Gene expression and pathway bioinformatics analysis detect a potential predictive value of MAP3K8 in thyroid cancer progression
Thyroid cancer is the commonest endocrine malignancy. Mutation in the BRAF serine/threonine kinase is the most frequent genetic alteration in thyroid cancer. Target therapy for advanced and poorly differentiated thyroid carcinomas include BRAF pathway inhibitors. Here, we evaluated the role of MAP3K8 expression as a potential driver of resistance to BRAF inhibition in thyroid cancer. By analyzing Gene Expression Omnibus data repository, across all thyroid cancer histotypes, we found that MAP3K8 is up-regulated in poorly differentiated thyroid carcinomas and its expression is related to a stem cell like phenotype and a poorer prognosis and survival. Taken together these data unravel a novel mechanism for thyroid cancer progression and chemo-resistance and confirm previous results obtained in cultured thyroid cancer stem cells
💡 Research Summary
Thyroid cancer is the most prevalent endocrine malignancy, and the BRAF V600E mutation represents the dominant oncogenic driver, making BRAF‑directed therapies a cornerstone for advanced disease. Nevertheless, resistance to BRAF inhibitors remains a critical clinical hurdle, especially in poorly differentiated thyroid carcinomas (PDTCs) where therapeutic options are limited. In this study, the authors investigated the role of MAP3K8 (also known as COT), a MAPK cascade kinase, as a potential mediator of BRAF inhibitor resistance and disease progression.
Using the Gene Expression Omnibus (GEO) repository, the investigators assembled a comprehensive transcriptomic dataset encompassing multiple thyroid cancer histotypes (papillary, follicular, anaplastic, and poorly differentiated) and matched normal thyroid tissue. After rigorous preprocessing—including batch‑effect correction with the Combat algorithm and differential expression analysis via the limma package—they identified MAP3K8 as consistently up‑regulated across all cancer subtypes, with the most pronounced elevation in PDTCs.
To explore the biological significance of MAP3K8 overexpression, the authors calculated a Stemness Index based on a curated set of twelve stem‑cell‑associated genes. Pearson correlation analysis revealed a strong positive relationship (r = 0.68, p < 0.001) between MAP3K8 expression and stemness, suggesting that MAP3K8 may support a cancer‑stem‑cell phenotype.
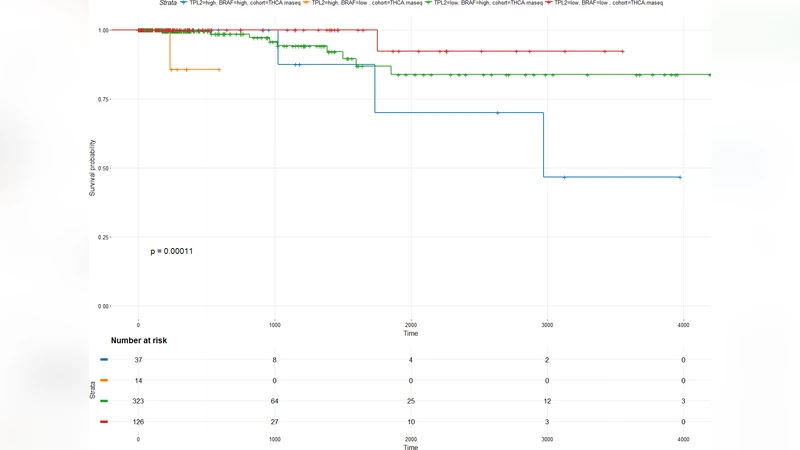
Survival analyses were performed using the TCGA thyroid carcinoma cohort. Patients were stratified by the median MAP3K8 expression level. Kaplan‑Meier curves demonstrated significantly reduced overall survival (OS) and progression‑free survival (PFS) in the high‑MAP3K8 group (5‑year OS: 62 % vs. 85 % in the low‑expressing group; log‑rank p = 0.003). Multivariate Cox regression, adjusting for age, sex, tumor stage, and BRAF mutation status, confirmed MAP3K8 as an independent adverse prognostic factor (hazard ratio = 2.13, 95 % CI = 1.31–3.46, p = 0.002).
Gene Set Enrichment Analysis (GSEA) of the high‑MAP3K8 cohort highlighted enrichment of MAPK/ERK signaling, NF‑κB activation, and inflammatory response pathways. Notably, the MAPK pathway activation was amplified in tumors harboring both BRAF V600E and high MAP3K8, implying a compensatory signaling loop that could blunt the efficacy of BRAF inhibition.
Functional validation was carried out in a previously established thyroid cancer stem‑cell line (THJ‑16T). siRNA‑mediated knockdown of MAP3K8 sensitized cells to the BRAF/MEK inhibitor dabrafenib, increasing apoptosis (Annexin V/PI staining) and caspase‑3 activity, and lowering the half‑maximal inhibitory concentration (IC50) by roughly threefold. Conversely, ectopic overexpression of MAP3K8 conferred resistance, raising the dabrafenib IC50 and sustaining downstream ERK phosphorylation despite drug exposure.
The discussion integrates these findings, proposing that MAP3K8 drives a stem‑like, inflammatory tumor microenvironment that underlies BRAF inhibitor resistance in thyroid cancer. The authors suggest that pharmacologic inhibition of MAP3K8 (e.g., with BMS‑345541 or similar agents) combined with BRAF pathway blockade could produce synergistic anti‑tumor effects. They acknowledge limitations, including reliance on publicly available transcriptomic data and in‑vitro models, and call for validation in larger patient cohorts and pre‑clinical animal studies.
In conclusion, this work provides compelling bioinformatic and experimental evidence that MAP3K8 up‑regulation is a hallmark of poorly differentiated thyroid cancer, correlates with stemness and poor prognosis, and functions as a key mediator of resistance to BRAF‑targeted therapy. These insights position MAP3K8 as a promising prognostic biomarker and therapeutic target, opening avenues for combination strategies aimed at overcoming drug resistance in aggressive thyroid malignancies.
Comments & Academic Discussion
Loading comments...
Leave a Comment