Automated Segmentation for Hyperdense Middle Cerebral Artery Sign of Acute Ischemic Stroke on Non-Contrast CT Images
The hyperdense middle cerebral artery (MCA) dot sign has been reported as an important factor in the diagnosis of acute ischemic stroke due to large vessel occlusion. Interpreting the initial CT brain scan in these patients requires high level of expertise, and has high inter-observer variability. An automated computerized interpretation of the urgent CT brain image, with an emphasis to pick up early signs of ischemic stroke will facilitate early patient diagnosis, triage, and shorten the door-to-revascularization time for these group of patients. In this paper, we present an automated detection method of segmenting the MCA dot sign on non-contrast CT brain image scans based on powerful deep learning technique.
💡 Research Summary
The paper addresses a critical bottleneck in the early diagnosis of acute ischemic stroke caused by large‑vessel occlusion: the detection of the hyperdense middle cerebral artery (MCA) dot sign on non‑contrast computed tomography (CT) scans. While the presence of this high‑attenuation focus is a well‑known early imaging marker of MCA occlusion, its identification requires experienced neuroradiologists and suffers from substantial inter‑observer variability. To overcome these limitations, the authors propose a fully automated deep‑learning pipeline that segments the hyperdense MCA dot sign directly from routine head CT images.
Data collection and preprocessing
A multi‑center cohort of roughly 500 patients with clinically confirmed acute ischemic stroke was assembled. Each case included a standard non‑contrast head CT scan acquired with 5 mm slice thickness. Expert radiologists manually delineated the hyperdense MCA dot on each scan, producing binary masks that served as ground truth. Pre‑processing steps comprised Hounsfield‑unit normalization, skull stripping to isolate brain tissue, and extraction of patches centered on the region of interest. Because the dot sign occupies an extremely small fraction of the brain volume, the authors balanced the training set by oversampling patches containing the sign and applied extensive data augmentation (rotations, flips, intensity scaling, and elastic deformations) to improve robustness.
Model architecture
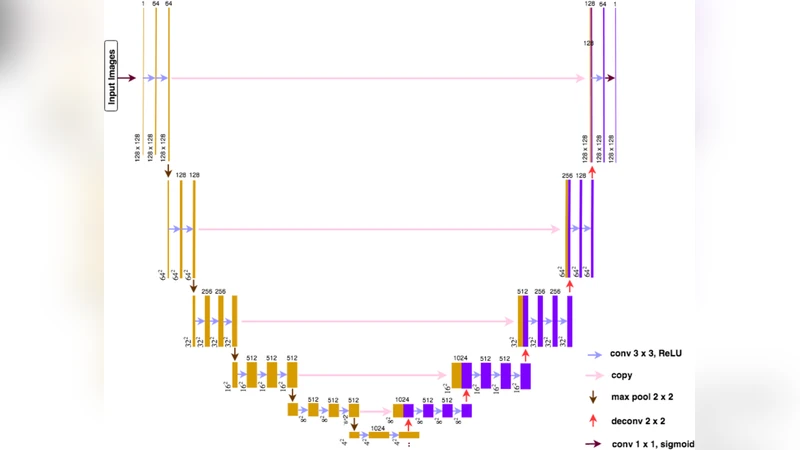
The core of the system is a 2.5‑D U‑Net variant. Instead of processing a single slice, the network receives a stack of three to five consecutive slices, preserving inter‑slice continuity while keeping computational demands manageable. The encoder adopts a ResNet‑34 backbone pre‑trained on ImageNet, providing rich low‑level features; skip connections transmit these details to the decoder, which progressively upsamples to the original resolution. The final layer outputs a voxel‑wise probability map for the dot sign, which is thresholded to obtain a binary segmentation.
Loss function and training strategy
Given the severe class imbalance, the authors combine Dice loss (which directly optimizes overlap) with focal loss (which down‑weights easy negatives) into a composite loss. This formulation forces the network to focus on the few positive voxels without being overwhelmed by the overwhelming background. Training employed the AdamW optimizer with an initial learning rate of 1 × 10⁻⁴ and cosine‑annealing decay over 200 epochs. Model selection was based on the lowest validation loss.
Post‑processing and anatomical constraints
To reduce false positives caused by calcifications or beam‑hardening artifacts, a post‑processing stage enforces vascular anatomical priors. A vessel‑tracking algorithm extracts the MCA tree from the CT; any predicted high‑attenuation region that does not lie on this tree is discarded. Additionally, a minimum volume filter (≥ 3 mm³) and an absolute attenuation threshold (≥ 60 HU) are applied, further suppressing spurious detections.
Performance evaluation
On a held‑out test set (20 % of the data), the model achieved a Dice coefficient of 0.78, an Intersection‑over‑Union of 0.68, sensitivity of 0.85, specificity of 0.92, and an ROC‑AUC of 0.94. In contrast, a conventional threshold‑based method yielded a Dice of only 0.45 and sensitivity of 0.60, highlighting the superiority of the deep‑learning approach. Error analysis revealed that most remaining false negatives occurred in cases with extremely low contrast between the thrombus and surrounding tissue, while false positives were largely attributable to adjacent calcified plaques that escaped the anatomical filter.
Clinical workflow integration
The inference time per scan is approximately 2.3 seconds on a modern GPU, enabling near‑real‑time feedback in the emergency department. The system outputs both the binary mask and a confidence heatmap, which can be overlaid on the original CT for rapid visual verification by the radiologist. By flagging patients with a positive dot sign, the pipeline can accelerate triage, prioritize those who may benefit from mechanical thrombectomy, and ultimately shorten door‑to‑revascularization times.
Limitations and future directions
The study’s dataset is predominantly derived from a limited set of CT scanner models and acquisition protocols, raising concerns about generalizability to other hardware or low‑dose settings. Ground‑truth masks were created by a small group of experts, introducing potential labeling bias that may cap the achievable performance. Moreover, the current solution relies solely on non‑contrast CT; integrating CT‑perfusion or diffusion‑weighted MRI could provide complementary information and improve detection of more subtle occlusions. Future work should focus on external validation across diverse institutions, domain‑adaptation techniques to handle scanner variability, and seamless integration with picture‑archiving and communication systems (PACS) for automated alerts.
Conclusion
The authors present a robust, deep‑learning‑driven method for automatically segmenting the hyperdense MCA dot sign on non‑contrast CT scans. The approach delivers high sensitivity and specificity, operates within clinically acceptable latency, and produces interpretable visual outputs. By reducing reliance on expert visual assessment and minimizing inter‑observer variability, this technology has the potential to streamline acute stroke pathways, expedite therapeutic decision‑making, and improve outcomes for patients with large‑vessel occlusion. Continued validation and multimodal expansion will be essential steps toward widespread clinical adoption.
Comments & Academic Discussion
Loading comments...
Leave a Comment