Multicellular rosettes drive fluid-solid transition in epithelial tissues
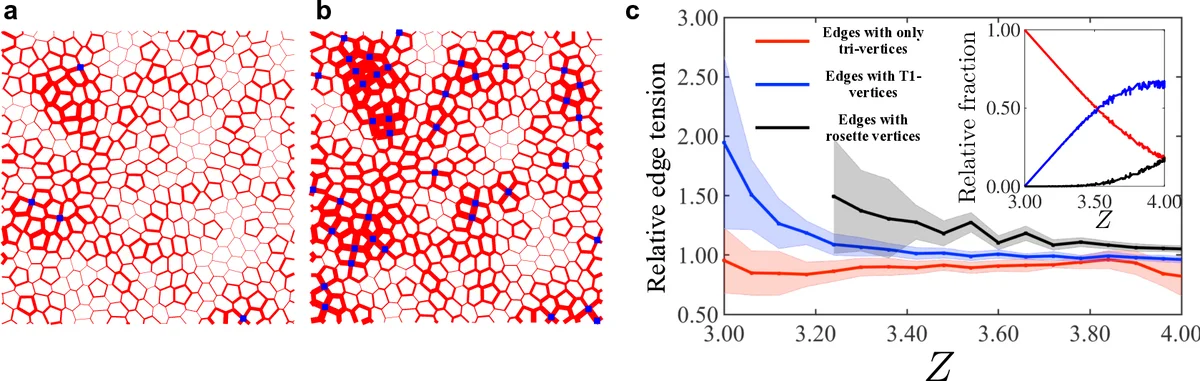
Models for confluent biological tissues often describe the network formed by cells as a triple-junction network, similar to foams. However, higher order vertices or multicellular rosettes are prevalent in developmental and {\it in vitro} processes and have been recognized as crucial in many important aspects of morphogenesis, disease, and physiology. In this work, we study the influence of rosettes on the mechanics of a confluent tissue. We find that the existence of rosettes in a tissue can greatly influence its rigidity. Using a generalized vertex model and effective medium theory we find a fluid-to-solid transition driven by rosette density and intracellular tensions. This transition exhibits several hallmarks of a second-order phase transition such as a growing correlation length and a universal critical scaling in the vicinity a critical point. Further, we elucidate the nature of rigidity transitions in dense biological tissues and other cellular structures using a generalized Maxwell constraint counting approach. This answers a long-standing puzzle of the origin of solidity in these systems.
💡 Research Summary
This paper investigates how multicellular rosettes—vertices where four or more epithelial cells meet—affect the mechanical rigidity of confluent tissues. Traditional vertex models treat epithelial sheets as networks of three‑fold junctions, analogous to soap foams, and predict a fluid‑to‑solid transition when the dimensionless target shape index (p_0=P_0/\sqrt{A_0}) exceeds a critical value (p_0^*\approx3.81). However, many developmental and in‑vitro systems display higher‑order vertices (T1‑junctions and rosettes) that are not captured by these models.
To incorporate rosettes, the authors extend the generic vertex model with an energy functional
(U=\sum_{\alpha}
Comments & Academic Discussion
Loading comments...
Leave a Comment