Fingering instabilities in tissue invasion: an active fluid model
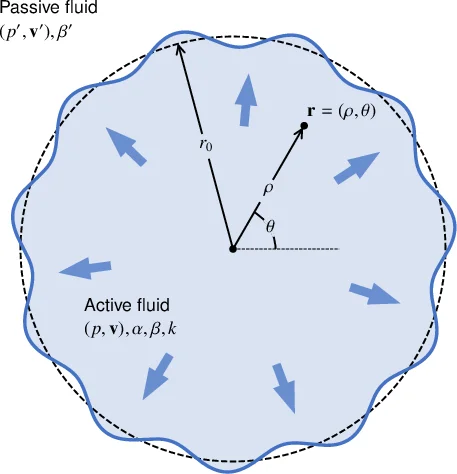
Metastatic tumors often invade healthy neighboring tissues by forming multicellular finger-like protrusions emerging from the cancer mass. To understand the mechanical context behind this phenomenon, we here develop a minimalist fluid model of a self-propelled, growing biological tissue. The theory involves only four mechanical parameters and remains analytically trackable in various settings. As an application of the model, we study the evolution of a 2D circular droplet made of our active and expanding fluid, and embedded in a passive non-growing tissue. This system could be used to model the evolution of a carcinoma in an epithelial layer. We find that our description can explain the propensity of tumor tissues to fingering instabilities, as conditioned by both the magnitude of active traction and the growth kinetics. We are also able to derive predictions for the tumor size at the onset of metastasis, and for the number of subsequent invasive fingers. Our active fluid model may help describe a wider range of biological processes, including wound healing and developmental patterning.
💡 Research Summary
This paper presents a minimalist continuum‐fluid framework to explain why invasive tumors develop multicellular finger‑like protrusions as they expand into surrounding healthy tissue. The authors model the tumor as an “active growing fluid” that both exerts a constant traction on the substrate (parameter α) and expands at a uniform volumetric growth rate k, while experiencing passive friction against the substrate (parameter β). The surrounding non‑malignant tissue is treated as a passive, incompressible fluid with its own friction coefficient β₀ and no growth. The governing equations are
∇p = –β v + α v|v|, ∇·v = k (active tumor)
∇p₀ = –β₀ v₀, ∇·v₀ = 0 (passive tissue)
together with continuity of normal velocity and a Laplace pressure jump across the interface, the latter involving the interfacial surface tension γ.
Using experimentally motivated estimates (k≈10⁻⁴ s⁻¹, β≈10¹⁵–10¹⁶ Pa·s·m⁻², α≈10⁸–10¹⁰ Pa·m⁻¹, γ≈10⁻³–10⁻² Pa·m), the authors introduce a characteristic length ℓ = (2γβ/k)¹⁄³ (≈10 µm) and a reference activity α* = βℓk (≈10⁷ Pa·m⁻¹) to nondimensionalize the problem.
A linear stability analysis is performed on a circular tumor of radius r₀ perturbed by azimuthal modes r(θ)=r₀+δrₙe^{inθ}. The growth rate σₙ of each mode n is derived analytically and shown to depend on the four key parameters (α, β, k, γ) as well as the viscosity ratio φ = β₀/β. When α = 0 the result reduces to the classic viscous fingering (Saffman–Taylor) dispersion relation, confirming consistency with previous work. For α > 0 the activity term adds a positive contribution to σₙ, especially for intermediate mode numbers (n≈5–10) when the tumor radius is on the order of a few ℓ. Consequently, active traction dramatically lowers the critical radius r_c at which the interface becomes unstable. The authors obtain a scaling law
r_c ≈ ℓ (α/α*)^{1/2} (γ/γ₀)^{1/3}
and predict the most unstable mode n_max from the maximum of σₙ. The expected number of fingers N≈n_max follows, yielding N≈4–12 for realistic parameter ranges, in agreement with observations of carcinoma and wound‑healing fronts.
The analysis also reveals that higher surface tension γ stabilizes the interface (larger r_c), while a larger viscosity ratio φ (more viscous surrounding tissue) suppresses the instability. These dependencies provide a mechanistic explanation for why certain tumors become highly invasive while others remain relatively smooth.
The paper discusses limitations: the model is two‑dimensional, assumes spatially uniform and time‑independent parameters, and neglects heterogeneity such as leader‑cell dynamics. Nevertheless, the framework is readily extensible to three dimensions, spatially varying α(r,t), or coupling to biochemical fields (e.g., nutrient diffusion). The authors suggest experimental validation using microfluidic tumor‑on‑a‑chip platforms or organoid cultures where traction forces and growth rates can be measured.
In summary, the work offers a concise yet powerful analytical description of tumor fingering, linking cellular activity, growth, friction, and interfacial tension. It provides quantitative predictions for the onset of metastasis (critical tumor size) and the number of invasive protrusions, thereby bridging biophysical theory with potential clinical relevance.
Comments & Academic Discussion
Loading comments...
Leave a Comment