Ultrasensitive and highly accurate long-range surface plasmon resonance biosensors based on two-dimensional transition metal dichalcogenides
Two-dimensional transition metal dichalcogenides (TMDCs), as promising alternative plasmonics supporting materials to graphene, exhibit potential applications in sensing. Here, we propose an ultrasensitive, accurate long-range surface plasmon resonance (LRSPR) imaging biosensor with two-dimensional TMDC layers, which shows higher detection accuracy than that of conventional SPR biosensor. It is found that the imaging sensitivity of the proposed LRSPR biosensor can be enhanced by the integration of TMDC layers, which is different from the previous graphene-based LRSPR or SPR imaging sensor, whose imaging sensitivity usually decreases with the number of graphene layers. The sensitivity enhancement or degradation effect for the proposed chalcogenide-cytop-gold-TMDCs based biosensor depends on the thickness of gold thin film and cytop layer. Imaging sensitivity of more than 4000 $\text{RIU}^{-1}$ can be obtained with a high detection accuracy of more than 120 $\text{deg}^{-1}$. We expect that the proposed TMDCs mediated LRSPR imaging sensor could provide potential applications in chemical sensing and biosensing for a highly sensitive and accurate simultaneous detection of multiple biomolecular interactions.
💡 Research Summary
The manuscript presents a novel long‑range surface plasmon resonance (LRSPR) imaging biosensor that incorporates two‑dimensional transition metal dichalcogenide (TMDC) layers to achieve ultra‑high sensitivity and detection accuracy. Conventional surface plasmon resonance (SPR) sensors, typically operated in the Kretschmann configuration, rely on the excitation of surface plasmon waves (SPWs) at a metal–dielectric interface. While SPR offers high refractive index (RI) sensitivity, its angular interrogation mode suffers from a broad reflectance curve, limiting detection accuracy (DA) and preventing simultaneous monitoring of multiple biomolecular interactions. Imaging SPR overcomes the parallel‑monitoring limitation but still inherits the broad resonance linewidth of conventional SPR.
To address these drawbacks, the authors adopt a LRSPR architecture. In LRSPR, a thin gold (Au) film (15–30 nm) is sandwiched between a high‑index 2S2G glass prism and a low‑index Cytop layer (n≈1.3395). This configuration supports two coupled SPWs—one at the Au–Cytop interface and another at the Au–sensing‑layer interface. Their strong coupling yields a much narrower reflectance dip (small full‑width at half‑maximum, FWHM) and a deeper evanescent field penetration (≈545 nm versus ≈189 nm for conventional SPR). Consequently, both the angular sensitivity and the imaging sensitivity improve, and the DA—defined as the reciprocal of FWHM—rises dramatically (from <1 deg⁻¹ to >7 deg⁻¹).
