Why Pacing Frequency Affects the Production of Early Afterdepolarizations in Cardiomyocytes: An Explanation Revealed by Slow/Fast Analysis of a Minimal Model
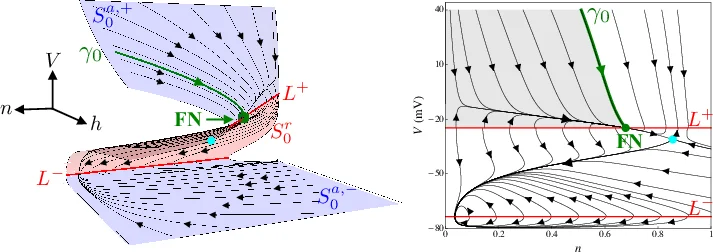
Early afterdepolarizations (EADs) are pathological voltage oscillations in cardiomyocytes that have been observed in response to a number of pharmacological agents and disease conditions. EADs are small voltage fluctuations that occur during the plateau of an action potential. Although a single-cell behavior, EADs can lead to tissue-level arrhythmias, including ventricular tachycardia. Much is currently known about the biophysical mechanisms (i.e., the roles of ion channels and intracellular calcium stores) for EADs, due partially to the development and analysis of mathematical models. This includes the application of slow/fast analysis, which takes advantage of timescale separation inherent in the system to simplify its analysis. We take this further, using a minimal 3D model to demonstrate that the phase-2 EADs are canards that are formed in the neighborhood of a folded node singularity. This knowledge allows us to determine the number of EADs that can be produced for a given parameter set without performing computer simulations, and provides guidance on parameter changes that can facilitate or inhibit EAD production. With this approach, we demonstrate why periodic stimulation, as would occur in an intact heart, preferentially facilitates EAD production when applied at low frequencies,. We also explain the origin of complex alternan dynamics that can occur with intermediate-frequency stimulation, in which varying numbers of EADs are produced with each stimulation. These revelations fall out naturally from an understanding of folded node singularities, but are hard or impossible to glean from a knowledge of the biophysical mechanism for EADs alone. Therefore, an understanding of the canard mechanism is a useful complement to an understanding of the biophysical mechanism that has been developed over years of experimental and computational investigations.
💡 Research Summary
The paper investigates why the pacing frequency of electrical stimulation in cardiac tissue strongly influences the occurrence of early afterdepolarizations (EADs), which are small voltage oscillations that appear during the plateau phase of the cardiac action potential and can trigger life‑threatening arrhythmias. Using a minimal three‑dimensional model of a ventricular myocyte that includes the membrane voltage V, the activation gate n of the delayed‑rectifier K⁺ current, and the inactivation gate h of the L‑type Ca²⁺ current, the authors explore the dynamics under periodic square‑pulse stimulation of varying cycle length (PCL).
Previous analyses treated V and n as fast variables and h as the sole slow variable (a 1‑slow/2‑fast split). That approach identified a delayed subcritical Hopf bifurcation of the fast subsystem as the origin of EADs, but it could not predict how many EADs would appear or why their number depends on pacing rate. The present study adopts the opposite decomposition: V is the only fast variable, while n and h are both slow. This 2‑slow/1‑fast split reveals a three‑timescale structure (fast voltage ≈ 5 ms, n ≈ 80 ms, h ≈ 300 ms) amenable to geometric singular perturbation theory (GSPT).
In the singular limit (ε → 0) the fast subsystem reduces to dv/dt = f(V,n,h) with n and h frozen, defining a critical manifold C₀ given by f(V,n,h)=0. C₀ is an S‑shaped surface that folds at two points, creating a folded‑node singularity. Near this folded node, trajectories can follow canard solutions that spend a long time near the repelling sheet of C₀ before jumping back to the attracting sheet. These canard excursions correspond precisely to the observed EADs.
The folded‑node geometry provides a clear rule for the number of EADs generated by a single stimulus: it depends on where the stimulus injects the trajectory into the “singular funnel” surrounding the folded node. Low pacing frequencies (large PCL) allow the slow variables to relax fully toward the stable branch of C₀ before the next stimulus, so the stimulus lands deep inside the funnel, producing multiple rotations (typically 2–3 EADs). High frequencies (short PCL) leave the system near the funnel’s edge; the stimulus only grazes the funnel, yielding at most one or no EAD. At intermediate frequencies the stimulus repeatedly lands near the funnel boundary, causing the depth of entry to alternate from beat to beat. This produces alternating patterns such as 1 2 1 3 (one action potential with two EADs followed by one with three) and more complex “dynamical chaos” observed experimentally.
Parameter sensitivity analysis shows that decreasing the K⁺ conductance g_K (or pharmacologically blocking K⁺ channels) and increasing the Ca²⁺ conductance g_Ca shift the folded node and enlarge the funnel, thereby facilitating EAD formation. This explains why hypokalemia and K⁺‑channel blockers are pro‑arrhythmic. Similarly, increasing membrane capacitance C_m enlarges the small parameter ε, strengthening the timescale separation and making the canard mechanism more robust.
A key contribution is the derivation of an explicit formula, based on folded‑node theory, that predicts the maximal number of EADs possible for any given set of conductances and pacing interval, without the need for time‑consuming numerical simulations. This bridges the gap between biophysical intuition (ion‑channel currents) and rigorous dynamical‑systems analysis, offering a practical tool for drug development and for designing pacing protocols that avoid arrhythmogenic EADs.
In summary, the authors demonstrate that EADs are not merely a consequence of altered ionic currents but are organized by a folded‑node canard structure in the three‑dimensional phase space of the minimal cardiac model. This geometric insight explains the frequency‑dependent propensity for EADs, the emergence of alternans and chaotic patterns at intermediate pacing rates, and the impact of pharmacological or electrolyte perturbations. The work provides a unified, mathematically grounded framework that complements traditional electrophysiological explanations and opens new avenues for controlling arrhythmia risk.
Comments & Academic Discussion
Loading comments...
Leave a Comment