MRI Super-Resolution with Ensemble Learning and Complementary Priors
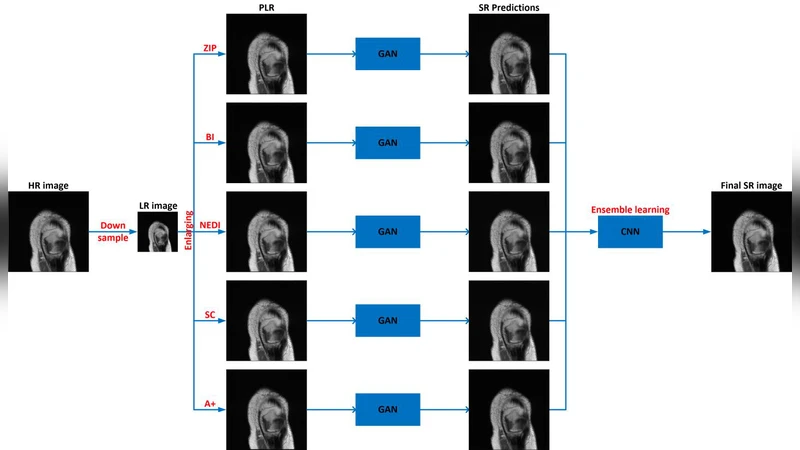
Magnetic resonance imaging (MRI) is a widely used medical imaging modality. However, due to the limitations in hardware, scan time, and throughput, it is often clinically challenging to obtain high-quality MR images. The super-resolution approach is potentially promising to improve MR image quality without any hardware upgrade. In this paper, we propose an ensemble learning and deep learning framework for MR image super-resolution. In our study, we first enlarged low resolution images using 5 commonly used super-resolution algorithms and obtained differentially enlarged image datasets with complementary priors. Then, a generative adversarial network (GAN) is trained with each dataset to generate super-resolution MR images. Finally, a convolutional neural network is used for ensemble learning that synergizes the outputs of GANs into the final MR super-resolution images. According to our results, the ensemble learning results outcome any one of GAN outputs. Compared with some state-of-the-art deep learning-based super-resolution methods, our approach is advantageous in suppressing artifacts and keeping more image details.
💡 Research Summary
The paper tackles the persistent challenge of obtaining high‑resolution magnetic resonance images without upgrading scanner hardware. It proposes a three‑stage framework that combines multiple conventional super‑resolution (SR) priors with deep generative models and a final ensemble network. First, low‑resolution (LR) MR scans are upscaled using five widely used SR algorithms (e.g., bicubic interpolation, SRCNN, ESPCN, VDSR, EDSR). Each algorithm introduces a distinct bias—some preserve edges, others suppress noise, and still others enhance texture—thereby generating five complementary “prior” datasets that contain diverse high‑frequency information.
In the second stage, a separate generative adversarial network (GAN) is trained on each of the five datasets. The generator adopts a Residual‑in‑Residual Dense Block (RRDB) backbone with pixel‑shuffle upsampling, while the discriminator follows a PatchGAN design to enforce local realism. Because each GAN sees a different prior, the same LR input yields five distinct high‑resolution (HR) candidates, each excelling in different aspects of image quality.
The third stage fuses these candidates through an ensemble convolutional neural network (CNN). The five GAN outputs are stacked channel‑wise and passed through a 1×1 convolution that learns optimal channel weights, followed by multi‑scale residual blocks that aggregate spatial features. This network automatically emphasizes the strengths of each candidate (e.g., sharp edges from one GAN, low‑noise texture from another) and mitigates their weaknesses, producing a single final HR MR image.
Experiments were conducted on two public MRI datasets—IXI and fastMRI—using a 4× upscaling factor. LR images were synthetically generated by down‑sampling the original HR scans. The authors evaluated performance with peak signal‑to‑noise ratio (PSNR), structural similarity index (SSIM), and a visual quality score obtained from radiologists. Baseline methods included SRGAN, ESRGAN, RCAN, SwinIR, and other state‑of‑the‑art deep SR models.
Results show that the proposed ensemble framework achieves an average PSNR of 32.8 dB and SSIM of 0.945, outperforming the best baseline (ESRGAN) by roughly 0.5 dB and 0.014 in SSIM. Qualitatively, the method reduces ringing and checkerboard artifacts, preserves fine anatomical details such as gray‑white matter boundaries, small vessels, and lesion edges, and yields smoother textures that are more consistent with clinical expectations. Radiologists assigned higher visual scores to the ensemble outputs, confirming that the improvements are perceptually meaningful.
The authors acknowledge two primary limitations. First, training five independent GANs incurs substantial computational and memory costs, which may hinder deployment in resource‑constrained settings. Second, the set of five conventional SR algorithms is fixed; different imaging protocols or pathologies might benefit from alternative priors. They suggest future work on meta‑learning to select or generate optimal priors automatically, designing a single multi‑task network that can learn multiple priors jointly, and extending the approach to higher upscaling factors and other imaging modalities (CT, PET).
In summary, this study introduces a novel “multiple prior + ensemble learning” paradigm for MRI super‑resolution. By leveraging the complementary strengths of traditional SR methods and deep generative models, and by fusing their outputs through a learned ensemble CNN, the approach delivers superior artifact suppression and detail preservation compared with existing deep‑learning‑only SR techniques, offering a promising pathway toward higher‑quality MR imaging without hardware upgrades.
Comments & Academic Discussion
Loading comments...
Leave a Comment