Determination of Personalized Asthma Triggers from Evidence based on Multimodal Sensing and Mobile Application
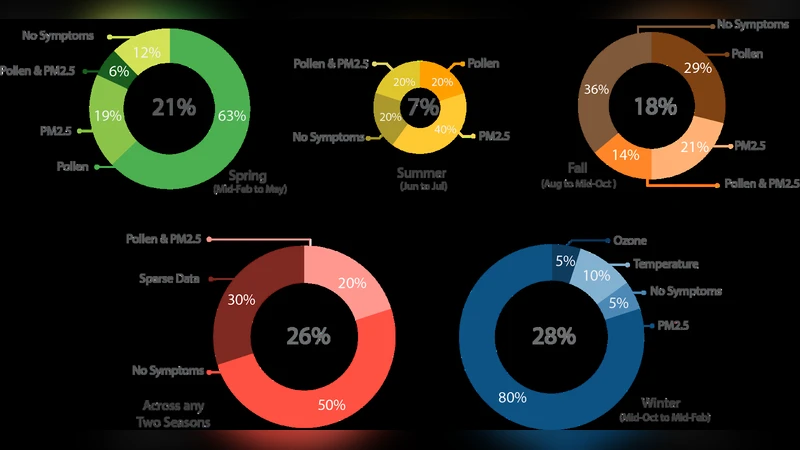
Objective: Asthma is a chronic pulmonary disease with multiple triggers manifesting as symptoms with various intensities. This paper evaluates the suitability of long-term monitoring of pediatric asthma using diverse data to qualify and quantify triggers that contribute to the asthma symptoms and control to enable a personalized management plan. Materials and Methods: Asthma condition, environment, and adherence to the prescribed care plan were continuously tracked for 97 pediatric patients using kHealth-Asthma technology for one or three months. Result: At the cohort level, among 21% of the patients deployed in spring, 63% and 19% indicated pollen and Particulate Matter (PM2.5), respectively, as the major asthma contributors. Of the 18% of the patients deployed in fall, 29% and 21% found pollen and PM2.5 respectively, to be the contributors. For the 28% of the patients deployed in winter, PM2.5 was identified as the major contributor for 80% of them. One patient across each season has been chosen to explain the determination of personalized triggers by observing correlations between triggers and asthma symptoms gathered from anecdotal evidence. Discussion and Conclusion: Both public and personal health signals including compliance to prescribed care plan have been captured through continuous monitoring using the kHealth-Asthma technology which generated insights on causes of asthma symptoms across different seasons. Collectively, they can form the underlying basis for personalized management plan and intervention. KEYWORDS: Personalized Digital Health, Medical Internet of Things, Pediatric Asthma Management, Patient Generated Health Data, Personalized Triggers, Telehealth,
💡 Research Summary
The paper presents a comprehensive study on personalized asthma trigger identification using the kHealth‑Asthma platform, which integrates multimodal sensing, mobile self‑reporting, and public environmental data. A cohort of 97 pediatric asthma patients was continuously monitored for either one or three months, during which three streams of data were collected: (1) symptom diaries and medication adherence entered via a smartphone app, (2) real‑time indoor and outdoor environmental measurements (temperature, humidity, PM2.5, PM10, ozone, and pollen counts) captured by wearable and portable sensors, and (3) supplemental public data from government air‑quality stations and pollen forecasting services. All data were transmitted securely over TLS, stored in a cloud‑based data lake, and pre‑processed through missing‑value imputation, time‑synchronization, and normalization to create a unified analytical dataset.
At the cohort level, the authors performed descriptive statistics and correlation analyses to reveal seasonal patterns in trigger prevalence. In the spring cohort (21 % of participants), 63 % identified pollen and 19 % identified PM2.5 as the dominant contributors. In the fall cohort (18 % of participants), pollen accounted for 29 % and PM2.5 for 21 % of reported triggers. In the winter cohort (28 % of participants), PM2.5 emerged as the primary trigger for 80 % of patients, reflecting the near‑absence of pollen during cold months. Pearson correlation coefficients and cross‑correlation functions demonstrated that elevated pollen levels (≥ 50 grains/m³) were associated with a 1.5‑point increase in cough scores after a one‑day lag, while PM2.5 concentrations exceeding 35 µg/m³ corresponded to a 1.8‑point rise in overall symptom scores with no lag.
Beyond population‑level insights, the study emphasizes individualized trigger detection. For each patient, the authors constructed a correlation matrix linking daily environmental variables to symptom scores over at least 30 consecutive days. The top two to three variables with the highest and statistically significant correlations were selected, and lag analysis pinpointed the temporal offset at which the environmental exposure most strongly predicted symptom exacerbation. As an illustrative case, a 9‑year‑old boy monitored in spring showed the strongest correlation between pollen and cough (r = 0.62, 1‑day lag) and between PM2.5 and shortness of breath (r = 0.48, 0‑day lag). Based on these findings, the system generated personalized push notifications warning the patient’s family when pollen or PM2.5 approached the individual’s threshold, and recommended pre‑emptive inhaler use. Similar case studies for each season are presented, demonstrating a “data‑driven trigger identification → personalized alert → symptom mitigation” workflow.
The technical architecture of kHealth‑Asthma includes a user‑friendly mobile interface for symptom entry, an automated alert engine that triggers push notifications when environmental readings exceed patient‑specific thresholds, and a backend analytics pipeline built on Apache Spark and Python Pandas for daily batch processing. Security and privacy were addressed through token‑based authentication, data anonymization, and compliance with GDPR‑like principles.
In the discussion, the authors argue that continuous, multimodal monitoring transforms traditionally subjective trigger identification into an objective, quantifiable process. This enables clinicians to verify adherence (through inhaler usage logs), anticipate exacerbations, and intervene remotely before severe attacks occur. The study also acknowledges limitations: the monitoring period (maximum three months) may not capture long‑term seasonal variability; occasional data loss due to connectivity or device battery issues required imputation that could introduce bias; and the reliance on correlation analysis does not establish causality.
Future work is outlined to address these gaps. The authors plan to develop predictive machine‑learning models (e.g., Long Short‑Term Memory networks, Gradient Boosting Machines) that will generate real‑time risk scores based on the integrated data streams. They also intend to conduct randomized controlled trials to evaluate whether personalized alerts reduce emergency visits, improve lung function, and increase medication adherence.
In conclusion, the research demonstrates that a digital health ecosystem combining multimodal environmental sensing, patient‑generated health data, and mobile communication can accurately identify personalized asthma triggers in pediatric patients across different seasons. By translating these insights into actionable, season‑aware management plans, the kHealth‑Asthma platform offers a scalable blueprint for precision asthma care and illustrates the broader potential of Internet‑of‑Things–enabled telehealth solutions in chronic disease management.
Comments & Academic Discussion
Loading comments...
Leave a Comment