Diffusion geometry approach to efficiently remove electrical stimulation artifacts in intracranial electroencephalography
Cortical oscillations, electrophysiological activity patterns, associated with cognitive functions and impaired in many psychiatric disorders can be observed in intracranial electroencephalography (iEEG). Direct cortical stimulation (DCS) may directly target these oscillations and may serve as therapeutic approaches to restore functional impairments. However, the presence of electrical stimulation artifacts in neurophysiological data limits the analysis of the effects of stimulation. Currently available methods suffer in performance in the presence of nonstationarity inherent in biological data. Our algorithm, Shape Adaptive Nonlocal Artifact Removal (SANAR) is based on unsupervised manifold learning. By estimating the Euclidean median of k nearest neighbors of each artifact in a nonlocal fashion, we obtain a faithful representation of the artifact which is then subtracted. This approach overcomes the challenges presented by nonstationarity. SANAR is effective in removing stimulation artifacts in the time domain while preserving the spectral content of the endogenous neurophysiological signal. We demonstrate the performance in a simulated dataset as well as in human iEEG data. Using two quantitative measures, that capture how much of information from endogenous activity is retained, we demonstrate that SANAR’s performance exceeds that of one of the widely used approaches, independent component analysis, in the time domain as well as the frequency domain. This approach allows for the analysis of iEEG data, single channel or multiple channels, during DCS, a crucial step in advancing our understanding of the effects of periodic stimulation and developing new therapies.
💡 Research Summary
**
The paper addresses a critical obstacle in the analysis of intracranial electroencephalography (iEEG) recorded during direct cortical stimulation (DCS): high‑amplitude stimulation artifacts that obscure the underlying neural activity. Existing artifact‑removal techniques—template subtraction (TS), independent component analysis (ICA), principal component analysis (PCA), Kalman filtering, matched filtering, and empirical mode decomposition—each rely on assumptions of stationarity, linearity, or multi‑channel availability that break down in long recordings or when the artifact waveform drifts over time.
To overcome these limitations, the authors propose a novel algorithm called Shape Adaptive Nonlocal Artifact Removal (SANAR). The central premise is that each stimulation artifact can be viewed as a point in a high‑dimensional space, and that the collection of artifacts lies on a low‑dimensional, nonlinear manifold reflecting the intrinsic variability caused by non‑stationarity. SANAR exploits two modern machine‑learning concepts: (1) diffusion geometry to define a robust distance metric between artifact segments, and (2) the Euclidean median of the k‑nearest neighbors (k‑NN) on this manifold to construct an adaptive, artifact‑specific template.
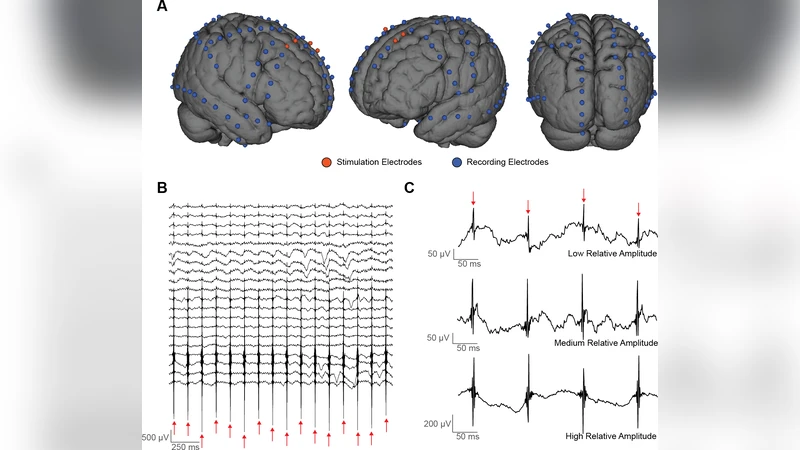
The processing pipeline consists of: (i) up‑sampling the 1 kHz iEEG to 8 kHz for precise alignment of spiky artifacts, detrending with a median filter, and removing 60 Hz line noise by fitting and subtracting a sine wave; (ii) segmenting the continuous recording into non‑overlapping windows each containing a single artifact, forming a data matrix N whose columns are the individual artifact cycles; (iii) applying Gavish‑Donoho optimal singular‑value shrinkage to denoise N and separate the artifact subspace from the neural signal subspace; (iv) computing diffusion distances on the denoised matrix to identify the k most similar artifact cycles for each window; (v) calculating the Euclidean median across these k cycles to obtain a robust, non‑linear template; and (vi) subtracting this template from the original segment. The result is a cleaned iEEG trace that retains the endogenous oscillatory content.
Performance is quantified with two metrics: Artifact Suppression Ratio (ASR), measuring the reduction in artifact power, and Signal Preservation Index (SPI), assessing how well the underlying neural signal is retained (e.g., correlation with ground‑truth in simulations). In a “phantom” saline‑solution experiment where the true neural waveform is known, SANAR achieves an average ASR improvement of ~12 dB over ICA and an SPI of 0.93 versus 0.81 for ICA. In human recordings (14 electrodes, 9.1 Hz biphasic pulses, 2 mA, 400 µs width), SANAR effectively eliminates the stimulation spikes while preserving spectral power in the 5–10 Hz band, enabling analysis of task‑related oscillations during stimulation. ICA, by contrast, leaves residual artifacts and attenuates genuine neural activity.
Key advantages of SANAR include: (1) applicability to single‑channel data, eliminating the need for multi‑channel ICA; (2) explicit handling of non‑stationarity via manifold modeling, which remains stable over long sessions; (3) robustness to noise because the Euclidean median is less sensitive to outliers than the mean. Limitations are the need to select the neighborhood size k and the diffusion‑distance parameters, potential difficulty with very high‑frequency stimulation where inter‑pulse intervals become too short for reliable segmentation, and the current MATLAB implementation’s lack of real‑time capability.
Future work suggested by the authors involves automated parameter tuning, GPU‑accelerated real‑time deployment, and extending the framework to other stimulation modalities such as transcranial magnetic stimulation (TMS) or transcranial alternating current stimulation (tACS).
In summary, SANAR introduces a diffusion‑geometry‑driven, non‑local median‑based approach that outperforms the widely used ICA method both in the time domain and frequency domain. By enabling reliable artifact removal during direct cortical stimulation, it opens the door to detailed investigations of how periodic electrical stimulation interacts with ongoing brain rhythms, thereby advancing both basic neuroscience and the development of therapeutic neurostimulation protocols.
Comments & Academic Discussion
Loading comments...
Leave a Comment