On the dynamic suction pumping of blood cells in tubular hearts
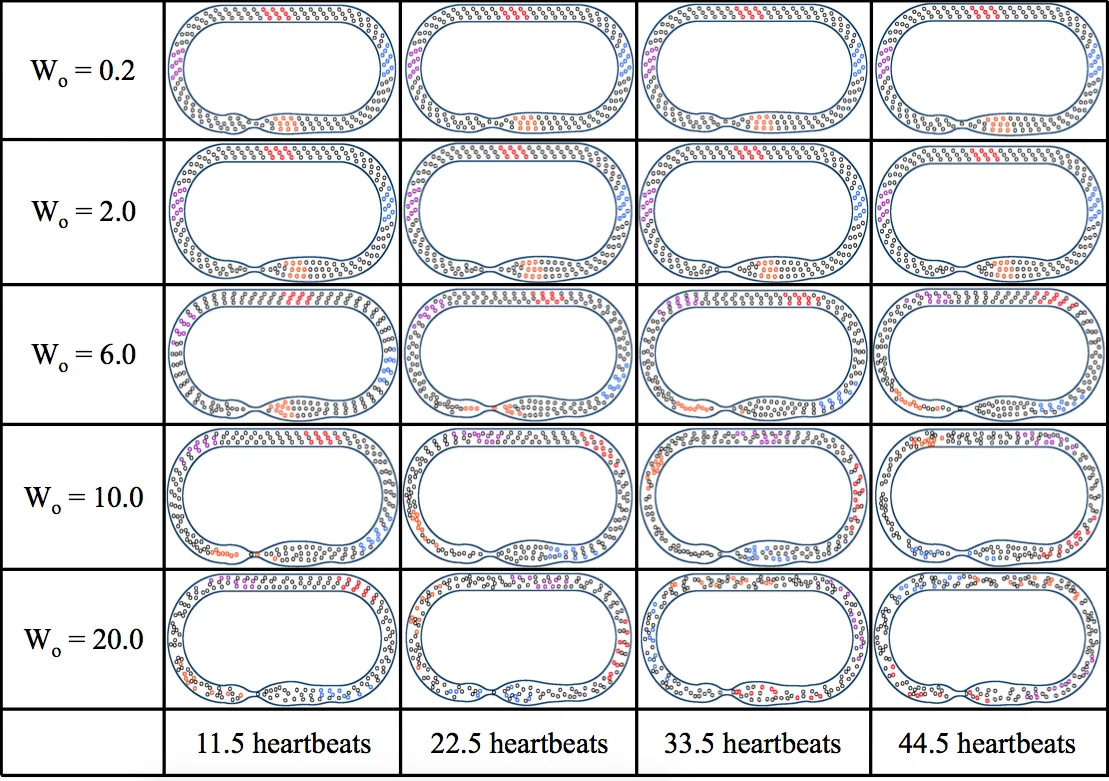
Around the third week after gestation in embryonic development, the human heart consists only of a valvless tube, unlike a fully developed adult heart, which is multi-chambered. At this stage in development, the heart valves have not formed and so net flow of blood through the heart must be driven by a different mechanism. It is hypothesized that there are two possible mechanisms that drive blood flow at this stage - Liebau pumping (dynamic suction pumping or valveless pumping) and peristaltic pumping. We implement the immersed boundary method with adaptive mesh refinement (IBAMR) to numerically study the effect of hematocrit on the circulation around a valveless. Both peristalsis and dynamic suction pumping are considered. In the case of dynamic suction pumping, the heart and circulatory system is simplified as a flexible tube attached to a relatively rigid racetrack. For some Womersley number (Wo) regimes, there is significant net flow around the racetrack. We find that the addition of flexible blood cells does not significantly affect flow rates within the tube for Wo $\leq$ 10. On the other hand, peristalsis consistently drives blood around the racetrack for all Wo and for all hematocrit considered.
💡 Research Summary
This paper investigates the fluid‑mechanical mechanisms that drive blood circulation in the early embryonic heart, which at about three weeks gestation consists of a simple, valveless tube rather than the multi‑chambered, valve‑equipped adult organ. Two competing hypotheses have been proposed for this stage: Liebau‑type dynamic suction pumping (DSP), also known as valveless pumping, and peristaltic pumping. While many previous analytical and experimental studies have examined these mechanisms, they have largely ignored the presence of blood cells, whose size (≈4 µm) is comparable to the tube diameter and whose volume fraction (hematocrit) can reach 40 % during development.
To address this gap, the authors employ the immersed boundary method (IBM) with adaptive mesh refinement (IBAMR) to perform two‑dimensional, fully coupled fluid‑structure simulations that explicitly resolve deformable blood cells as elastic spring networks. The computational domain is a closed “racetrack” geometry: two straight sections joined by two semicircular arcs, with a uniform tube diameter. The lower straight segment is made flexible (beams and springs) and is the only region where active actuation is applied. For the DSP case, a localized band of “muscle” springs (10 % of the straight length) is programmed to shorten and lengthen sinusoidally, thereby mimicking the off‑center contraction that characterizes Liebau pumps. For the peristaltic case, a traveling wave of prescribed contraction is imposed along the lower wall while the upper straight segment is allowed to expand, ensuring volume conservation in the closed loop.
Blood cells are modeled as circular particles of diameter d/5 (≈0.8 µm) linked by springs; they occupy volume fractions (hematocrit) ranging from 0 % to 80 % in 10 % increments. The fluid is incompressible with constant density and viscosity, and the Navier–Stokes equations are solved on a hierarchically refined Cartesian grid (finest spacing D/1024, where D is the domain length). The key nondimensional parameter explored is the Womersley number, Wo = √(ω ρ R²/µ), which measures the relative importance of unsteady inertia to viscous effects. Simulations span low‑Wo regimes (Wo ≤ 10, where viscous forces dominate) and higher Wo regimes (Wo > 10, where inertial effects become significant).
Results show that in the low‑Wo regime the presence of flexible blood cells does not appreciably alter the net volumetric flow produced by DSP; average flow rates remain essentially unchanged across all hematocrit levels. This suggests that, when viscous damping is strong, the additional drag introduced by cells is balanced by the same increase in shear, leading to a negligible net effect. By contrast, peristaltic pumping drives robust, unidirectional circulation for every Wo and hematocrit tested, with flow rates increasing markedly as Wo grows, reflecting the more efficient conversion of wave motion into bulk transport when inertia is significant.
The DSP mechanism proves highly sensitive to geometric and actuation parameters. Varying the length of the active region (Ls), its offset from the tube’s left end (LA), and the stiffness of the surrounding rigid sections changes the amplitude of reflected elastic waves and can even reverse flow direction. In certain configurations, especially at higher Wo, DSP yields substantial net flow, but only within a narrow window of design choices.
Overall, the study makes two major contributions. First, it delivers the first 2‑D IBM simulations that incorporate deformable blood cells into models of embryonic heart pumping, thereby bridging a critical gap between idealized fluid‑structure analyses and the physiological reality of cellular suspensions. Second, it provides a systematic, quantitative comparison of DSP and peristaltic pumping across a physiologically relevant range of Womersley numbers and hematocrits. The findings support the view that peristalsis is a more reliable and efficient pumping strategy for the embryonic tubular heart, yet they also demonstrate that DSP can sustain circulation under low‑frequency, viscously dominated conditions. These insights have practical implications for the design of micro‑fluidic valveless pumps, the engineering of tissue‑based heart models, and the broader understanding of how mechanical forces shape early cardiac development.
Comments & Academic Discussion
Loading comments...
Leave a Comment