Accurate determination of the absolute 3He/4He ratio of a synthesized helium standard gas (Helium Standard of Japan, HESJ): Towards revision of the atmospheric 3He/4He ratio
The helium standard of Japan, referred to as HESJ, is an inter-laboratory standard for the 3He/4He ratio. While the ratio of 3He and 4He of the HESJ was previously determined by a relative comparison to atmospheric helium, the absolute value of the 3He/4He ratio of the HESJ has not been directly determined yet. Therefore, it relies on the early measurements of that of atmospheric helium. The accuracy of the absolute 3He/4He ratios of the atmosphere and other working standards including HESJ is crucial in some applications of helium isotopes, such as tritium-3He dating, surface-exposure age determination based on cosmogenic 3He, and the accurate measurement of the neutron lifetime. In this work, new control samples of helium gases with 3He/4He ratios of 14, 28, and 42 ppm were fabricated with accuracy of 0.25-0.38% using a gas-handling system for a neutron lifetime experiment at Japan Proton Accelerator Research Complex (J-PARC). The relative 3He/4He ratios of these samples and the HESJ were measured using a magnetic-sector-type, single-focusing, noble gas mass spectrometer with a double collector system. As a result, the absolute 3He/4He ratio of the HESJ was determined as 27.36 +/- 0.11 ppm. The atmospheric 3He/4He ratio was determined as 1.340 +/- 0.006 ppm, based on this work.
💡 Research Summary
The paper presents a rigorous determination of the absolute 3He/4He ratio of the Japanese helium standard gas (HESJ) and, using this result, a revised value for the atmospheric 3He/4He ratio. Historically, the HESJ ratio has been known only through relative comparison to atmospheric helium, whose own absolute ratio has been subject to debate and potential temporal or spatial variability. To eliminate this dependence, the authors directly measured the absolute ratio of HESJ with a high‑precision approach.
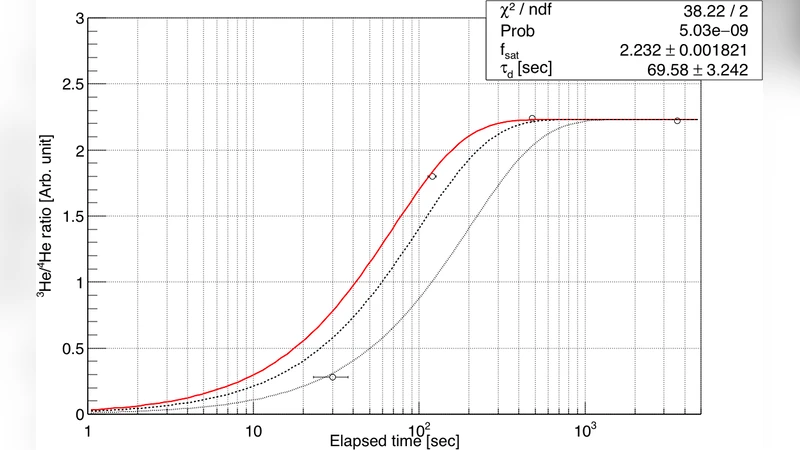
First, three control gases with nominal 3He/4He ratios of 14 ppm, 28 ppm, and 42 ppm were fabricated. The authors employed a gas‑handling system originally built for a neutron‑lifetime experiment at J‑PARC. The system consists of four defined volumes (V0–V3) connected by stainless‑steel tubing and Swagelok valves. By using a gas‑expansion method, the authors accurately diluted isotopically pure 3He with 4He. Volume ratios between the compartments were measured by pressure‑change experiments, corrected for non‑ideal gas behavior using the second virial coefficient of helium (11.83 cm³ mol⁻¹) and for temperature effects (Baratron gauge operated at 45 °C). The resulting volume ratios (A = 0.30783, D = 0.006139) have uncertainties of 0.02–0.22 %. These ratios, together with precisely measured initial pressures (P1–P3), allowed the preparation of the three control samples with absolute 3He/4He ratios of 14.01 ± 0.05 ppm, 28.05 ± 0.08 ppm, and 42.01 ± 0.11 ppm, corresponding to relative uncertainties of 0.38 %, 0.30 %, and 0.25 % respectively. Diffusion times were experimentally verified (τd = 70 ± 3 s) and mixing was allowed to proceed for >30 min to ensure homogeneity.
Second, the absolute ratio of HESJ was measured using a magnetic‑sector, single‑focusing noble‑gas mass spectrometer equipped with a dual‑collector system (electron‑multiplier ion counter for 3He and a Faraday cup with 10¹⁰ Ω feedback for 4He). The instrument operates at a fixed magnetic field where the 3He and 4He peaks coincide, minimizing field‑drift effects. Each sample was measured for 400 s, and the sequence of measurements alternated between HESJ and each control gas to cancel temporal drifts. The pressure range in the ion source (3–7 × 10⁻⁶ Pa) is well below the level where pressure‑dependent bias (2.4 % decrease above 6.4 × 10⁻⁴ Pa) becomes significant; the maximum pressure difference among samples would cause only a 0.015 % effect on the ratio. A “memory effect” (slow release of previously adsorbed gas) was observed but contributed less than 0.01 ppm over the typical 1000 s measurement, negligible compared with the 27 ppm signal.
Five replicate measurements yielded a mean HESJ ratio of 27.36 ± 0.11 ppm, corresponding to a combined relative uncertainty of 0.40 %. By anchoring this value to the previously accepted atmospheric ratio, the authors derived an absolute atmospheric 3He/4He ratio of 1.340 ± 0.006 ppm (1.340 × 10⁻⁶). This value lies within the range of earlier determinations (1.3–1.4 × 10⁻⁶) but with an order‑of‑magnitude improvement in precision.
The significance of these results extends to several fields that rely on precise helium isotope ratios. In tritium‑3He dating, the age calculation depends directly on the absolute amount of 3He; a 0.4 % uncertainty in the standard translates to a comparable uncertainty in groundwater or seawater ages. Cosmogenic 3He exposure dating of minerals also requires an absolute calibrator to convert measured 3He concentrations into exposure ages, especially when production rates are expressed per unit 3He. Finally, the J‑PARC neutron‑lifetime experiment uses a mixture of 3He, 4He, and CO₂ as a detection medium; the neutron flux is inferred from the 3He(n,p) reaction rate, which is proportional to the absolute 3He number density. The 0.3 % control over 3He density achieved in the gas‑handling system, combined with the newly established absolute HESJ ratio, reduces a major systematic uncertainty in the neutron‑lifetime measurement.
In summary, the authors have demonstrated a complete workflow—from the fabrication of high‑accuracy control gases to the dual‑collector mass‑spectrometric determination—that yields the first direct, high‑precision absolute 3He/4He ratio for the HESJ standard. By propagating this result to atmospheric helium, they provide a robust reference that can be adopted internationally, improving the reliability of helium‑isotope based geochronology, cosmochemistry, and fundamental physics experiments. Future work may focus on inter‑laboratory comparisons using the newly defined standard and on extending the methodology to other noble‑gas isotopic systems.
Comments & Academic Discussion
Loading comments...
Leave a Comment