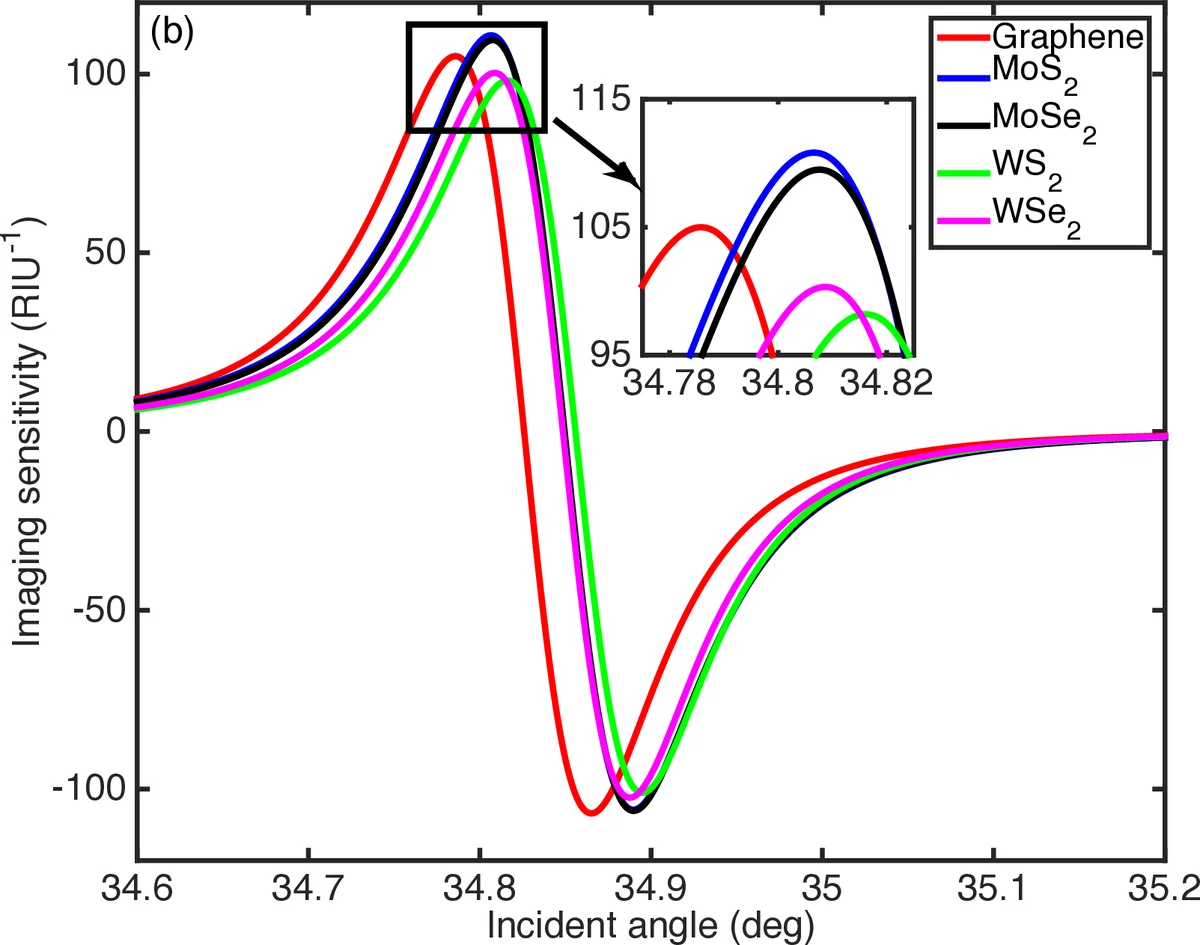
The key innovation lies in coating the Au surface with monolayer or few‑layer TMDCs (MoS₂, MoSe₂, WS₂, WSe₂). TMDCs possess high carrier mobility and strong light‑matter interaction, enabling efficient charge transfer to the Au film and thereby enhancing the local electric field at the sensing interface. Their complex refractive indices at λ = 633 nm (e.g., MoS₂: 5.0805 + i 1.1723) introduce modest absorption; when limited to a single or few layers (thickness 0.65–0.80 nm), this loss is outweighed by the field enhancement. The authors systematically explore the impact of Au film thickness (d_Au) and Cytop thickness (d_cytop) on sensor performance. Optimal performance is achieved for d_Au≈15 nm and d_cytop≈1.0–1.5 µm, where imaging sensitivities exceed 4000 RIU⁻¹ and DA surpasses 120 deg⁻¹.
Comparative simulations reveal that TMDC‑based LRSPR sensors outperform graphene‑based counterparts. While graphene layers typically reduce imaging sensitivity as their number increases (due to added loss), certain TMDC configurations actually increase sensitivity with additional layers, provided the metal and dielectric thicknesses are appropriately tuned. However, beyond a few layers, the imaginary part of the TMDC refractive index leads to increased plasmon damping, causing both sensitivity and DA to decline. Among the TMDCs studied, tungsten‑based WS₂ and WSe₂ deliver the highest sensitivities (≈1200 RIU⁻¹) for specific thickness combinations, whereas molybdenum‑based MoS₂ and MoSe₂ achieve ≈935–960 RIU⁻¹. The best overall sensor—MoS₂‑Au LRSPR—reaches an imaging sensitivity of 110.9 RIU⁻¹ with a DA of 7.10 deg⁻¹, representing a more than seven‑fold improvement over conventional SPR.
The authors also discuss practical implications. A typical biomolecular binding event changes the sensing layer RI by ~0.001, which translates into an angular shift of 0.1–0.2 deg in the LRSPR configuration—significantly larger than in conventional SPR. In imaging mode, the reflectance change per RI unit (dR/dn) exceeds 4000 RIU⁻¹, enabling real‑time, label‑free monitoring of multiple interactions across a sensor surface. The paper concludes that the Au‑Cytop‑TMDC trilayer design synergistically combines the narrow resonance of LRSPR with the field‑enhancing properties of TMDCs, delivering a platform suitable for high‑throughput chemical and biosensing applications. Future work is suggested to validate the theoretical predictions with experimental biomolecule assays, assess temperature and pH stability, and integrate the sensor with microfluidic delivery systems for practical deployment.
Comments & Academic Discussion
Loading comments...
Leave a Comment