Network models for characterization of trabecular bone
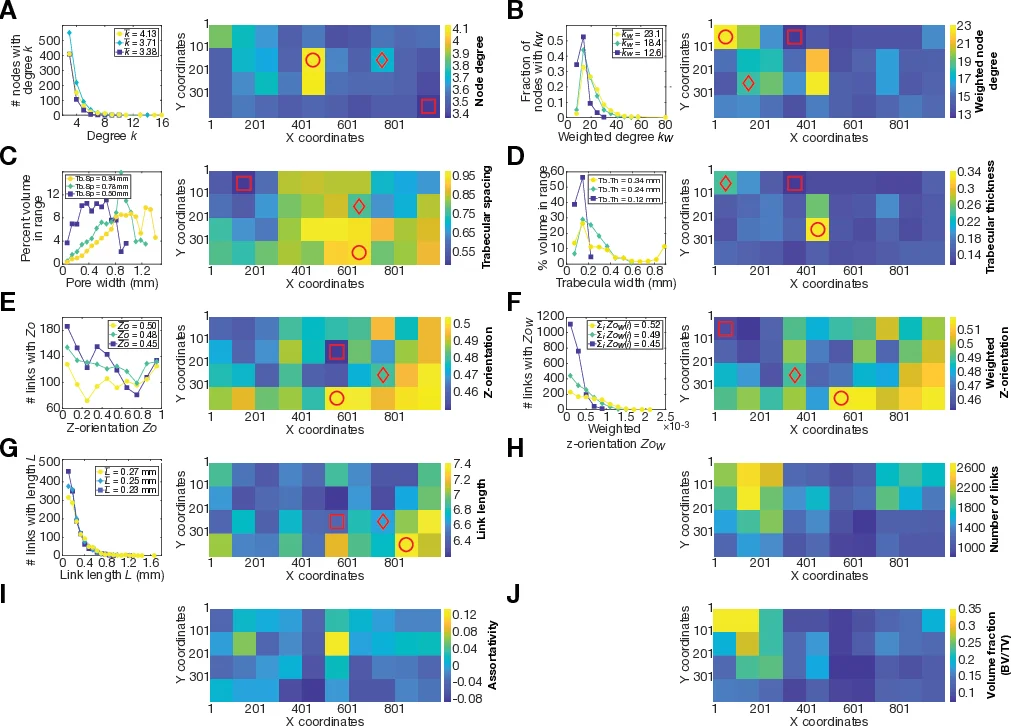
Trabecular bone is a lightweight, compliant material organized as a web of struts and rods (trabeculae) that erode with age and the onset of bone diseases like osteoporosis, leading to increased fracture risk. The traditional diagnostic marker of osteoporosis, bone mineral density (BMD), has been shown in ex vivo experiments to correlate poorly with fracture resistance when considered on its own, while structural features in conjunction with BMD can explain more of the variation in trabecular bone strength. We develop a network-based model of trabecular bone by creating graphs from micro-computed tomography images of human bone, with weighted links representing trabeculae and nodes representing branch points. These graphs enable calculation of quantitative network metrics to characterize trabecular structure. We also create finite element models of the networks in which each link is represented by a beam, facilitating analysis of the mechanical response of the bone samples to simulated loading. We examine the structural and mechanical properties of trabecular bone at the scale of individual trabeculae (of order 0.1 mm) and at the scale of selected volumes of interest (approximately a few mm), referred to as VOIs. At the VOI scale, we find significant correlations between the stiffness of VOIs and 10 different structural metrics. Individually, the volume fraction of each VOI is most strongly correlated to the stiffness of the VOI. We use multiple linear regression to identify the smallest subset of variables needed to capture the variation in stiffness. In a linear fit, we find that node degree, weighted node degree, Z-orientation, weighted Z-orientation, trabecular spacing, link length, and the number of links are the structural metrics that are most significant (p < 0.05) in capturing the variation of stiffness in trabecular networks.
💡 Research Summary
This paper presents a novel network‑based framework for quantifying the micro‑architectural and mechanical properties of human trabecular bone, with the aim of improving osteoporosis assessment beyond conventional bone mineral density (BMD) measurements. High‑resolution micro‑CT scans (37 µm voxel size) of a cadaveric L3 vertebral body were processed using Otsu thresholding and a despeckling filter, then partitioned into 100 × 100 pixel tiles (3.7 mm × 3.7 mm) forming 40 volumes of interest (VOIs), each comprising 100 axial slices (≈50 mm³).
Skeletonization (Skeleton3D) extracted the medial axis of the trabecular network, which was subsequently converted into an undirected graph via the Skel2Graph library. In this graph, nodes correspond to branch points where trabeculae intersect, and edges (links) represent individual trabeculae. Each link was assigned a weight equal to the average trabecular thickness measured with BoneJ, enabling the calculation of weighted node degree, weighted Z‑orientation, and other topological descriptors. Conventional histomorphometric parameters—trabecular thickness (Tb.Th), spacing (Tb.Sp), and bone volume fraction (BV/TV)—were also computed for each VOI.
Two mechanical modeling approaches were employed. First, a full continuum finite‑element (FE) model was generated directly from the binary bone images, providing a high‑fidelity reference for stress and strain distribution. Second, a computationally efficient beam‑element model was built by replacing each graph edge with a cylindrical beam whose cross‑sectional area was proportional to the link weight (thickness). Identical boundary conditions and compressive loading were applied to both models, and the effective stiffness (Young’s modulus) of each VOI was extracted from the beam model.
Statistical analysis revealed significant correlations (p < 0.05) between VOI stiffness and ten structural metrics. While BV/TV showed the strongest single‑variable correlation, multiple linear regression identified a minimal subset of eight descriptors that together explained >90 % of the stiffness variance: node degree, weighted node degree, Z‑orientation, weighted Z‑orientation, trabecular spacing, link length, number of links, and BV/TV. These metrics capture distinct aspects of the trabecular network: connectivity (degree), load‑bearing directionality (Z‑orientation), geometric scale (link length), and overall material content (BV/TV).
The study demonstrates that representing trabecular bone as a weighted graph provides a compact, mathematically tractable description of its architecture, enabling rapid computation of meaningful topological indices. When coupled with the beam‑element FE model, this approach yields accurate predictions of elastic stiffness while requiring far less computational resources than full continuum simulations. The authors argue that such a pipeline could be automated for large‑scale screening of bone quality, potentially augmenting or replacing BMD‑only diagnostics.
Limitations include the linear elastic assumption of the beam model, which does not capture post‑yield behavior or fracture propagation, and the reliance on ex‑vivo micro‑CT data, which is not yet feasible for routine in‑vivo clinical use due to radiation dose constraints. Future work should incorporate nonlinear material models, validate predictions against experimental mechanical testing, and explore low‑dose high‑resolution imaging techniques to translate this network‑based methodology into clinical practice.
Comments & Academic Discussion
Loading comments...
Leave a Comment