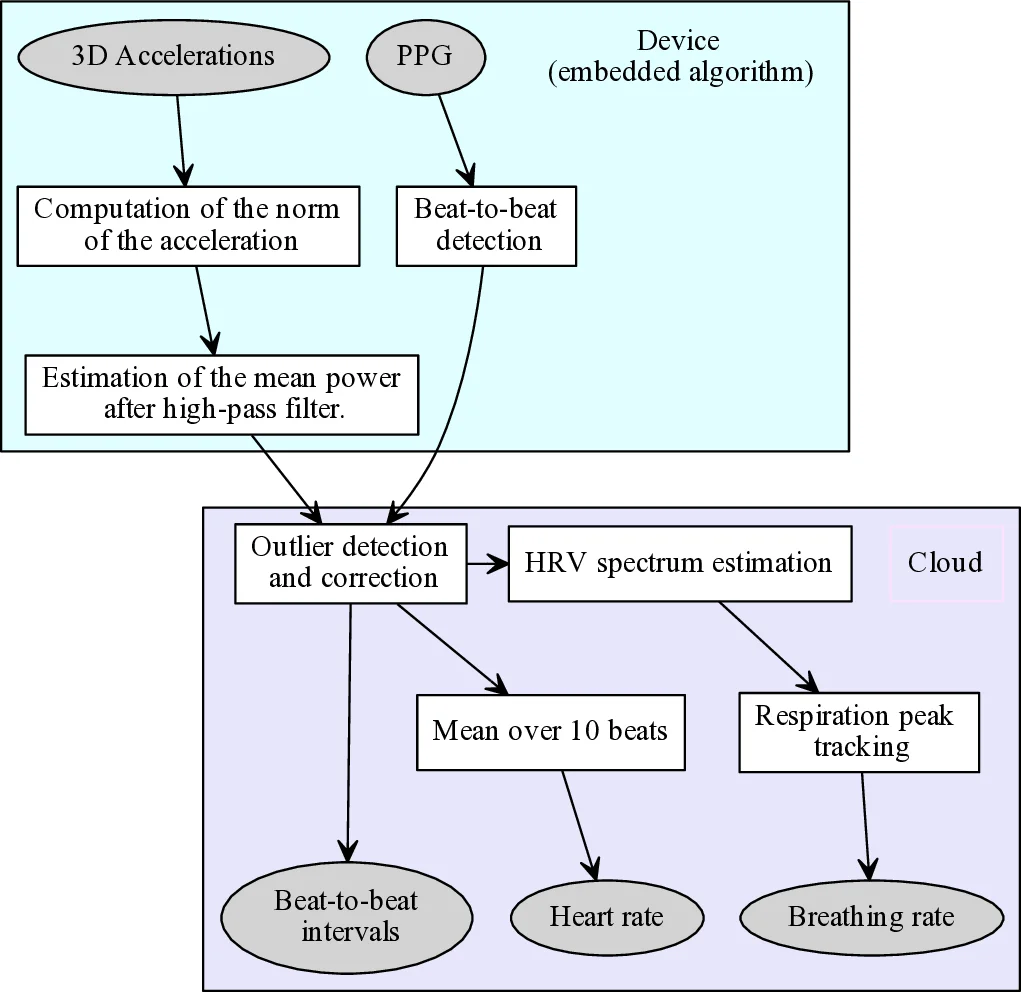
The algorithm is split between low‑power on‑device processing and a cloud‑based server. On the device, the first‑order derivative of the PPG signal is used to locate local maxima that correspond to pulse upstrokes. A motion indicator derived from the power of the high‑pass‑filtered acceleration norm flags periods where the optical signal is corrupted by movement. Physiological constraints (e.g., refractory period) discard implausibly short intervals, and any remaining outliers are corrected by linear interpolation. The corrected RR series is then transmitted to the server.
Server‑side processing resamples the RR series uniformly at 2 Hz, applies a band‑pass filter (0.04–0.5 Hz) to isolate autonomic nervous system (ANS) modulation, and fits a 20‑th‑order autoregressive (AR) model using a normalized least‑mean‑square (NLMS) adaptive algorithm. The HRV spectrum obtained from the AR model is examined in the respiratory frequency band (0.1–0.5 Hz, corresponding to 6–30 breaths per minute). The dominant peak frequency is taken as the instantaneous breathing rate, and a recursive update uses the ratio of the peak power to total band power as a learning gain.
Performance evaluation employed mean absolute error (MAE) and mean absolute percentage error (MAPE). Dynamic time warping (DTW) aligned the estimated and reference RR series; HR was derived from the inverse of ten‑beat averaged RR; BR was interpolated to the PSG respiration timestamps. Results showed a mean RR MAE of 17.4 ms (1.8 % MAPE), HR MAE of 0.13 bpm (0.20 % MAPE), and BR MAE of 0.9 breaths per minute (6.7 % MAPE). Approximately 75 % of the breathing‑rate estimates were within 1 cpm of the reference, and HR errors were consistently below 0.2 bpm.
The authors discuss several sources of error. Wrist‑based PPG measures the peripheral pulse wave, which is delayed and morphologically altered relative to the ECG R‑peak, limiting the fidelity of RR for high‑resolution HRV analysis. The embedded detection algorithm must operate under strict memory and computational constraints, leading to sub‑optimal pulse detection. Motion artifacts, even minor, can degrade signal quality despite the accelerometer‑based correction. Moreover, the indirect estimation of respiration from HRV assumes normal ANS modulation; pathological or elderly populations with altered autonomic control may require dedicated validation.
In conclusion, the study demonstrates that a wrist‑worn optical device can reliably estimate heart rate and breathing rate during sleep, provided the user remains relatively still. Beat‑to‑beat interval accuracy is sufficient for many applications but remains an approximation of ECG‑derived intervals. Future work will incorporate additional PPG features such as amplitude modulation and baseline wander, which carry further respiratory and autonomic information, and will extend validation to clinical cohorts.
Comments & Academic Discussion
Loading comments...
Leave a Comment