Identifying Ketamine Responses in Treatment-Resistant Depression Using a Wearable Forehead EEG
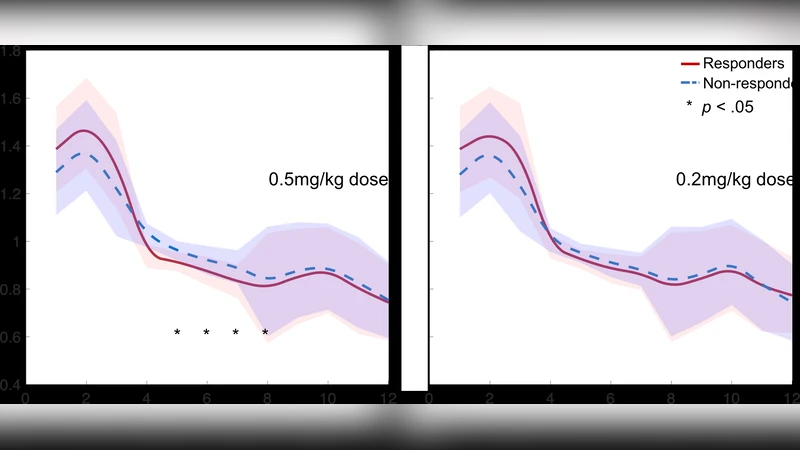
This study explores the responses to ketamine in patients with treatment-resistant depression (TRD) using a wearable forehead electroencephalography (EEG) device. We recruited fifty-five outpatients with TRD who were randomised into three approximately equal-sized groups (A: 0.5 mg/kg ketamine; B: 0.2 mg/kg ketamine; and C: normal saline) under double-blind conditions. The ketamine responses were measured by EEG signals and Hamilton Depression Rating Scale (HDRS) scores. At baseline, responders showed a significantly weaker EEG theta power than did non- responders (p < 0.05). Responders exhibited a higher EEG alpha power but lower EEG alpha asymmetry and theta cordance at post-treatment than at baseline (p < 0.05). Furthermore, our baseline EEG predictor classified responders and non-responders with 81.3 +- 9.5% accuracy, 82.1 +- 8.6% sensitivity and 91.9 +- 7.4% specificity. In conclusion, the rapid antidepressant effects of mixed doses of ketamine are associated with prefrontal EEG power, asymmetry and cordance at baseline and early post-treatment changes. The prefrontal EEG patterns at baseline may account for recognising ketamine effects in advance. Our randomised, double- blind, placebo-controlled study provides information regarding clinical impacts on the potential targets underlying baseline identification and early changes from the effects of ketamine in patients with TRD.
💡 Research Summary
This paper investigates whether a wearable forehead electroencephalography (EEG) system can predict and monitor rapid antidepressant responses to ketamine in patients with treatment‑resistant depression (TRD). Fifty‑five out‑patients meeting DSM‑IV‑TR criteria for major depressive disorder and demonstrating resistance to at least two prior antidepressant trials were enrolled. Participants were randomly assigned in a 1:1:1 ratio to receive a single intravenous infusion of either 0.5 mg/kg ketamine (Group A), 0.2 mg/kg ketamine (Group B), or normal saline placebo (Group C) under double‑blind conditions. The primary clinical outcome was change in the 17‑item Hamilton Depression Rating Scale (HDRS‑17) measured at multiple time points up to two weeks post‑infusion; a “responder” was defined as ≥45 % reduction in HDRS score at 240 minutes relative to baseline.
EEG data were collected using the “Mindo‑4S Jellyfish” wearable device, which employs four dry electrodes placed on the prefrontal scalp (Fp1, Fp2, AF7, AF8) plus a reference electrode (A2). The system samples at 512 Hz with 16‑bit resolution and incorporates a real‑time artifact‑removal pipeline based on canonical correlation analysis (CCA) and Gaussian mixture modeling (GMM). Baseline recordings consisted of a 10‑minute eyes‑closed session; after the infusion (which was not recorded due to patient discomfort), a second 10‑minute eyes‑closed session was obtained 240 minutes later. EEG signals were band‑pass filtered (1–12 Hz), transformed via a 256‑point fast Fourier transform with 0.5 Hz resolution, and used to compute the following features: absolute and relative power in the theta (4–7 Hz) and alpha (8–12 Hz) bands, alpha asymmetry
Comments & Academic Discussion
Loading comments...
Leave a Comment