Physiological Closed-Loop Control (PCLC) Systems: Review of a Modern Frontier in Automation
Over the past decade, there has been an unprecedented international focus on improved quality and availability of medical care, which has reignited interest in clinical automation and drawn researchers toward novel solutions in the field of physiological closed-loop control systems (PCLCs). Today, multidisciplinary groups of expert scientists, engineers, clinicians, mathematicians, and policy-makers are combining their knowledge and experience to develop both the next generation of PCLC-based medical equipment and a collaborative commercial/academic infrastructure to support this rapidly expanding frontier. In the following article, we provide a robust introduction to the various aspects of this growing field motivated by the recent and ongoing work supporting two leading technologies: the artificial pancreas (AP) and automated anesthesia. Following a brief high-level overview of the main concepts in automated therapy and some relevant tools from systems and control theory, we explore – separately – the developments, challenges, state-of-the-art, and probable directions for AP and automated anesthesia systems. We then close the review with a consideration of the common lessons gleaned from these ventures and the implications they present for future investigations and adjacent research.
💡 Research Summary
The paper provides a comprehensive review of physiological closed‑loop control (PCLC) systems, focusing on two flagship applications: the artificial pancreas (AP) for type‑1 diabetes and automated anesthesia. It begins by defining PCLCs as feedback‑driven medical devices that continuously sense a physiological variable, compute a therapeutic command via a control algorithm, and actuate treatment (e.g., insulin infusion, drug delivery) without direct manual intervention. The authors stress that PCLCs are intended to augment, not replace, clinicians; they shift the clinician’s role from low‑level pump operation to high‑level goal setting and supervision.
A substantial portion of the review is devoted to the modeling foundations required for safe and effective PCLC design. Human physiology is represented using compartmental models that capture pharmacokinetic (PK) and pharmacodynamic (PD) processes. The paper outlines how each compartment is described by ordinary differential equations, illustrates a generic two‑compartment example, and points to extensive literature for more complex multi‑compartment representations. Accurate models of the patient, the sensor, and the actuator are emphasized as prerequisites for any control strategy.
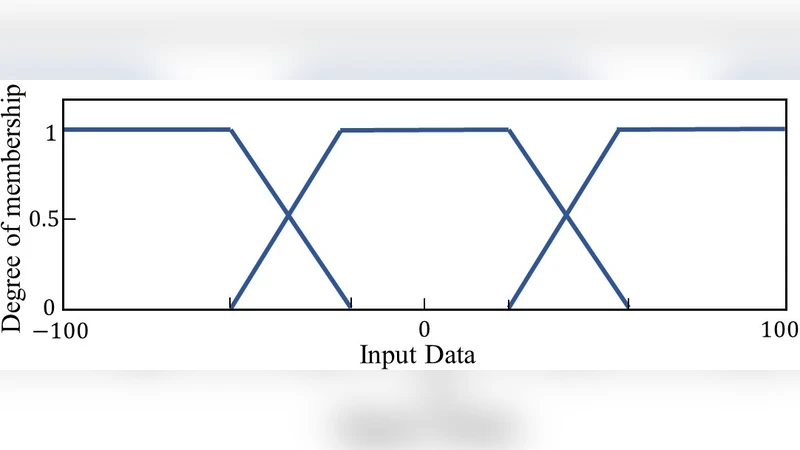
Three control methodologies dominate current PCLC research: proportional‑integral‑derivative (PID), model predictive control (MPC), and fuzzy‑logic (F‑L). PID is praised for its simplicity and real‑time applicability but is limited by patient‑to‑patient variability and non‑linearity. MPC offers optimal performance under constraints (dose limits, safety bounds) by solving a receding‑horizon optimization problem at each sampling instant; however, it demands reliable models and significant computational resources. Fuzzy‑logic encodes expert knowledge into rule‑based membership functions, making it suitable for highly nonlinear or poorly understood dynamics, yet its performance hinges on the quality of the rule base and tuning. The authors note that hybrid schemes (e.g., PID + MPC, fuzzy + reinforcement learning) are increasingly explored to combine strengths.
In the artificial pancreas case study, the review details the closed‑loop architecture: continuous glucose monitoring (CGM) provides glucose readings every few minutes, which feed a controller that commands an insulin pump. Key challenges discussed include sensor lag and dropout, inter‑day insulin sensitivity changes, meal‑time carbohydrate estimation, and the risk of hypoglycemia. Safety mechanisms such as low‑glucose suspend, predictive low‑glucose alerts, and adaptive parameter estimation (Bayesian, machine‑learning) are described. The authors summarize clinical trial outcomes, noting that modern AP systems can keep glucose within target ranges >70 % of the time while markedly reducing severe hypoglycemia.
The automated anesthesia section examines control of depth of anesthesia (DOA) using bispectral index (BIS) and hemodynamic variables. Dual‑drug delivery (propofol and remifentanil) is modeled with PK/PD equations that capture drug interactions and rapid effect site equilibration. Control strategies include sliding‑mode control for robustness, MPC for dose constraints, and fuzzy‑logic for handling abrupt surgical stimuli. Safety protocols—maximum infusion limits, emergency manual override, and continuous clinician monitoring—are highlighted as essential because over‑ or under‑dosage can cause catastrophic outcomes.
Regulatory considerations are thoroughly addressed. The paper references the IEC 60601‑1‑10 standard, FDA’s Center for Devices and Radiological Health (CDRH) definition of PCLC devices, and the concept of Level of Automation (LOA) that categorizes devices from clinician‑driven to fully autonomous. The authors outline a staged validation pathway: in‑silico simulations, animal studies, pilot clinical trials, and full‑scale regulatory submissions.
Finally, the review identifies future research directions: (1) advanced data‑driven and personalized modeling to capture inter‑patient variability; (2) real‑time cybersecurity and privacy safeguards for networked PCLCs; (3) multidisciplinary collaboration platforms that integrate engineering, medicine, policy, and social science; and (4) development of ambulatory, wearable, and remote‑monitoring PCLCs enabled by low‑power communication and energy‑harvesting technologies. The authors conclude that, with continued progress in modeling, control, safety assurance, and regulatory alignment, PCLCs have the potential to transform chronic disease management, peri‑operative care, and a broad spectrum of physiologic therapies.
Comments & Academic Discussion
Loading comments...
Leave a Comment