Shared features of endothelial dysfunction between sepsis and its preceding risk factors (aging and chronic disease)
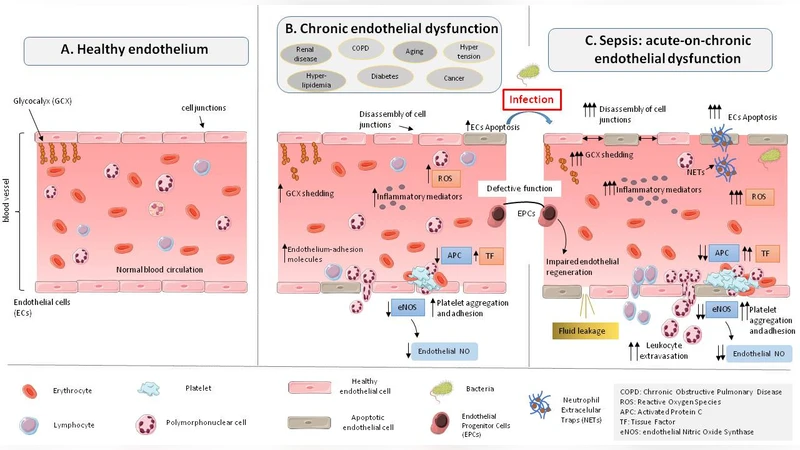
Acute vascular endothelial dysfunction is a central event in the pathogenesis of sepsis,increasing vascular permeability, promoting activation of the coagulation cascade, tissue edema and compromising perfusion of vital organs. Aging and chronic diseases(hypertension,dyslipidaemia,diabetes mellitus,chronic kidney disease,cardiovascular disease,cerebrovascular disease, chronic pulmonary disease,liver disease or cancer)are recognized risk factors for sepsis. In this article we review the features of endothelial dysfunction shared by sepsis,aging and the chronic conditions preceding this disease. Clinical studies and review articles on endothelial dysfunction associated to sepsis,aging and chronic diseases published in PubMed were considered. The main features of endothelial dysfunction shared by sepsis,aging and chronic diseases were 1.increased oxidative stress and systemic inflammation, 2.glycocalyx degradation and shedding, 3.disassembly of intercellular junctions,endothelial cell death,blood tissue barrier disruption, 4.enhanced leukocyte adhesion and extravasation, 5.induction of a pro-coagulant and anti-fibrinolytic state. In addition,chronic diseases impair the mechanisms of endothelial reparation. In conclusion,sepsis,aging and chronic diseases induce similar features of endothelial dysfunction. The potential contribution of the pre-existent degree of endothelial dysfunction to sepsis pathogenesis deserves to be further investigated
💡 Research Summary
This narrative review examines the overlapping mechanisms of endothelial dysfunction (ED) that occur in sepsis, aging, and a broad spectrum of chronic diseases (including hypertension, dyslipidemia, diabetes, chronic kidney disease, cardiovascular disease, cerebrovascular disease, chronic pulmonary disease, liver disease, and cancer). The authors performed a PubMed search focused on articles published within the last ten years, prioritizing studies that investigated endothelial pathology in these contexts. They identified five core pathological features shared across all conditions: (1) heightened oxidative stress and systemic inflammation, (2) degradation and shedding of the endothelial glycocalyx (GCX), (3) disassembly of intercellular junctions together with endothelial cell death leading to barrier disruption, (4) increased leukocyte adhesion and extravasation, and (5) a shift toward a pro‑coagulant and anti‑fibrinolytic state.
In sepsis, a “molecular storm” of pathogen‑associated molecular patterns (PAMPs), damage‑associated molecular patterns (DAMPs), cytokines, bradykinin, platelet‑activating factor, VEGF, fibrin‑degradation products, and reactive oxygen species (ROS) initiates and amplifies these five mechanisms. ROS and inducible nitric oxide synthase (iNOS) generate excessive nitric oxide, while endothelial nitric oxide synthase (eNOS) activity falls, impairing vasodilation. The inflammatory milieu triggers GCX shedding (elevated plasma syndecan‑1, heparan sulfates), exposing adhesion molecules and facilitating leukocyte tethering. NETs released by activated neutrophils further degrade the glycocalyx, proteolyze junctional proteins, and directly kill endothelial cells. The resulting loss of VE‑cadherin, claudins, and occludins creates intercellular gaps, increasing vascular permeability and tissue edema. Concurrently, up‑regulation of P‑selectin, E‑selectin, ICAM‑1, and VCAM‑1 promotes leukocyte adhesion and migration, while tissue factor (TF) expression, reduced thrombomodulin, and diminished protein C receptor activity shift the coagulation balance toward thrombosis. NETs also act as scaffolds for fibrin deposition, reinforcing microvascular clot formation.
Aging and chronic diseases reproduce these same pathways, albeit in a chronic, low‑grade fashion. Age‑related increases in NADPH oxidase activity, mitochondrial ROS production, and uncoupled eNOS generate persistent oxidative stress. Hypertension, diabetes, and hyperlipidemia are linked to reduced GCX thickness and increased shedding of heparan sulfate. Chronic kidney disease, COPD, liver cirrhosis, and cancer similarly impair glycocalyx integrity through inflammatory cytokines, oxidative damage, and direct endothelial injury. Senescent endothelial cells display a pro‑oxidant phenotype, down‑regulate tight‑junction proteins, and become more permeable. Hyperglycemia and oxidized LDL disrupt adherens junctions via protein kinase C‑β‑mediated VE‑cadherin phosphorylation. Chronic inflammatory states (e.g., in atherosclerosis, hypertension, COPD) elevate NF‑κB activity, driving over‑expression of adhesion molecules and facilitating leukocyte recruitment. Moreover, chronic conditions diminish the number and function of endothelial progenitor cells (EPCs), hampering endothelial repair and regeneration.
Epidemiological data cited (Rhee et al., 173,690 patients; Donnelly et al., 1,080 patients) reveal that sepsis patients are typically elderly (mean age ≈ 67–70 years) and carry multiple comorbidities (≥ 97% have at least one chronic disease). This suggests that acute sepsis often strikes an endothelium already compromised by age‑related or disease‑related dysfunction, potentially amplifying vascular injury and organ failure.
The review highlights gaps in the current literature: most evidence is observational or derived from animal models, and quantitative comparisons of endothelial injury across disease states are scarce. The authors call for prospective cohort studies that correlate circulating endothelial injury biomarkers (e.g., syndecan‑1, angiopoietin‑2, soluble thrombomodulin) with sepsis incidence and outcomes, as well as interventional trials testing glycocalyx‑protective agents, antioxidants, or EPC‑enhancing therapies. Preventive strategies targeting endothelial health in older adults and patients with chronic diseases could reduce sepsis susceptibility and improve survival.
In summary, sepsis, aging, and chronic diseases converge on a common set of endothelial pathophysiological processes. Understanding and therapeutically targeting these shared mechanisms may offer a unified approach to mitigate sepsis severity, especially in high‑risk, elderly, and comorbid populations.
Comments & Academic Discussion
Loading comments...
Leave a Comment