Monocular 3D Sway Tracking for Assessing Postural Instability in Cerebral Hypoperfusion During Quiet Standing
Postural instability is prevalent in aging and neurodegenerative disease, decreasing quality of life and independence. Quantitatively monitoring balance control is important for assessing treatment efficacy and rehabilitation progress. However, existing technologies for assessing postural sway are complex and expensive, limiting their widespread utility. Here, we propose a monocular imaging system capable of assessing sub-millimeter 3D sway dynamics during quiet standing. Two anatomical targets with known feature geometries were placed on the lumbar and shoulder. Upper and lower trunk 3D kinematic motion was automatically assessed from a set of 2D frames through geometric feature tracking and an inverse motion model. Sway was tracked in 3D and compared between control and hypoperfusion conditions in 14 healthy young adults. The proposed system demonstrated high agreement with a commercial motion capture system (error $1.5 \times 10^{-4}~\text{mm}$, [$-0.52$, $0.52$]). Between-condition differences in sway dynamics were observed in anterior-posterior sway during early and mid stance, and medial-lateral sway during mid stance commensurate with decreased cerebral perfusion, followed by recovered sway dynamics during late stance with cerebral perfusion recovery. This inexpensive single-camera system enables quantitative 3D sway monitoring for assessing neuromuscular balance control in weakly constrained environments.
💡 Research Summary
This paper presents a low‑cost, high‑precision system for quantifying three‑dimensional postural sway during quiet standing using a single monocular camera and two custom geometric markers. The authors attach asymmetric markers with known 3‑D feature layouts to the lumbar region (L3 vertebra) and the left shoulder (acromion). After a one‑time intrinsic and extrinsic calibration of the camera (including radial distortion correction), each video frame is processed to locate the marker features with sub‑pixel accuracy via multi‑orientation kernel convolution and gradient‑orthogonal minimization.
The 2‑D image coordinates of the detected features are then fitted to a rigid‑body kinematic model of the marker geometry. The model has six degrees of freedom: three translations and three Euler rotations. An initial closed‑form solution provides the pose for the first frame; subsequent frames are initialized with the previous pose and refined using Levenberg‑Marquardt non‑linear least‑squares optimization. This frame‑to‑frame continuity exploits the fact that sway during quiet standing changes only by sub‑millimeter amounts, ensuring rapid convergence and avoidance of local minima.
From the fitted pose, the shoulder marker directly yields an upper‑trunk 3‑D coordinate, while a virtual point 10 cm deep along the torso axis (derived from the lumbar marker’s orientation) provides a lower‑trunk coordinate. Both points are transformed from camera space into an anatomical reference frame defined by anterior‑posterior (AP), medial‑lateral (ML), and superior‑inferior (SI) axes, enabling biomechanically meaningful analysis of sway in each direction.
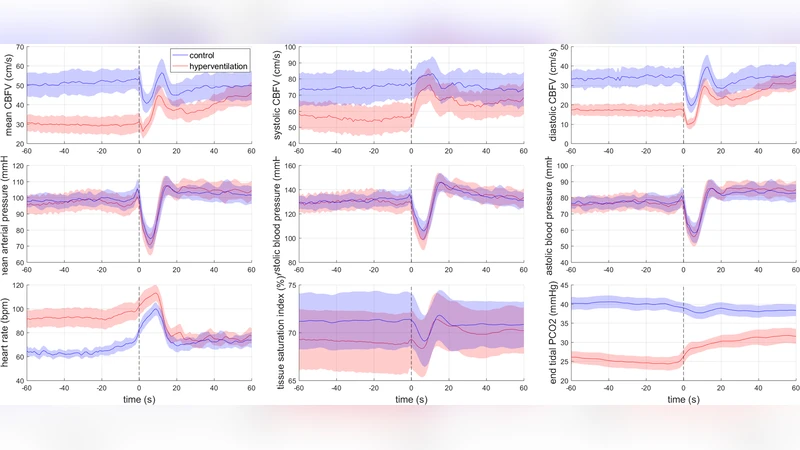
The system was evaluated on fourteen healthy young adults (average age 24.7 ± 4.3 years). Each participant performed two standing trials: (1) a normal supine‑to‑stand transition followed by 60 s of quiet standing, and (2) a 2‑minute guided hyperventilation (≈20 breaths min⁻¹) to lower end‑tidal CO₂ and thereby induce cerebral hypoperfusion, immediately followed by the same standing protocol. Physiological signals—including ECG, finger arterial pressure, stroke volume (ModelFlow), transcranial Doppler cerebral blood‑flow velocity, end‑tidal CO₂, and near‑infrared spectroscopy tissue oxygenation—were recorded simultaneously.
Comparisons with a commercial Vicon motion‑capture system showed an average absolute error of 1.5 × 10⁻⁴ mm and a 95 % confidence interval of ±0.52 mm, confirming sub‑millimeter accuracy. During the hypoperfusion condition, AP sway was significantly larger in early and mid‑stance, while ML sway peaked in mid‑stance, reflecting the destabilizing effect of reduced cerebral blood flow. In the late‑stance phase, sway returned toward baseline as cerebral perfusion recovered, demonstrating the system’s sensitivity to dynamic physiological changes.
Key technical contributions include: (i) leveraging known asymmetric marker geometry to resolve depth from a single view, (ii) a two‑step pose estimation pipeline that combines a closed‑form initializer with iterative refinement, (iii) projection of a virtual lower‑trunk point to capture independent trunk segments, and (iv) transformation into an anatomically aligned coordinate system for clinically relevant interpretation.
The authors argue that this approach overcomes the cost, setup complexity, and expertise barriers of traditional multi‑camera or marker‑based systems, making continuous balance monitoring feasible in clinics, long‑term care facilities, or even home environments. Potential applications span fall‑risk screening, rehabilitation progress tracking, and objective assessment of interventions that modulate cerebral perfusion (e.g., breathing exercises, pharmacologic agents). Future work will extend validation to older adults and patients with neurodegenerative disorders, explore long‑term monitoring, and investigate marker‑less implementations using deep‑learning pose estimation while preserving sub‑millimeter fidelity.
Comments & Academic Discussion
Loading comments...
Leave a Comment