Surround suppression explained by long-range recruitment of local competition, in a columnar V1 model
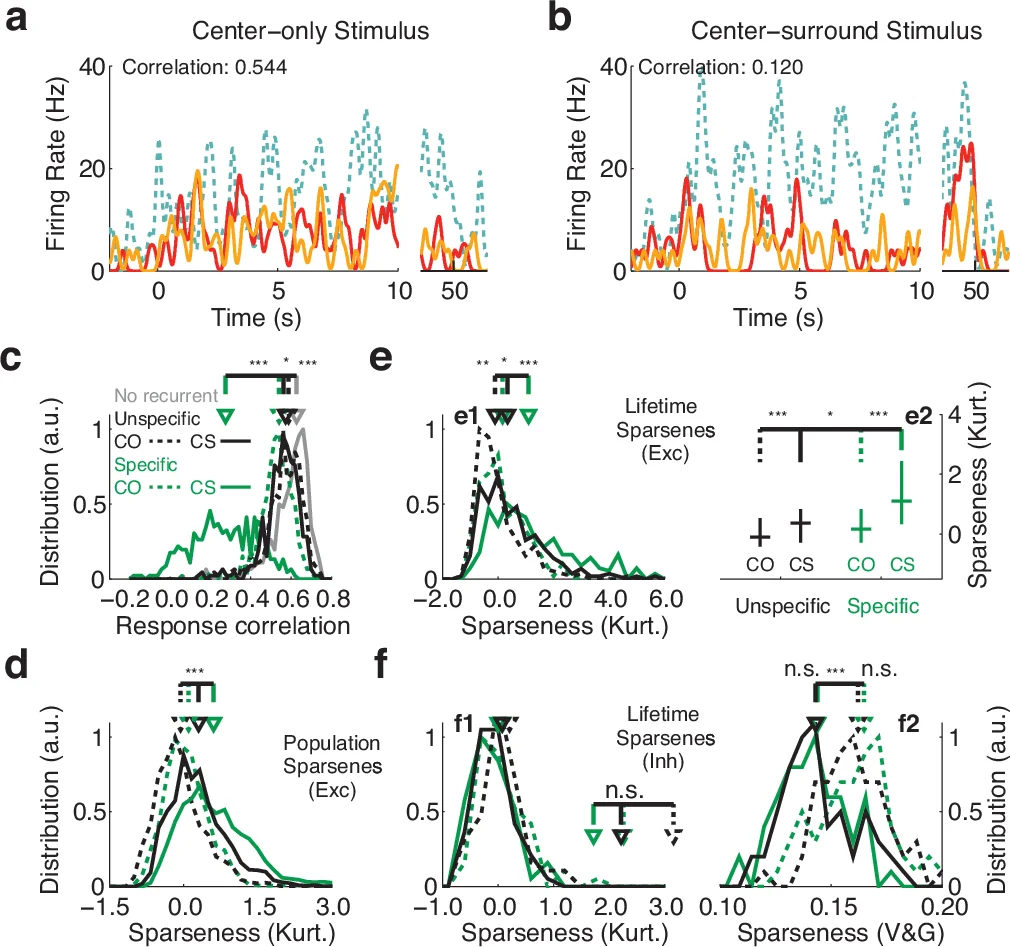
Although neurons in columns of visual cortex of adult carnivores and primates share similar orientation tuning preferences, responses of nearby neurons are surprisingly sparse and temporally uncorrelated, especially in response to complex visual scenes. The mechanisms underlying this counter-intuitive combination of response properties are still unknown. Here we present a computational model of columnar visual cortex which explains experimentally observed integration of complex features across the visual field, and which is consistent with anatomical and physiological profiles of cortical excitation and inhibition. In this model, sparse local excitatory connections within columns, coupled with strong unspecific local inhibition and functionally-specific long-range excitatory connections across columns, give rise to competitive dynamics that reproduce experimental observations. Our results explain surround modulation of responses to simple and complex visual stimuli, including reduced correlation of nearby excitatory neurons, increased excitatory response selectivity, increased inhibitory selectivity, and complex orientation-tuning of surround modulation.
💡 Research Summary
The authors present a biologically grounded spiking network model of primary visual cortex (V1) that explains how surround suppression and decorrelation of neuronal activity arise from a combination of sparse local excitatory connectivity, dense non‑specific local inhibition, and functionally specific long‑range excitatory projections. The model consists of seven populations, each representing a distinct region of visual space. Within each population, neurons are organized into orientation columns arranged on a ring (a hypercolumn), and each column contains excitatory (E) and inhibitory (I) cells. Excitatory cells are further divided into several specific subnetworks (SSNs). Local E‑E connections are sparse and preferentially link neurons belonging to the same SSN, controlled by a parameter P⁺ that determines the degree of SSN‑specificity. In contrast, local E‑I and I‑I connections are dense and non‑specific, providing strong, broadly tuned inhibition. Long‑range excitatory connections link columns of different populations that share similar preferred orientations (θ‑specific) and are also SSN‑specific, governed by a second parameter P_M.
Two stimulus conditions are simulated: (i) a “center‑only” grating presented to the classical receptive field (cRF) of the central population, and (ii) a “center‑surround” condition where the same grating is also presented to the surrounding populations. Under center‑only stimulation, neurons within a column fire in synchrony according to their shared orientation preference, but their spike trains are only weakly negatively correlated (median ≈ ‑0.06). When the surround is added, long‑range excitatory input recruits competition among SSNs: activity in one SSN suppresses activity in the others via the shared inhibitory pool. This competition manifests as a marked increase in negative correlation (median ≈ ‑0.24), an increase in inhibitory firing rates (≈ 21 → 26 Hz) and a decrease in excitatory firing rates (≈ 16 → 12 Hz), reproducing experimental observations of surround‑induced suppression and decorrelation.
Systematic exploration of P⁺ and P_M shows that both local and long‑range SSN‑specificity are required for strong competition. When either parameter is low (connections essentially random), neurons remain positively correlated; when both approach 1 (highly specific wiring), competition is maximal and correlations become strongly negative. The authors further analyze a reduced mean‑field version of the network, defining a Competition Index (CI). They find that increasing excitatory synaptic strength (J_E) enhances competition, whereas stronger inhibition (J_I) diminishes it. Within realistic cortical parameter ranges, the network operates in a “soft winner‑take‑all” (sWTA) regime: multiple excitatory neurons can be active, but an increase in one neuron’s activity suppresses the others without silencing them completely. If J_E is increased further, the system shifts to a “hard winner‑take‑all” (hWTA) regime where only a single neuron dominates.
The model accounts for several key experimental findings: (1) surround stimulation suppresses excitatory responses while enhancing inhibitory activity; (2) neuronal responses within a column become less correlated; (3) excitatory orientation selectivity sharpens, whereas inhibitory selectivity broadens slightly; (4) the orientation tuning of surround modulation is complex, showing both suppression and facilitation depending on relative orientations. By linking these phenomena to a single mechanistic principle—competition induced by SSN‑specific long‑range excitation—the study provides a unified explanation for how V1 achieves sparse, efficient coding despite highly correlated natural inputs.
In summary, this work demonstrates that the interplay of sparse, specific local excitation, dense non‑specific inhibition, and orientation‑specific long‑range excitatory projections can generate the observed surround suppression and decorrelation in columnar V1. The findings highlight the importance of structured excitatory connectivity in shaping cortical dynamics and suggest that competition among subnetworks is a fundamental operation underlying efficient visual processing.
Comments & Academic Discussion
Loading comments...
Leave a Comment