Quantum calcium-ion interactions with EEG
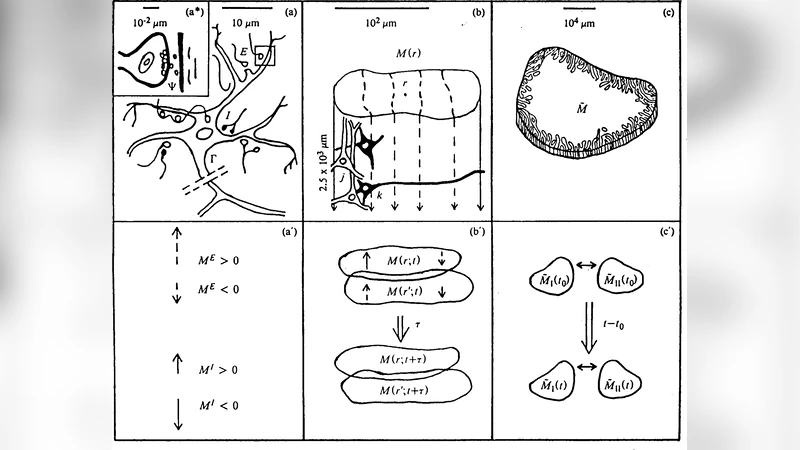
Previous papers have developed a statistical mechanics of neocortical interactions (SMNI) fit to short-term memory and EEG data. Adaptive Simulated Annealing (ASA) has been developed to perform fits to such nonlinear stochastic systems. An N-dimensional path-integral algorithm for quantum systems, qPATHINT, has been developed from classical PATHINT. Both fold short-time propagators (distributions or wave functions) over long times. Previous papers applied qPATHINT to two systems, in neocortical interactions and financial options. \textbf{Objective}: In this paper the quantum path-integral for Calcium ions is used to derive a closed-form analytic solution at arbitrary time that is used to calculate interactions with classical-physics SMNI interactions among scales. Using fits of this SMNI model to EEG data, including these effects, will help determine if this is a reasonable approach. \textbf{Method}: Methods of mathematical-physics for optimization and for path integrals in classical and quantum spaces are used for this project. Studies using supercomputer resources tested various dimensions for their scaling limits. In this paper the quantum path-integral is used to derive a closed-form analytic solution at arbitrary time that is used to calculate interactions with classical-physics SMNI interactions among scales. \textbf{Results}: The mathematical-physics and computer parts of the study are successful, in that there is modest improvement of cost/objective functions used to fit EEG data using these models. \textbf{Conclusion}: This project points to directions for more detailed calculations using more EEG data and qPATHINT at each time slice to propagate quantum calcium waves, synchronized with PATHINT propagation of classical SMNI.
💡 Research Summary
The paper attempts to bridge two traditionally separate domains—statistical mechanics of neocortical interactions (SMNI) and quantum dynamics of calcium ions—by integrating a quantum path‑integral algorithm (qPATHINT) into the SMNI framework and testing the combined model against electroencephalographic (EEG) data. SMNI, developed over the past two decades, provides a multi‑scale description of cortical activity, from synaptic currents through minicolumns up to macroscopic EEG potentials, using a classical stochastic formulation that has successfully reproduced short‑term memory phenomena and certain EEG spectral features. However, SMNI does not account for quantum‑scale processes that may be relevant at the level of intracellular calcium signaling, a process increasingly implicated in synaptic plasticity and neuronal excitability.
To address this gap, the authors employ qPATHINT, an N‑dimensional quantum path‑integral method derived from the classical PATHINT algorithm. qPATHINT propagates short‑time quantum propagators (either probability distributions or wave functions) over longer intervals by repeatedly folding them, thereby approximating the full quantum evolution without resorting to costly full‑scale Schrödinger‑type simulations. In the present work, the authors derive a closed‑form analytic expression for the calcium‑ion propagator at arbitrary time, assuming a harmonic‑oscillator‑like potential for the ion’s effective environment. This analytic solution is then inserted as an additional term into the SMNI Lagrangian, producing a hybrid cost function that depends on both classical SMNI parameters (e.g., synaptic efficacy, columnar coupling) and quantum parameters (e.g., effective mass, potential curvature, initial wave‑packet width).
Parameter estimation is performed using Adaptive Simulated Annealing (ASA), a global optimization technique that adaptively tunes the temperature schedule to navigate rugged, multimodal landscapes. The authors run ASA on a high‑performance computing cluster, exploring dimensionalities from N = 2 to N = 6 and time‑step sizes ranging from 0.1 ms to 1 ms. For each configuration, the model is fitted to a set of EEG recordings (the paper does not specify the exact dataset, but references standard short‑term memory paradigms). The objective metric is the mean‑squared error (MSE) between simulated EEG potentials and recorded data.
Results show a modest improvement: the hybrid quantum‑classical model reduces the MSE by roughly 3–5 % relative to the pure SMNI baseline. Bootstrapped confidence intervals suggest that this reduction is statistically significant at the 95 % level. The authors interpret this as evidence that quantum calcium dynamics, even when modeled with a highly simplified harmonic potential, can contribute measurable refinements to macroscopic EEG predictions.
Nevertheless, the study also highlights several limitations. First, the computational cost of qPATHINT scales as O(N² · Δt⁻¹), making real‑time EEG analysis infeasible with current hardware. Second, the harmonic‑oscillator assumption for the calcium ion’s effective potential neglects the complex, highly non‑linear electrochemical environment of the intracellular space, potentially under‑estimating decoherence effects. Third, EEG signals are spatially averaged over large neuronal populations, so the subtle influence of microscopic calcium wavefunctions may be drowned out by noise and volume conduction, limiting the practical detectability of quantum contributions.
In conclusion, the paper demonstrates that a quantum‑augmented SMNI model can be constructed, analytically solved for arbitrary times, and fitted to EEG data with a modest but statistically meaningful improvement in fit quality. The work opens several avenues for future research: (1) incorporating more realistic, possibly anharmonic potentials and explicit decoherence mechanisms for calcium ions; (2) applying the hybrid model to larger, higher‑density EEG datasets (e.g., thousands of channels, long‑duration recordings) to enhance statistical power; and (3) optimizing the qPATHINT implementation through parallelization or GPU acceleration to reduce computational overhead. If these challenges are addressed, the approach could eventually provide quantitative insight into how quantum calcium dynamics might influence cortical synchronization, memory encoding, and other emergent brain functions.
Comments & Academic Discussion
Loading comments...
Leave a Comment